International Journal of Cancer and Clinical Research
Second Line and Maintenance Therapy for Advanced Non-Small Cell Lung Cancer without Driver Mutation: An Evolving Paradigm
Stephanie Brulé* and Paul Wheatley-Price
The Ottawa Hospital Cancer Centre, Ottawa, Canada
*Corresponding author:
Stephanie Brulé, The Ottawa Hospital Cancer Centre, 501 Smyth Road, Ottawa, ONK1H 8L6, Canada, Tel: 6137227000, E-mail: sbrule@toh.on.ca
Int J Cancer Clin Res, IJCCR-3-055, (Volume 3, Issue 3), Review Article; ISSN: 2378-3419
Received: February 24, 2016 | Accepted: April 22, 2016 | Published: May 02, 2016
Citation: Brulé S, Wheatley-Price P (2016) Second Line and Maintenance Therapy for Advanced Non-Small Cell Lung Cancer without Driver Mutation: An Evolving Paradigm. Int J Cancer Clin Res 3:055. 10.23937/2378-3419/3/3/1055
Copyright: © 2016 Brulé S, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Introduction
Lung cancer is the most commonly diagnosed cancer amongst men and women combined, and is responsible for the greatest number of cancer-related deaths worldwide [1]. Non-small cell lung cancer (NSCLC) represents about 85% of all lung cancers, and is the umbrella term that includes the most common histological subtypes: adenocarcinoma, squamous cell carcinoma and large cell carcinoma. Due to the fact that it is frequently diagnosed at an advanced stage or recurs after potentially curative treatment, the five-year survival rate for all patients diagnosed with NSCLC is only 17% [2]. Therefore, at some point in their cancer course, even those initially treated with curative intent, most patients will be faced with a diagnosis of incurable cancer with the potential to undergo palliative systemic therapy.
Importantly, of all patients diagnosed with advanced NSCLC, a significant proportion remain untreated in a real world setting. However, with the advent of more effective and tolerable therapies, this proportion is becoming less and less. Depending upon the population and timeframe examined, 24%-55% of patients diagnosed with advanced NSCLC undergo first line systemic therapy. Then, there is a significant drop off rate with each subsequent line of therapy owing to rapid disease progression, decline in performance status, or toxicity from previous therapy. Only approximately only one third to one half of treated patients receive each subsequent line of therapy [3-6]. Further, the landscape of systemic therapy for advanced NSCLC has seen significant changes over the past two decades [7]. Standard first line therapy for patients without an identified actionable mutation (such as EGFR, ALK, or ROS1) consists of platinum doublet chemotherapy with or without an antiangiogenic biologic agent [8]. Treatment beyond first line therapy has been in a state of flux during recent times. Maintenance therapy has found a place within standard treatment plans, while targeted and immunotherapy are finding their way into second line therapy, where previously the limited options involved primarily single-agent cytotoxic chemotherapy.
Given the recent developments in maintenance and second line systemic therapy for advanced NSCLC, this article will present the current state of the evidence for disease without an identifiable driver mutation (Figure 1).
Maintenance Therapy
Historically, upon completion of 4-6 cycles of first line chemotherapy for advanced NSCLC, a patient would be monitored without therapy until disease progression, at which time second line therapy was initiated. However, in an effort to improve outcomes and maximize therapy with a potentially less toxic and tolerable single agent, a strategy of maintenance therapy has emerged.
There are two forms of maintenance therapy: switch maintenance and continuation maintenance. Switch maintenance entails introducing a new, non-cross-resistant agent immediately after completion of first line platinum doublet therapy. In continuation maintenance, administration of the non-platinum agent used in the first line setting is continued beyond the induction phase.
Biological and clinical rationales for maintenance therapy have been proposed [9]. Switch maintenance therapy leads to increased exposure of tumour cells to non-cross-resistant therapies. Therefore, this may lead to a decreased incidence of chemotherapy resistance and to increased antitumor effects. Further, switch maintenance therapy can act as an early form of second line therapy. As previously outlined, a relatively low proportion of patients receive second line therapy, and thus maintenance therapy leads to an increased number of patients receiving exposure to systemic therapy after completion of first line treatment.
Improvements in PFS and OS have been demonstrated in both the switch and continuation maintenance settings. As a result, maintenance therapy is often considered a standard of care option in the palliative treatment of advanced NSCLC.
Switch maintenance
With the switch maintenance strategy, trials investigating cytotoxic chemotherapy, targeted agents, and biologic agents have been performed, with some showing improved survival outcomes (Table 1).
![]()
Table 1: Phase III randomized, controlled trials investigating switch maintenance therapy.
View Table 1
Amongst cytotoxic chemotherapy agents, vinorelbine, paclitaxel, pemetrexed, and docetaxel have been investigated in the switch maintenance setting. Neither vinorelbine [10] nor paclitaxel [11] maintenance therapy demonstrated improvements in PFS or OS. In the JMEN trial [12], patients who had not progressed after four cycles of platinum-based chemotherapy were randomly assigned to receive maintenance pemetrexed or placebo, which was continued until disease progression or unacceptable toxicity. Patients who received maintenance pemetrexed demonstrated significantly improved PFS (4.3 vs. 2.6 months, p < 0.0001), and OS (13.4 vs. 10.6 months, p = 0.012). In subgroup analyses, the benefit of pemetrexed was limited to patients with non-squamous histology. Drug-related toxicity was increased in the pemetrexed group, though remained reasonably low, with 16% versus 4% experiencing grade 3 or higher adverse events. However a criticism of this trial was that only 19% of patients in the placebo arm received pemetrexed at any point on progression, while other non-standard therapies were commonly used. Had the randomization been to pemetrexed maintenance versus pemetrexed upon progression, the results may have been clearer to interpret. The docetaxel trial by Fidias et al. [13] randomized patients to receive docetaxel immediately after completion of four cycles of carboplatin-gemcitabine versus delayed administration of docetaxel upon disease progression. A maximum of six cycles of docetaxel were given. PFS was significantly improved amongst the patients who received immediate docetaxel as compared to those who received delayed docetaxel (5.7 vs. 2.7 months, p = 0.0001). Median OS was numerically superior in the immediate docetaxel arm, but not statistically significantly different (12.3 vs. 9.7 months, p = 0.0853). However, when those patients in the delayed docetaxel arm who received docetaxel upon progression were analyzed alone, and thus removing the patients who never received docetaxel, OS was 12.5 months. This was identical to the OS observed in the safety population in the immediate docetaxel arm. In the immediate docetaxel arm, most patients (94.8%) actually received the docetaxel, whereas 37.2% never received it in the delayed arm. Many of the patients in the delayed arm who never received docetaxel were unable to do so as a result of symptomatic progression, despite the rigorous follow-up schedule dictated by the clinical trial. Therefore, the improved OS in the immediate docetaxel arm was largely driven by the higher proportion of patients who actually received docetaxel. Quality of life, toxicity, and response rates were similar between the immediate and delayed groups.
Targeted therapy with erlotinib has been investigated in several trials in the switch maintenance setting. The SATURN trial [14] randomized patients to receive the EGFR-tyrosine kinase inhibitor, erlotinib, versus placebo after completion of four cycles of platinum-based chemotherapy that resulted in at least stable disease. Progression free survival (12.3 vs. 11.1 weeks, p < 0.0001) and overall survival (12.0 vs. 11.0 months, p = 0.0088) were both significantly prolonged with erlotinib maintenance therapy regardless of EGFR status. PFS was significantly improved in both EGFR mutation-positive patients (HR 0.10, p < 0.0001), and EGFR wild-type patients (HR 0.78, p = 0.0185). The ATLAS trial [15] also investigated erlotinib in the switch maintenance setting. Patients received four cycles of chemotherapy plus bevacizumab, and then were randomized to continuing maintenance bevacizumab with or without erlotinib. PFS was improved with the addition of erlotinib to maintenance bevacizumab (4.8 vs. 3.7 months, p < 0.0001), but OS was not (14.4 vs. 13.3 months, p = 0.5341). There were more grade 3/4 adverse events in the group receiving erlotinib. In the French trial by Perol et al., patients who received first line therapy with cisplatin-gemcitabine were then assigned one of three groups: switch maintenance with erlotinib, continuation maintenance with gemcitabine, or observation [16]. In the erlotinib arm, PFS was significantly improved as compared to patients in the observation arm (2.9 vs. 1.9 months, p = 0.003). However, a significant benefit in OS with erlotinib therapy was not observed (11.4 vs. 10.8 months, p = 0.304). However, while switch maintenance therapy with erlotinib is an acceptable option with statistically significant survival benefits, this approach has not been widely adopted. This is related its modest numerical benefits, as well as the fact that most patients are now tested for EGFR mutation prior to first line therapy.
Continuation maintenance
Several phase III trials of the continuation maintenance therapy strategy with non-platinum cytotoxic chemotherapy have been performed. Three studies, PARAMOUNT [17], AVAPERL [18], and Point Break [19], investigated the role of maintenance pemetrexed in non-squamous histology, while gemcitabine as continuation maintenance therapy has also been investigated in three studies. Amongst these, only the continuation of pemetrexed alone in PARAMOUNT has demonstrated improved overall survival.
In the PARAMOUNT [17,20] trial, patients with advanced non-squamous NSCLC received four cycles of cisplatin-pemetrexed induction therapy, followed by randomization to continuation of pemetrexed versus placebo if disease did not progress. With continuation of pemetrexed, median PFS was improved (4.1 vs. 2.8 months, p < 0.0001), and there was a 22% reduction in the risk of death (median 13.9 vs. 11.0 months, p = 0.0195). Between the two groups, there was no difference in quality of life. Grade 3/4 adverse events were more frequent in the group receiving maintenance pemetrexed, with 4% of patients experiencing each of grade 3/4 anemia, neutropenia, and fatigue. In the AVAPERL [18,21] and Point Break [19] trials, bevacizumab was continued beyond induction chemotherapy with or without pemetrexed. While these trials do not give insight as to whether the continuation of bevacizumab as maintenance therapy improves outcomes, they did demonstrate that the addition of pemetrexed to maintenance bevacizumab resulted in improved PFS but not OS.
In the Perol study described above [16], patients who received maintenance gemcitabine had significantly improved PFS (3.8 vs. 1.9 months, p < 0.001), and non-significantly improved OS (13.0 vs. 11.0 months, p = 0.195) as compared to patients in the observation group. In a subgroup analysis, it appeared that maintenance gemcitabine may provide OS benefit to patients in whom induction chemotherapy resulted in objective response (15.2 vs. 10.8 months) (Table 2).
![]()
Table 2: Phase III randomized, controlled trials investigating continuation maintenance therapy.
View Table 2
Thus, there are a variety of options after completion of first line platinum doublet therapy. Many believe that the standard of care would be to offer maintenance therapy, given the observed survival benefits. Significant overall survival benefits have been demonstrated with pemetrexed, docetaxel and erlotinib in the switch maintenance setting and with pemetrexed in the continuation maintenance setting. However, some continue to debate the value of maintenance therapy, arguing that it just represents early second line therapy. But, even when patients are very closely monitored, such as in a clinical trial setting, there is still a large proportion who do not go on to receive second line therapy and thus benefit from maintenance therapy. Some may also worry about prolonged exposure to systemic therapy leading to increased toxicity, though generally a less toxic treatment is used as the maintenance therapy. Indeed the treatment cessation rate due to toxicity is low, and data indicates that quality of life is comparable while on maintenance therapy. In practice, single agent pemetrexed is most frequently used as maintenance therapy in nonsquamous NSCLC, whereas there is less of a consensus for squamous NSCLC. Ultimately, decisions should be made on an individual basis, with consideration of the potential toxicity and impact on quality of life in this generally poor prognosis population.
Second Line Systemic Therapy
Until recently, few clinical trials of systemic therapy demonstrated an improvement in overall survival in the second line setting. However, with the advent of targeted therapy and immunotherapy and their subsequent incorporation into second line therapy, the landscape of treatment options is quickly changing.
Monotherapy
The TAX317 trial [24] was the first phase III trial to demonstrate an improvement in overall survival with the use of second line chemotherapy. In this trial, 204 patients with advanced NSCLC who had previously received platinum-based chemotherapy were randomized to receive docetaxel (at a dose of 75 mg/m2 versus 100 mg/m2) versus best supportive care. OS (7.0 vs. 4.6 months, p = 0.047) and PFS (10.6 vs. 6.7 weeks, p = 0.001) were significantly improved with docetaxel treatment. OS was similar amongst the two patient groups receiving the two doses of docetaxel, though only the 75 mg/m2 dose was significantly better than best supportive care. There were more treatment related deaths and cases of febrile neutropenia in the higher dose group, and thus the 100 mg/m2 dose was discontinued. The response rate was 7.1%, and quality of life was improved in docetaxel-treated patients. Thus, based on the TAX317 trial, docetaxel was moved into standard of care as second line therapy. Pemetrexed can also be used in the second line setting, as it has been shown to have similar clinical efficacy as compared to docetaxel with less febrile neutropenia and a lower rate of hospitalization [25]. However, pemetrexed efficacy appears to be limited to nonsquamous histology. Patients with nonsquamous NSCLC appeared to derive increased benefit from pemetrexed as compared to docetaxel, whereas those with squamous cell carcinomas experienced shorter survival with pemetrexed [26].
Targeted therapy
Targeted therapy also plays a role in the second line treatment of patients with wildtype EGFR, albeit a potentially controversial one. The first trial to demonstrate this role was NCIC CTG BR.21, in which erlotinib was shown to be superior to placebo in the second or third line setting, for patients not considered suitable for further cytotoxic chemotherapy [27]. In BR.21, patients with ECOG performance status 0-3 were randomized to receive erlotinib or placebo after failure of standard chemotherapy. Erlotinib resulted in improved OS (7.0 vs. 4.6 months, p < 0.001) and PFS (10.6 vs. 6.7 weeks, p < 0.001). Quality of life scores for cough, pain, and dyspnea were better in the erlotinib group. At the same time, gefitinib was also tested against placebo in the second- or third-line setting in the ISEL trial [28]. A similar, though non-significant, trend for OS was demonstrated for gefitinib over placebo in this trial.
The INTEREST trial, a phase III non-inferiority trial, compared gefitinib to docetaxel in patients with previously treated advanced NSCLC [29]. Overall survival associated with gefitinib therapy was non-inferior to the OS seen with docetaxel treatment (7.6 vs. 8.0 months, p = 0.50). In a preplanned subgroup analysis, patients receiving second line treatment had similar OS whether treated with gefitinib or docetaxel, whereas in the third line setting, OS was improved with docetaxel as compared to gefinitib. Meanwhile, Erlotinib has also been compared against chemotherapy in three separate phase III trials. In the TAILOR trial, patients treated with docetaxel experienced significantly improved OS and PFS compared to those treated with erlotinib [30]. Conversely, treatment with erlotinib versus chemotherapy resulted in similar efficacy in the TITAN [31] and HORG [32] trials.
More recently, the LUX-Lung 8 trial sought to improve outcomes seen in BR.21 amongst [33] patients with squamous cell carcinoma, a histology with traditionally poor outcomes. Patients with advanced squamous cell carcinoma were randomized to afatinib, an irreversible ErbB family inhibitor, versus erlotinib. Survival outcomes were modestly improved with afatanib, while toxicity was comparable between the two agents.
The use of EGFR TKIs in patients with EGFR wildtype tumours is controversial given the modest and somewhat inconsistent results from randomized trials. For instance, a biomarker analysis in BR.21 demonstrated that there was no OS benefit for erlotinib compared to placebo in patients with EGFR wildtype tumours [27]. For clarification of this topic, two recent meta analyses investigated the efficacy of EGFR TKIs versus chemotherapy in advanced EGFR-wildtype NSCLC [34,35]. These meta analyses came to the same conclusion: in patients with wildtype EGFR, PFS was inferior with the use of EGFR TKIs as compared to chemotherapy. However, OS was similar between the two treatment strategies, and thus the inferior PFS did not translate into inferior OS.
Therefore, despite the low response rates and modest survival benefits seen with EGFR TKIs in patients with wildtype EGFR, they remain a viable treatment option which generally results in disease stabilization for some patients. Unfortunately, there are no biomarkers to direct treatment choice in these unselected patients. Thus, erlotinib (with any NSCLC histology) or afatinib (for squamous cell carcinoma) represent treatment options for those who are not candidates for further cytotoxic chemotherapy.
Combination therapy
Recently, the addition of antiangiogenic agents to second line docetaxel in phase III trials has resulted in modestly improved survival outcomes. Nintedanib, an oral angiokinase inhibitor, was added to docetaxel and compared to docetaxel alone in the second line treatment setting in LUME-Lung 1 [36]. Results from the study revealed that in the entire study population, PFS was improved (3.4 vs. 2.7 months, p = 0.0019) with the addition of nintedanib, though OS was not (10.1 vs. 9.1 months, p = 0.2720). However, upon subgroup analysis, patients treated with nintedanib who had adenocarcinoma histology experienced improved OS (12.6 vs. 10.3 months, p = 0.036), particularly if they had experienced progressive disease within nine months after start of first line chemotherapy. Toxicity was manageable, with increased rates of diarrhea, nausea/vomiting, and liver enzyme elevation. The incidence of antiangiogenic agent class adverse events (such as hypertension, bleeding, perforation, and thromboembolism) was not increased with nintedanib. In the REVEL trial [37], ramucirumab, an anti-VEGFR-2 monoclonal antibody, was investigated in combination with docetaxel in the second line setting. As compared to docetaxel plus placebo, docetaxel with ramucirumab resulted in significantly increased PFS (4.5 vs. 3.0 months, p < 0.0001) and OS (10.5 vs. 9.1 months, p = 0.023). The survival benefit was seen across subgroups and histologies. There was an increased rate of grades 1-2 bleeding, as well as neutropenia and febrile neutropenia in the ramucirumab group. Based on these results, nintedanib has been approved in the EU and ramucirumab has been approved by the FDA in the United States for use in patients with advanced adenocarcinoma of the lung after first line chemotherapy.
Other combination therapies that have been investigated in phase III trials in the second-line setting have not resulted in improved survival outcomes. This includes both combinations of two cytotoxic therapies together, two targeted therapies together, as well as a targeted therapy in addition to chemotherapy. For instance, the addition of gemcitabine [38], capecitabine [39], or vinorelbine [39] to docetaxel did not result in improved overall survival as compared to docetaxel alone. Similarly, the addition of bevacizumab [40] or sunitinib [41] to erlotinib did not prolong overall survival. In the LUME-Lung 2 trial, nintedanib was added to pemetrexed in patients with nonsquamous NSCLC, and compared against single agent pemetrexed. The trial was halted early based on an interim futility analysis, yet the primary endpoint of PFS was actually met. No difference was observed in OS [42].
The combination of pemetrexed and vandetanib was also investigated without observed OS benefit as compared to pemetrexed alone [43]. Cetuximab has been combined with both pemetrexed and docetaxel in an molecularly unselected patient population, and OS was not improved with the combination compared to either single agent chemotherapy [44]. Finally, the docetaxel was combined with aflibercept in the VITAL trial [45] and vandetanib in the ZODIAC trial [46] and compared to docetaxel monotherapy. The combination did not result in prolonged OS in either trial.
Immunotherapy
Immunotherapy is taking off in leaps and bounds in the treatment of multiple different tumour types. Immune checkpoint inhibitors, including PD-1, PDL-1, and CTLA-4 inhibitors, function by disrupting inhibition of the immune response, which leads to T-cell activation, thereby restoring antitumour immunity [47].
Nivolumab, an anti-PD-1 antibody, has recently been shown to have superior efficacy when compared with docetaxel in the second line treatment of advanced NSCLC. The landmark phase III trials, CheckMate 017 [48] and CheckMate 057 [49], randomized patients with squamous and non-squamous NSCLC, respectively, to receive nivolumab or docetaxel after first line platinum chemotherapy. In CheckMate 017, PFS (3.5 vs. 2.8 months, p = 0.008) and OS (9.0 vs. 6.3 months, p < 0.001) were significantly longer in patients treated with nivolumab. In CheckMate 057, median OS was significantly improved with nivolumab (12.2 vs. 9.4 months, p = 0.002), but median PFS was not (2.3 vs. 4.2 months, p = NR). Response rates with nivolumab were 20% and 19% in CheckMate 017 and CheckMate 057, respectively. Remarkably, in those patients who did experience a tumour response to nivolumab, median duration of response was dramatically longer than that seen with docetaxel (median not reached vs. 8.4 months in CheckMate 017, and 17.2 vs. 5.6 months in CheckMate 057). Nivolumab was well tolerated with a favourable safety profile. Serious adverse events were less frequent with nivolumab than with docetaxel (7-10% vs. 54-55%), and serious immune-related adverse events were infrequent. The search for a predictive biomarker turned up inconsistent results between the two trials: in CheckMate 017, PD-L1 expression level did not correlate with outcomes, but in CheckMate 057, PD-L1 expression levels of 1% or greater were associated with even greater efficacy with nivolumab. Therefore, the CheckMate trials have demonstrated that a subset of patients treated with nivolumab experience very favourable outcomes with significantly less toxicity. Currently, use of nivolumab is not restricted to any biomarker subgroup.
Pembrolizumab, another PD-1 inhibitor, was also evaluated against docetaxel in previously treated NSCLC in the KEYNOTE-010 trial [50]. However, unlike in the CheckMate trials, in this study only patients with PD-L1 positive tumours, defined as expression of PD-L1 on at least 1% of tumour cells, were eligible to be enrolled. Two different doses of pembrolizumab, 2 mg/kg or 10 mg/kg every 3 weeks were evaluated against docetaxel. Both doses of pembrolizumab significantly prolonged OS as compared to docetaxel (10.4 vs. 8.5 months in 2 mg/kg, p = 0.0008, and 12.7 vs. 8.5 months in 10 mg/kg, p < 0.0001). OS between the two groups receiving pembrolizumab was similar. In the total population, PFS was similar amongst all three groups (3.9 vs. 4.0 vs. 4.0 months). The magnitude of benefit improved with increasing PD-L1 expression. Subgroup analysis of patients with at least 50% of tumour cells expressing PD-L1 demonstrated more impressive survival benefits. OS was significantly improved with both doses of pembrolizumab as compared to docetaxel (14.9 vs. 17.3 vs. 8.2 months), as was PFS (5.0 vs. 5.2 vs. 4.1 months). Pembrolizumab was well tolerated, with significantly less grade 3-5 adverse events occurring as compared to patients receiving docetaxel.
Currently, nivolumab is approved by the FDA for use pretreated patients with either squamous or nonsquamous NSCLC, while pembrolizumab is approved for use in pretreated patients with PD-L1 positive NSCLC. In addition, there are currently several other immune checkpoint monoclonal antibodies under evaluation for use in advanced NSCLC, including atezolizumab, durvalumab, avelumab, and ipilimumab. Thus, immunotherapy is now a standard second line therapy for patients with advanced squamous and non-squamous NSCLC (Table 3).
![]()
Table 3: Phase III trials with improved overall survival in second-line treatment.
View Table 3
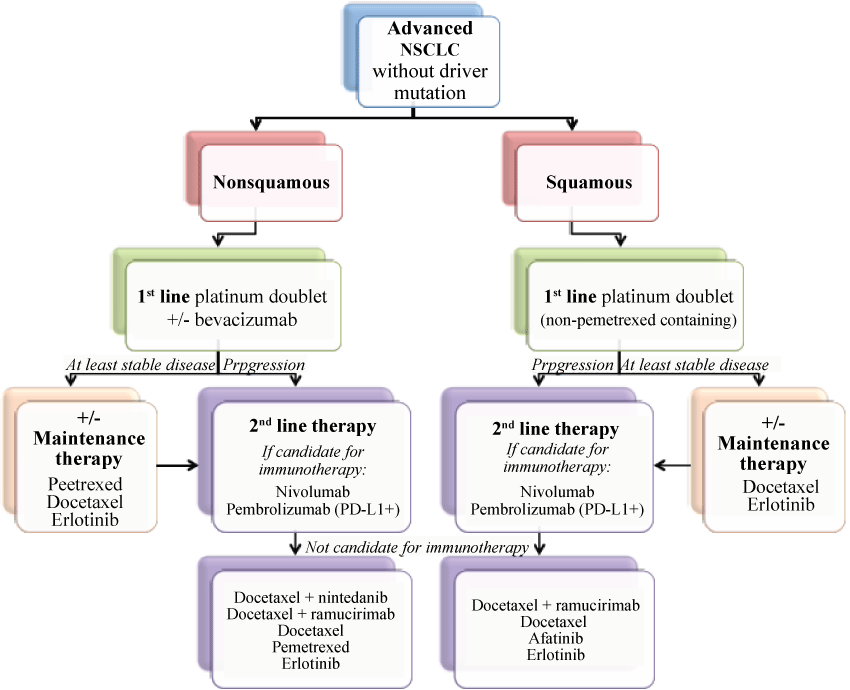
Proposed Treatment Algorithm for Advanced NSCLC without a Driver Mutation
Thus, there are several options during the treatment course for a patient with advanced NSCLC. Currently, standard first line therapy remains chemotherapy with a platinum doublet for four to six cycles. Whether or not the addition of immunotherapy to first line chemotherapy improves clinical outcomes has yet to be seen, but trials are ongoing. If first line doublet therapy results in at least stable disease, maintenance therapy should then be considered in those who are tolerating therapy. Upon progression, treatment with an immune checkpoint inhibitor should be considered the standard approach, as long as the patient is a candidate for immunotherapy. In the third line, docetaxel with a targeted agent (if available) would be the next step. Of course, enrollment in clinical trials throughout the course of treatment is always encouraged (Figure 2).
.
Figure 2: Proposed treatment sequencing options for advanced NSCLC without a driver mutation.
View Figure 2
Future Directions and Challenges
In advanced NSCLC, a disease with a generally poor outlook, recent developments in therapy type, sequence and efficacy provide grounds for optimism. Beyond first line therapy, there now exist many options for maintenance and second line treatment that provide a survival benefit. These options include chemotherapy, targeted therapy, and immunotherapy.
With these new developments come several significant challenges. One important question for patients and clinicians will be how to piece together the evidence. Further, as we strive more to achieve an approach of personalized medicine, there remains a paucity of suitable biomarkers to guide the appropriate treatment choice. Finally, one cannot ignore the significant price tag that accompanies these new agents. These challenges must be addressed without delay, with research seeking to provide answers and guidance.
The optimal treatment sequence, timing, and combinations have yet to be determined. Immunotherapy will now be commonly used and considered the standard of care in the second line. But, can those studies which used docetaxel as the control arm still be considered valid in the third line given the paucity of evidence for their use in this setting? Should all previous lines of systemic therapy be pushed back to accommodate immunotherapy? There is not a clear evidence-based answer, and yet, it would be an inefficient use of time and resources to repeat these studies in the immunotherapy era. In the CheckMate trials, 29% of patients in the Nivolumab arms went on to receive a taxane as their next line of therapy, which represented the most common choice. With the available data, we believe that pushing back previous standard lines of therapy by one line to accommodate immunotherapy is a reasonable approach, with the understanding that the evidence is lacking and thus uncertainty exists. As we move towards the future and newer trials will be designed to take standard immunotherapy into account, more solid evidence should ensue. We also await results of ongoing clinical trials, which combine chemotherapy and immunotherapy in the first line setting for advanced NSCLC. If these trials demonstrate positive results, then the ongoing challenge of optimal sequencing and combinations will yet again be of prime importance. In this regard, it is also appealing to believe that for selected patients, first line immunotherapy may provide durable disease control without the need for cytotoxic chemotherapy.
As the demonstrated response rates and survival curves for the checkpoint inhibitors suggest, the majority of patients still do not obtain benefit from immunotherapy in the second line setting. Furthermore, in CheckMate 057, the survival curves initially favour docetaxel until a crossing over of the curves occurs around the six month point, perhaps indicating not only a delayed time to observation of benefit from immunotherapy, but also an early complete lack of benefit for some patients. Thus, determining the optimal treatment choice on an individual patient basis would be ideal. For this to occur, an effective biomarker with a suitable diagnostic companion must be found. The data on the predictive value of PD-L1 expression level is inconsistent, as outlined above. In CheckMate 017, the expression of PD-L1 was neither prognostic nor predictive of benefit from Nivolumab, whereas in CheckMate 057, increasing PD-L1 expression was associated with improved efficacy, though efficacy was demonstrated at all levels of expression. Keynote 010 only enrolled patients with positive PD-L1 expression of at least 1% on tumour biopsy, and efficacy was greater in those with 50% or more PD-L1 expression. To augment the inconsistencies, the cutoffs defining PD-L1 positivity studied and the diagnostic tests used differ between trials. Optimal cutoffs and tests have not yet been defined. Furthermore, questions remain regarding the use of fresh or archival tissue and primary or metastatic site for determination of PD-L1 status, as well as the fluctuating nature of PD-L1 expression, which can change over time and with treatment [51]. Thus, at this point, PD-L1 expression is not an adequate biomarker for patient selection for immune checkpoint inhibitors, in our opinion. It will be of utmost important moving forward for the scientific community to take steps to identify an appropriate biomarker for immunotherapy, and also for the other targeted and cytotoxic therapies that are in the treatment algorithm.
As is becoming increasingly apparent, new developments in systemic therapy have come not only with improved clinical outcomes, but also with a significant price tag that is challenging the sustainability of the health care system. As a cancer community, we strive to make cancer care attainable for all, yet this will not be feasible in the current climate. As Dr. Saltzso eloquently discussed in his ASCO plenary session [52], the true value of therapies must be determined, taking into account not only efficacy, but also toxicity and price, and difficult discussions must take place as to what society can bear. Once again, determining appropriate biomarkers for appropriate patient selection leading to less ineffective use of therapies will also be helpful.
Ultimately, the objective of these therapeutic options will be to provide patients with an individualized treatment approach ensuring that quality of life and quality of care needs are met. The horizon of treatment options is looking brighter than ever, particularly in the second line setting of palliative systemic therapy.
References
-
Ferlay J, Soerjomataram I, Ervik M, Dikshit R, Eser S, et al. (2013) GLOBOCAN 2012 v1.0, cancer incidence and mortality worldwide: IARC Cancer Base No. 11. International Agency for Research on Cancer, Lyon, France.
-
(2015) Surveillance, Epidemiology, and End Results Program. SEER stat fact sheets: Lung and bronchus cancer.
-
Brule SY, Al-Baimani K, Jonker H, Zhang T, Wheatley-Price P, et al. (2015) Palliative chemotherapy (CT) for advanced non-small cell lung cancer (NSCLC): Investigating disparities between patients who are treated versus those who are not. J Clin Oncol 33.
-
Lang K, Marciniak , Faries D, Stokes M, Buesching D, et al. (2009) Trends and predictors of first-line chemotherapy use among elderly patients with advanced non-small cell lung cancer in the united states. Lung Cancer 63: 264-270.
-
Sacher AG, Le LW, Lau A, Earle CC, Leighl NB (2015) Real-world chemotherapy treatment patterns in metastatic non-small cell lung cancer: Are patients undertreated? Cancer 121: 2562-2569.
-
Krista Noonan, King Mong Tong, Sophie Sun, Barbara L. Melosky, Nevin Murray, et al. (2014) Population-based analysis of time to palliative chemotherapy (TPC) and survival in advanced non small cell lung cancer (NSCLC). J Clin Oncol 32.
-
Brule SY, Wheatley-Price P (2015) Non-small cell lung cancer: Two decades of progress. Oncology Exchange 14: 12-20.
-
Masters GA, Temin S, Azzoli CG, Giaccone G, Baker S, et al. (2015) Systemic therapy for stage IV non-small-cell lung cancer: American society of clinical oncology clinical practice guideline update. J Clin Oncol 33: 3488-3515.
-
Gerber DE, Schiller JH (2013) Maintenance chemotherapy for advanced non-small-cell lung cancer: New life for an old idea. J Clin Oncol 31: 1009-1020.
-
Westeel V, Quoix E, Moro-Sibilot D, Mercier M, Breton JL, et al. (2005) Randomized study of maintenance vinorelbine in responders with advanced non-small-cell lung cancer. J Natl Cancer Inst. 97: 499-506.
-
Sculier JP, Lafitte JJ, Lecomte J, Alexopoulos CG, Van Cutsem O, et al. (2007) A phase III randomised trial comparing sequential chemotherapy using cisplatin-based regimen and paclitaxel to cisplatin-based chemotherapy alone in advanced non-small-cell lung cancer. Ann Oncol 18: 1037-1042.
-
Ciuleanu T, Brodowicz T, Zielinski C, Kim JH, Krzakowski M, et al. (2009) Maintenance pemetrexed plus best supportive care versus placebo plus best supportive care for non-small-cell lung cancer: A randomised, double-blind, phase 3 study. Lancet 374: 1432-1440.
-
Fidias PM, Dakhil SR, Lyss AP, Loesch DM, Waterhouse DM, et al. (2009) Phase III study of immediate compared with delayed docetaxel after front-line therapy with gemcitabine plus carboplatin in advanced non-small-cell lung cancer. J Clin Oncol 27: 591-598.
-
Cappuzzo F, Ciuleanu T, Stelmakh L, Cicenas S, Szczésna A, et al. (2010) Erlotinib as maintenance treatment in advanced non-small-cell lung cancer: A multicentre, randomised, placebo-controlled phase 3 study. Lancet Oncol 1: 521-529.
-
Johnson BE, Kabbinavar F, Fehrenbacher L, Hainsworth J, Kasubhai S et al. (2013) ATLAS: Randomized, double-blind, placebo-controlled, phase IIIB trial comparing bevacizumab therapy with or without erlotinib, after completion of chemotherapy, with bevacizumab for first-line treatment of advanced non-small-cell lung cancer. J Clin Oncol 31: 3926-3934.
-
Perol M, Chouaid C, Perol D, Barlesi F, Gervais R, et al. (2012) Randomized, phase III study of gemcitabine or erlotinib maintenance therapy versus observation, with predefined second-line treatment, after cisplatin-gemcitabine induction chemotherapy in advanced non-small-cell lung cancer. J Clin Oncol 30: 3516-3524.
-
Paz-Ares L, de Marinis F, Dediu M, Thomas M, Pujol JL, et al. (2012) Maintenance therapy with pemetrexed plus best supportive care versus placebo plus best supportive care after induction therapy with pemetrexed plus cisplatin for advanced non-squamous non-small-cell lung cancer (PARAMOUNT): A double-blind, phase 3, randomised controlled trial. Lancet Oncol 13: 247-255.
-
Barlesi F, Scherpereel A, Rittmeyer A, Antonio Pazzola, Neus Ferrer Tur, et al. (2013) Randomized phase III trial of maintenance bevacizumab with or without pemetrexed after first-line induction with bevacizumab, cisplatin, and pemetrexed in advanced nonsquamous non-small-cell lung cancer: AVAPERL (MO22089). J Clin Oncol 31: 3004-3011.
-
Patel JD, Socinski MA, Garon EB, Reynolds CH, Spigel DR, et al. (2013)PointBreak: A randomized phase III study of pemetrexed plus carboplatin and bevacizumab followed by maintenance pemetrexed and bevacizumab versus paclitaxel plus carboplatin and bevacizumab followed by maintenance bevacizumab in patients with stage IIIB or IV nonsquamous non-small-cell lung cancer. J Clin Oncol 31: 4349-4357.
-
Paz-Ares LG, de Marinis F, Dediu M, Thomas M, Pujol JL, et al. (2013) PARAMOUNT: Final overall survival results of the phase III study of maintenance pemetrexed versus placebo immediately after induction treatment with pemetrexed plus cisplatin for advanced nonsquamous non-small-cell lung cancer. J Clin Oncol 31: 2895-2902.
-
Barlesi F, Scherpereel A,Gorbunova V, Gervais R, Vikstrom A, et al. (2014) Maintenance bevacizumab-pemetrexed after first-line cisplatin-pemetrexed-bevacizumab for advanced nonsquamousnonsmall-cell lung cancer: Updated survival analysis of the AVAPERL (MO22089) randomized phase III trial. Ann Oncol 25: 1044-1052.
-
Brodowicz T, Krzakowski M, Zwitter M, Tzekova V, Ramlau R, et al. (2006) Cisplatin and gemcitabine first-line chemotherapy followed by maintenance gemcitabine or best supportive care in advanced non-small cell lung cancer: A phase III trial. Lung Cancer 52: 155-163.
-
CP Belani, DM Waterhouse, H Ghazal, SS Ramalingam, R Bordoni, et al. (2010) Phase III study of maintenance gemcitabine (G) and best supportive care (BSC) versus BSC, following standard combination therapy with gemcitabine-carboplatin (G-cb) for patients with advanced non-small cell lung cancer (NSCLC). J Clin Oncol 28.
-
Shepherd FA, Dancey J, Ramlau R, Mattson K, Gralla R, et al. (2000) Prospective randomized trial of docetaxel versus best supportive care in patients with non-small-cell lung cancer previously treated with platinum-based chemotherapy. J Clin Oncol 18: 2095-2103.
-
Hanna N, Shepherd FA, Fossella FV, Pereira JR, De Marinis F, et al. (2004) Randomized phase III trial of pemetrexed versus docetaxel in patients with non-small-cell lung cancer previously treated with chemotherapy. J Clin Oncol 22: 1589-1597.
-
Scagliotti G, Hanna N, Fossella F, Sugarman K, Blatter J, et al. (2009) The differential efficacy of pemetrexed according to NSCLC histology: A review of two phase III studies. Oncologist 14: 253-263.
-
Shepherd FA, Rodrigues Pereira J, Ciuleanu T, Tan EH, Hirsh V, et al. (2005) Erlotinib in previously treated non-small-cell lung cancer. N Engl J Med 353: 123-132.
-
Thatcher N, Chang A, Parikh P, Rodrigues Pereira J, Ciuleanu T, et al. (2005) Gefitinib plus best supportive care in previously treated patients with refractory advanced non-small-cell lung cancer: Results from a randomised, placebo-controlled, multicentre study (Iressa survival evaluation in lung cancer). Lancet 366: 1527-1537.
-
Kim ES, Hirsh V, Mok T, Socinski MA, Gervais R, et al. (2008) Gefitinib versus docetaxel in previously treated non-small-cell lung cancer (INTEREST): A randomised phase III trial. Lancet 372: 1809-1818.
-
Garassino MC, Martelli O, Broggini M, Farina G, Veronese S, et al. (2013) Erlotinib versus docetaxel as second-line treatment of patients with advanced non-small-cell lung cancer and wild-type EGFR tumours (TAILOR): A randomised controlled trial. Lancet Oncol 14: 981-988.
-
Ciuleanu T, Stelmakh L, Cicenas S, Miliauskas S, Grigorescu AC, et al. (2012) Efficacy and safety of erlotinib versus chemotherapy in second-line treatment of patients with advanced, non-small-cell lung cancer with poor prognosis (TITAN): A randomised multicentre, open-label, phase 3 study. Lancet Oncol 13: 300-308.
-
Karampeazis A,Voutsina A, Souglakos J, Kentepozidis N, Giassas S, et al. (2013) Pemetrexed versus erlotinib in pretreated patients with advanced non-small cell lung cancer: A hellenic oncology research group (HORG) randomized phase 3 study. Cancer 119: 2754-2764.
-
Soria JC, Felip E, Cobo M, Lu S, Syrigos K, et al. (2015) Afatinib versus erlotinib as second-line treatment of patients with advanced squamous cell carcinoma of the lung (LUX-lung 8): An open-label randomised controlled phase 3 trial. Lancet Oncol 16: 897-907.
-
Lee JK, Hahn S, Kim DW, Suh KJ, Keam B, et al. (2014) Epidermal growth factor receptor tyrosine kinase inhibitors vs conventional chemotherapy in non-small cell lung cancer harboring wild-type epidermal growth factor receptor: A meta-analysis. JAMA 311: 1430-1437.
-
Zhao N, Zhang XC, Yan HH, Yang JJ, Wu YL (2014) Efficacy of epidermal growth factor receptor inhibitors versus chemotherapy as second-line treatment in advanced non-small-cell lung cancer with wild-type EGFR: A meta-analysis of randomized controlled clinical trials. Lung Cancer 85: 66-73.
-
Reck M, Kaiser R, Mellemgaard A, Douillard JY, Orlov S, et al. (2014) Docetaxel plus nintedanib versus docetaxel plus placebo in patients with previously treated non-small-cell lung cancer (LUME-lung 1): A phase 3, double-blind, randomised controlled trial. Lancet Oncol 15: 143-155.
-
Garon EB, Ciuleanu TE, Arrieta O, Prabhash K, Syrigos KN, et al. (2014) Ramucirumab plus docetaxel versus placebo plus docetaxel for second-line treatment of stage IV non-small-cell lung cancer after disease progression on platinum-based therapy (REVEL): A multicentre, double-blind, randomised phase 3 trial. Lancet 384: 665-673.
-
Takeda K, Negoro S, Tamura T, Nishiwaki Y, Kudoh S, et al. (2009) Phase III trial of docetaxel plus gemcitabine versus docetaxel in second-line treatment for non-small-cell lung cancer: Results of a japan clinical oncology group trial (JCOG0104). Ann Oncol 20: 835-841.
-
Gebbia V, Gridelli C, Verusio C, Frontini L, Aitini E, et al. (2009) Weekly docetaxel vs. docetaxel-based combination chemotherapy as second-line treatment of advanced non-small-cell lung cancer patients. the DISTAL-2 randomized trial. Lung Cancer 63: 251-258.
-
Herbst RS, Ansari R, Bustin F, Flynn P, Hart L, et al. (2011) Efficacy of bevacizumab plus erlotinib versus erlotinib alone in advanced non-small-cell lung cancer after failure of standard first-line chemotherapy (BeTa): A double-blind, placebo-controlled, phase 3 trial. Lancet 377: 1846-1854.
-
Scagliotti GV, Krzakowski M, Szczesna A, Strausz J, Makhson A, et al. (2012) Sunitinib plus erlotinib versus placebo plus erlotinib in patients with previously treated advanced non-small-cell lung cancer: A phase III trial. J Clin Oncol 30: 2070-2078.
-
Nasser H Hanna, Rolf Kaiser, Richard N Sullivan, Osvaldo Rudy Aren, Myung-JuAhn, et al. (2013) Lume-lung 2: A multicenter, randomized, double-blind, phase III study of nintedanib plus pemetrexed versus placebo plus pemetrexed in patients with advanced nonsquamous non-small cell lung cancer (NSCLC) after failure of first-line chemotherapy. J Clin Oncol 31.
-
de Boer RH, Arrieta Ó, Yang CH, Gottfried M, Chan V, et al. (2011)Vandetanib plus pemetrexed for the second-line treatment of advanced non-small-cell lung cancer: A randomized, double-blind phase III trial. J Clin Oncol 29: 1067-1074.
-
Kim ES, Neubauer M, Cohn A, Schwartzberg L, Garbo L, et al. (2013) Docetaxel or pemetrexed with or without cetuximab in recurrent or progressive non-small-cell lung cancer after platinum-based therapy: A phase 3, open-label, randomised trial. Lancet Oncol 14: 1326-1336.
-
Ramlau R, Gorbunova V, Ciuleanu TE, Novello S, Ozguroglu M, et al. (2012) Aflibercept and docetaxel versus docetaxel alone after platinum failure in patients with advanced or metastatic non-small-cell lung cancer: A randomized, controlled phase III trial. J Clin Oncol 30: 3640-3647.
-
Herbst RS, Sun Y, Eberhardt WE, Germonpré P, Saijo N, et al. (2010)Vandetanib plus docetaxel versus docetaxel as second-line treatment for patients with advanced non-small-cell lung cancer (ZODIAC): A double-blind, randomised, phase 3 trial. Lancet Oncol 11: 619-626.
-
La-Beck NM, Jean GW, Huynh C, Alzghari SK, Lowe DB (2015) Immune checkpoint inhibitors: New insights and current place in cancer therapy. Pharmacotherapy 35: 963-976.
-
Brahmer J, Reckamp KL, Baas P,LucioCrinò, Wilfried, EE, et al. (2015) Nivolumab versus docetaxel in advanced squamous-cell non-small-cell lung cancer. N Engl J Med 373: 123-135.
-
Borghaei H, Paz-Ares L, Horn L, Spigel DR, Steins M, et al. (2015) Nivolumab versus docetaxel in advanced nonsquamous non-small-cell lung cancer. N Engl J Med 373: 1627-1639.
-
Herbst RS, Baas P, Kim DW, EnriquetaFelip, José L Pérez-Gracia, et al. (2015)Pembrolizumab versus docetaxel for previously treated, PD-L1-positive, advanced non-small-cell lung cancer (KEYNOTE-010): A randomised controlled trial. Lancet 387: 1540–1550.
-
Reck M, Paz-Ares L (2015) Immunologic checkpoint blockade in lung cancer. Semin Oncol 42: 402-417.
-
Saltz L (2015) ASCO plenary session 2015, Perspectives on value, Chicago, Updated ASCO Annual Meeting.