International Journal of Cancer and Clinical Research
Metastatic Squamous Cell Carcinoma of the Neck from Unknown Primary Sites: the Ottawa Regional Cancer Centre (ORCC) Experience
Osama Marglani1*, Ameen Zaid Alherabi1, Ali Safar2, Suliman Alghamdi3, Libni Eapen4, Helen Caetano5 and Martin Corsten5
1Department of otolaryngology, Head and Neck Surgery, Faculty of Medicine, Umm Al-Qura University, Makkah, Saudi Arabia
2Department of Otolaryngology, Head and Neck Surgery, Al-Sabah and Zain Hospital, Kuwait city, Kuwait
3Department Oncology, King Abdulaziz Medical City, Jeddah, Saudi Arabia
4Radiation Oncologist, Ottawa Regional Cancer Center, Ontario, Canada
5Department of Otolaryngology, Head and Neck Surgery, The Ottawa Hospital, Ontario, Canada
*Corresponding author:
Osama Marglani, Associate Professor, Department of otolaryngology, Head and Neck Surgery, Faculty of Medicine, Umm Al-Qura University, Makkah, Saudi Arabia, Fax: 00966-2-5270000, Ext-4110, E-mail: osamamergalani@yahoo.com, marglani1@yahoo.com
Int J Cancer Clin Res, IJCCR-2-027, (Volume 2, Issue 4), Original Research; ISSN: 2378-3419
Received: September 04, 2015 | Accepted: September 23, 2015 | Published: September 25, 2015
Citation: Marglani O, Alherabi AZ, Safar A, Alghamdi S, Eapen L, et al. (2015) Metastatic Squamous Cell Carcinoma of the Neck from Unknown Primary Sites: the Ottawa Regional Cancer Centre (ORCC) Experience. Int J Cancer Clin Res 2:027. 10.23937/2378-3419/2/4/1027
Copyright: © 2015 Marglani O, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Background: Metastatic squamous cell carcinoma (SCC) to the neck from an unknown primary site presents a challenging diagnostic and therapeutic disease entity, despite a thorough diagnostic workup. The optimal management of patients who have this syndrome is still unclear and controversial. We present our experience in management of patients with unknown primary.
Objectives: We conducted this study to evaluate our experience in management and survival of patient with unknown primary. We also compared the difference in survival rate in the patients receiving combined treatment as compare to the patients receiving single modality treatment.
Materials and Methods: We reviewed retrospectively patients with cervical nodal metastases from unknown primary sites, who presented to the Ottawa Regional Cancer Center (ORCC). The subjects were treated either radiation therapy or combined therapy. Overall survival percentage was calculated by performing Kaplan Meier survival analysis.
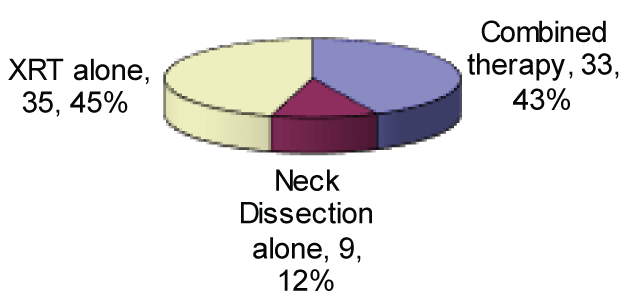
Results: Unknown primary disease was diagnosed in 78 patients. The commonest positive lymph nodes were present at level II (33.33%), commonest stage of the nodal disease in our study was at N2. Comparing the group receiving the radiotherapy alone (44.9%) and the group receiving the combined modality treatment (43.6%) in terms of survival, the survival rates were significantly higher in combined modality treatment group.
Conclusion: In patients who are diagnosed with unknown primary, the combined modality of the treatment that is surgery followed by radiotherapy forms the best mode of management with significantly better survival rates at the end of five years as compare to single modality of treatment.
Keywords
Unknown primary, Squamous cell carcinoma, Head and neck cancer
Introduction
The carcinoma of unknown primary is defined as histological diagnosis of nodal metastasis without detection of primary tumor. In 1882 the Volkmann was the first author to describe three cases of lateral cervical lymph node metastasis in the region of level II while primary malignant tumor could not be diagnosed [1]. In 1957 Commes et al. [2] described a cervical lymph node metastasis without primary tumor and for the first time defined the disease as carcinoma of unknown primary (CUP) [2].
The primary site for metastatic cervical adenopathy remains unidentified in 3-9% of patients [3]. Despite many investigations; the primary site is never located in the majority of patients. The overall five year survival rate is approximately 50% [3,4]. The management of patient with cervical CUP represents both a diagnostic and therapeutic challenge. Optimal treatment is controversial in these patients. The lack of randomized controlled trials and decreasing incidence of CUP make it difficult to draw conclusion regarding an optimal treatment plan. The aim of our study was to evaluate different treatment modalities and to find if there is a significant difference in survival between these patients. A retrospective study of squamous cell carcinoma (SCC) lymph node metastases with unknown primary site was undertaken to evaluate our experience in management and survival.
Materials and Methods
Charts from the Ottawa Hospital Cancer Center registered as SCC Mets to Neck (Primary Unknown) were reviewed. The exclusion criteria were pathology other than SCC. We included the patients with squamous cell carcinoma as it is the commonest histopathological diagnosis in patients presenting with carcinoma of unknown primary in our institute. A total of 154 charts were retrieved and 78 charts met the study criteria (n = 78). A detailed review of demographics, investigations, staging (TNM 2002), treatment and survival was performed on these charts. The demographics studied were gender, age, histology and nodal levels. Investigations recorded were chest x-ray, panendoscopy, and CT scan of neck. The treatment groups were radiation therapy, combined therapy (radiation and surgery). The external beam radiotherapy (XBRT) total dose was recorded in Gray. Radiotherapy treatment either preoperative and/or postoperative was noted. Types of surgical neck dissections performed were modified radical neck dissection, radical neck dissection or selective neck dissection. Combined treatment was defined as surgery and radiation treatment within 3 months of each other.
Data were analyzed using SPSS version 16 (SPSS Inc., Chicago, IL, USA). Overall survival percentage was calculated by performing Kaplan Meier survival analysis.
Results
Seventy-eight patients had histology of SCC with 63 (77.7%) of them were males. The average age of the patient was 66.5 years. Investigations included chest x-rays, panendoscopy and CT scan of the neck, none of our patients had a PET scan. The N2 was the commonest stage of the disease at presentation (46%). 35 (44.9%) patients received only radiotherapy treatment. (Table 1). XBRT was given postoperatively in 20 and preoperatively in 14 patients. XBRT was delivered to a volume encompassing the naso-oro-hypopharynx, larynx, and bilateral neck (24-60 Grey with mean 48.1 Grey); and a small field boost of (4-28 Grey with mean 17.4 Grey). Neck dissections were performed in a total of 43 patients, 15 (34.9%) had radical neck dissection, 20 (46.5%) modified radical neck dissection, 3 (7%) functional neck dissection and 5 (11.6%) patients limited neck dissection.
![]()
Table 1: Disease extent with treatment modalities (N = 78).
View Table 1
Primary site was discovered following treatment in 7 (8.9%) of patients. Two nasopharyngeal tumors were discovered at four and five months respectively, and two hypopharyngeal tumors were discovered at seven months and four years respectively (Table 2). There was high mortality rate in the patients where primary tumors progresses even after the treatment.
![]()
Table 2: Discovery of primary site and survival.
View Table 2
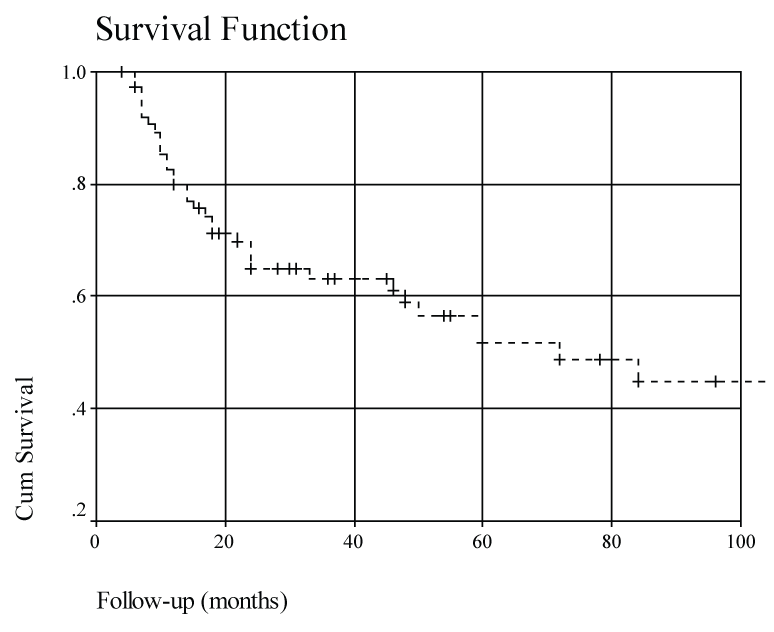
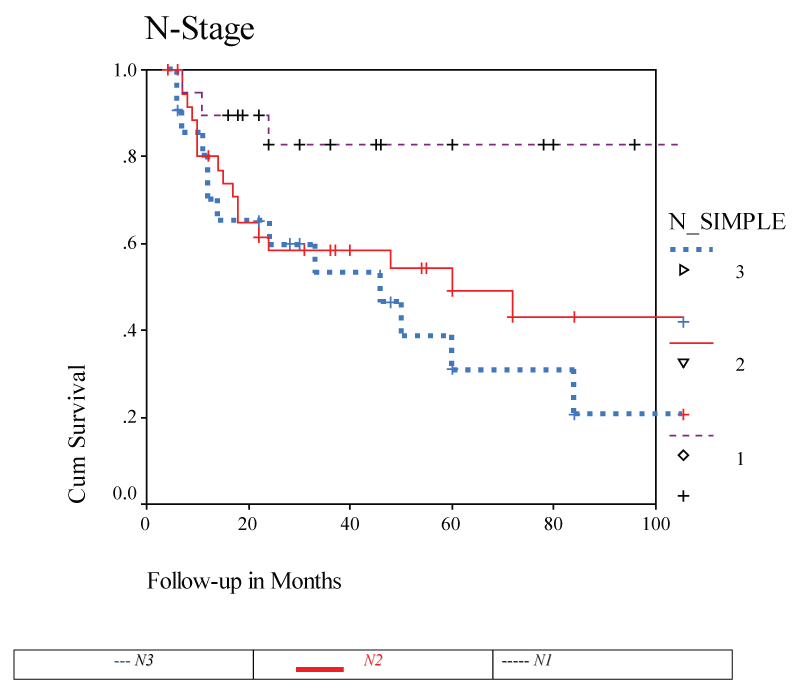
With a minimum follow up of five years overall patient survival was 50% (Kaplan Meier). Eleven (14.1%) of our patients had distant metastasis: lung, bone, brain, and liver. Overall survival by stage showed a survival rate of 80% in N-1, 45% in stage N-2 and 25% in stage N-3.
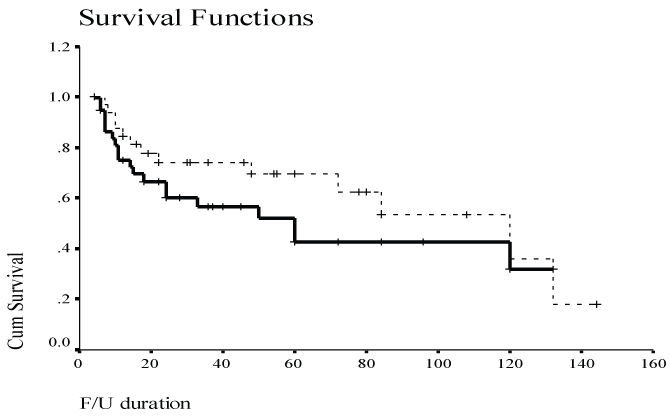
Survival curves comparing treatment groups XRT alone and combined treatment demonstrated a trend to improvement with combined treatment. (Figure 1, Figure 2, Figure 3 and Figure 4). The overall survival was 75.71% in combined group with standard deviation of 13.97%.
The survival in single modality group was 58.57% with standard deviation of 21.93. Mean period of follow up for both the groups was 5 years. Comparing the survival percentage between two groups using the log-rank test, the results were statistically significant (P < 0.05). Indicating the survival is better in the patients who received combined treatment.
Discussion
The management of patients who presents with CUP remains a clinical challenge. The aim of our study was to compare the different treatment modality for this disease.
In 1998, Medine et al. [5], Sjtrojan et al. [6], published independent studies on management of CUP [5,6], the overall survival rate in these studies varies from 54% to 64%. Even the diagnostic workup in the management of CUP remains controversial. Currently the treatment of unknown primary is based on non-randomized data and institutional policy. We present our experience in managing the cases with unknown primary. All the cases were approached with diagnostic CT scan of the Neck and Head neck region, followed by panendoscopy and multiple biopsies from all the suspicious areas. In our study the commonest presentation was positive lymphnodes at level III, and commonest stage of the disease was N2. The commonest histopathological type in all the studies reported to be squamous cell carcinoma, with reported incidences of 70-90%. We included only the cases of squamous cell carcinoma.
Discovery of the primary site after treatment occurred in 7 (8.9%) patients in this review. Previously reported primary discovery rates ranged from 3-40% [4,7,8]. Discovery of the primary site has been thought to be detrimental to survival [1].
Overall survival percentage at five years was 50% in this study. This was comparable to previous reports [3,4]. Also, it has previously been shown that survival correlated with nodal stage [3]. It has been demonstrated in our patient population as well. Due to the size of the study groups we were unable to draw conclusions with regards to treatment modalities and their effect on survival, but data suggested that the combined treatment group has improved survival compared to radiotherapy or surgical treatment alone [9,10]. Majority of disease progression occurred at the initial neck site. Radiotherapy was the primary method of treatment. The overall survival was 75.71% in combined group with standard deviation of 13.97%. The survival in single modality group was 58.57% with standard deviation of 21.93. Mean period of follow up for both the groups was 5 years. Comparing the survival percentage between two groups using the log-rank test the results were statistically significant (P < 0.05). Indicating the survival is better in the patients who received combined treatment. The objectives for the treatment of unknown primary SCC are control of regional lymph node metastases and control of possible primary sites. Because the majority of patients had disease progression at the initial neck site, Although SCC of the neck with an unknown primary continued to be a diagnostic and management challenge, all large studies were retrospective including ours' spanning many years, and difficult to compare given disparities in diagnostic workup and treatment subgroups. Therefore interpretation of the information is limited, and a multi-center study on a prospective basis given the problem of infrequent cases may help us determine the optimal strategy in terms of loco-regional control, survival and morbidity.
Conclusion
Overall five years survival was 50% with 80% in subjects with N-1 stage. Combined treatment demonstrated a trend towards improvement. SCC of the neck with an unknown primary continued to be a diagnostic and management challenge.
Ethical Statement
The Institutional Review Board of the institution granted us permission to conduct this study. We declare that we have no financial or personal relationship(s) which may have inappropriately influenced us in writing this paper.
References
-
Volkman R (1882) Das tiefe branchiogene Halcarcinom. Zbl Chirurgie 9: 49-51.
-
Comess MS, Beahrs OH, Dockerty MB (1957) Cervical metastasis from occult carcinoma. Surg Gynecol Obstet 104: 607-617.
-
Wang RC, Goepfert H, Barber AE, Wolf P (1990) Unknown primary squamous cell carcinoma metastatic to the neck. Arch Otolaryngol Head Neck Surg 116: 1388-1393.
-
Talmi YP, Wolf GT, Hazuka M, Krause CJ (1996) Unknown primary of the head and neck. J Laryngol Otol 110: 353-356.
-
Medini E, Medini AM, Lee CK, Gapany M, Levitt SH (1998) The management of metastatic squamous cell carcinoma in cervical lymph nodes from an unknown primary. Am J Clin Oncol 21: 121-125.
-
Strojan P, Anicin A (1998) Combined surgery and postoperative radiotherapy for cervical lymph node metastases from an unknown primary tumour. Radiother Oncol 49: 33-40.
-
Reddy SP, Marks JE (1997) Metastatic carcinoma in the cervical lymph nodes from an unknown primary site: results of bilateral neck plus mucosal irradiation vs. ipsilateral neck irradiation. Int J Radiat Oncol Biol Phys 37: 797-802.
-
Maulard C, Housset M, Brunel P, Huart J, Ucla L, et al. (1992) Postoperative radiation therapy for cervical lymph node metastases from an occult squamous cell carcinoma. Laryngoscope 102: 884-890.
-
Bataini JP, Rodriguez J, Jaulerry C, Brugere J, Ghossein NA (1987) Treatment of metastatic neck nodes secondary to an occult epidermoid carcinoma of the head and neck. Laryngoscope 97: 1080-1084.
-
Davidson BJ, Spiro RH, Patel S, Patel K, Shah JP (1994) Cervical metastases of occult origin: the impact of combined modality therapy. Am J Surg 168: 395-399.