International Journal of Cancer and Clinical Research
Laparoscopic-Assisted Surgery for Malignant Fibrous Histiocytoma Originating from the Descending Colon: A Case Report and Literature Review
Keiji Matsuda1*, Takahiro Yagi2, Mitsuo Tsukamoto2, Yoshihisa Fukushima2, Takuya Akahane2, Ryu Shimada2, Atsushi Horiuchi2, Keisuke Nakamura2, Takeshi Tsuchiya2, Tamuro Hayama2, Junko Tamura2, Hisae Iinuma2, Shoichi Fujii2, Keijiro Nozawa2, Takuo Tokairin2, Yuko Sasajima1, Fukuo Kondo1 and Yojiro Hashiguchi1
1The Department of Surgery, Teikyo University School of Medicine, Tokyo, Japan
2The Department of Pathology, Teikyo University School of Medicine, Tokyo, Japan
*Corresponding author:
Keiji Matsuda, The Department of Surgery, Teikyo University School of Medicine, 2-11-1 Kaga, Itabashi-ku, Tokyo, Japan, Tel: 81339641231, E-mail: keiji@med.teikyo-u.ac.jp
Int J Cancer Clin Res, IJCCR-2-022, (Volume 2, Issue 3), Case Report; ISSN: 2378-3419
Received: August 14, 2015 | Accepted: August 28, 2015 | Published: August 30, 2015
Citation: Matsuda K, Yagi T, Tsukamoto M, Fukushima Y, Akahane T, et al. (2015) Laparoscopic-Assisted Surgery for Malignant Fibrous Histiocytoma Originating from the Descending Colon: A Case Report and Literature Review. Int J Cancer Clin Res 2:022. 10.23937/2378-3419/2/3/1022
Copyright: © 2015 Matsuda K, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Primary malignant fibrous histiocytoma (MFH) localized in the large intestine is extremely rare. We report a first case of MFH in colon that underwent a laparoscopy-assisted colectomy. A 74-year-old female had a 3-month history of blood stool. Barium enema study showed protrusion in the descending colon. Colonoscopy revealed submucosal tumor-like lesion with central ulceration. A biopsy was obtained and the pathological report was that MFH was suspected. Laparoscopic-assisted left hemicolectomy with en-block regional lymph node dissection was performed. The tumor was a blackish brown and firm nodule projecting into the lumen, measuring 3.5 x 3 cm in diameter. The histopathological report revealed a highly pleomorphic neoplasm composed of large, pleomorphic giant cells as well as spindle cells, arranged in fascicular and storiform pattern mixed with severe inflammatory cells infiltration, including lymphocytes, plasma cells, polymorphonuclear leukocytes and multinucleated giant cells. Immunohistochemical staining showed positive reaction of tumor cells with CD68, α1-antitrypsin and α1-antichymotrypsin. Cytokeratin, c-kit and desmin were negative. These findings were consistent with MFH of the descending colon. Six months later no recurrence was detected. Intensive and careful follow-up should be performed because recurrence commonly occurs in peritoneum/retroperitoneum.
Keywords
Malignant fibrous, Histiocytoma, Laparoscopic surgery, Colectomy, Colon tumor, Immunohistochemical staining
Background
Malignant fibrous histiocytoma (MFH) is a malignant pleomorphic stromal tumor for which a definable line of differentiation has not been found. Because of its highly variable morphologic pattern, MFH has been classified into the following subtypes: pleomorphic, myxoid, giant cell, and inflammatory [1] . Most commonly, MFH arises in the soft tissues of the extremities, and less often in the peritoneum [2]. However, primary MFH localized in the large intestine is extremely rare. There are no reported cases of colorectal MFH which have undergone laparoscopic surgery. We report a first case of MFH in colon that underwent a laparoscopy-assisted colectomy.
Case presentation
A 74-year-old female was referred to our department, Teikyo University Hospital, with a 3-month history of blood stool.
On physical examination no abnormal findings were observed.
Laboratory tests revealed no abnormal data, for example, no anemia (hemoglobin; 11.7 g/dl) and a normal serum carcinoembryonic antigen (CEA) level of 1.7 ng/ml.
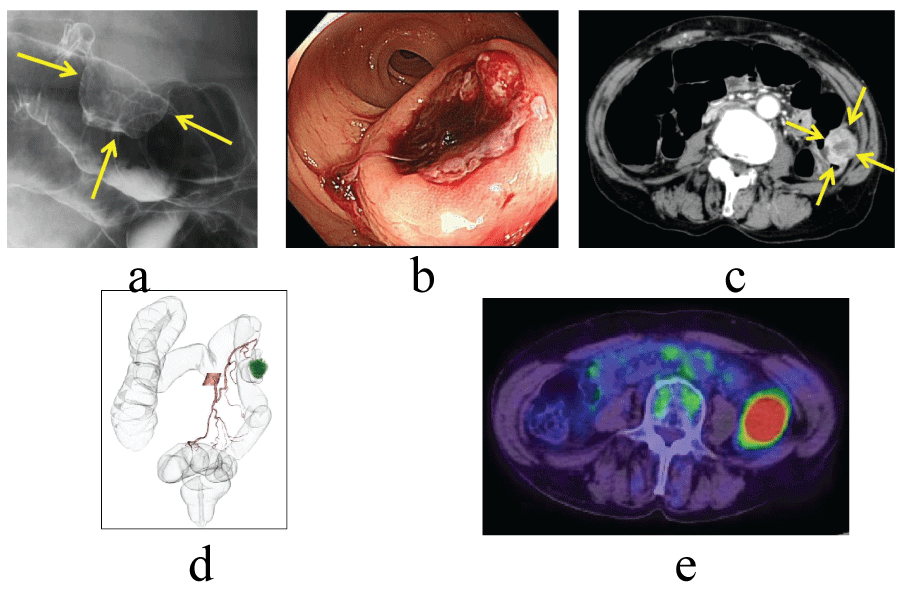
On radiologic investigation, barium enema study showed protrusion, 3.0 cm in diameter, in the descending colon (Figure 1a). Colonoscopy revealed submucosal tumor-like lesion with central ulceration which was easy to bleed (Figure 1b). Abdominopelvic CT scan yielded the thickening of the descending colon wall without any invasion to adjacent tissues and structures (Figure 1c, Figure 1d). A PET/CT using 18F-FDG depicted a tumor with intense FDG uptake in the descending colon (Figure 1e). A biopsy was obtained at colonoscopy and the pathological report was that MFH was suspected.
.
Figure 1: Examination findings. (a) Barium enema showed an intraluminal irregular mass in the descending colon (arrows). (b) Colonoscopy showed a submucosal tumor with central ulceration. (c) CT showed a large mass in the descending colon not extending to the adjacent organs (arrows). (d) CT colonography revealed left colic artery was a feeding vessel. (e) A PET/CT using 18F-FDG depicted a tumor with intense FDG uptake in the descending colon.
View Figure 1
From the findings which showed the tumor of descending colon causing daily bleeding, an operation was thought to be indicated.
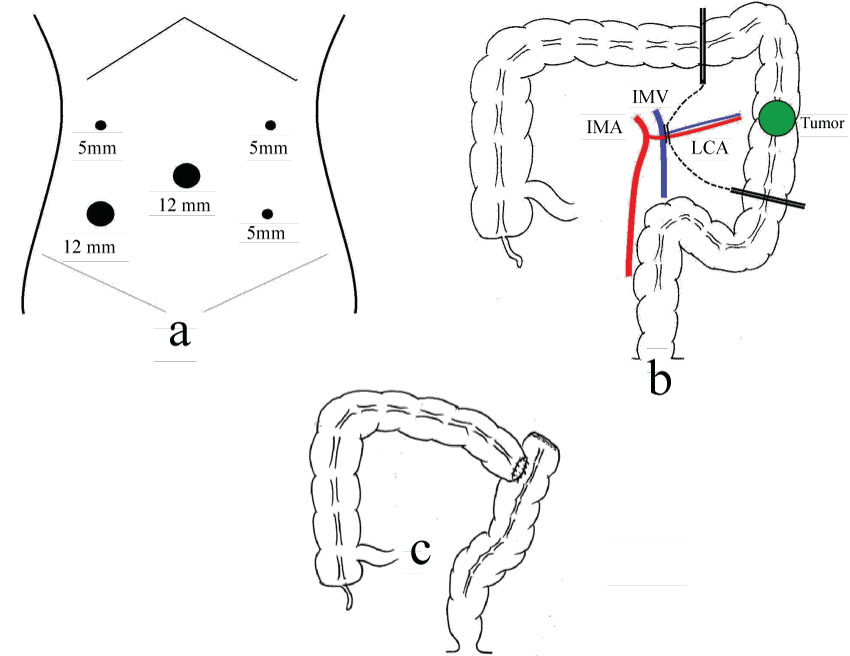
Laparoscopic-assisted left hemicolectomy with en-block regional lymph node dissection and a transverse-sigmoid colon anastomosis was performed. The details were as follows. A camera was inserted into the abdomen through the 12-mm trocar at the umbilicus, and four 5-mm trocars were placed at the left and right and upper and lower quadrants (Figure 2a). A tumor's position was observed, which had been marked by tattooing at a preoperative colonoscopy. There was no evidence of metastasis in the surrounding lymph nodes, omentum, colonic mesentery, liver or the rest of the abdomen. We began mobilization of the sigmoid and descending colon. The division proceeded cephalad and caudal. The left colonic artery and vein were divided by using a vascular clip, respectively. The phrenocolic and splenocolic attachments were divided. The dissection was completed. By extension of the umbilicus trocar opening, 4.5-cm minilaparotomy was performed. The transverse, descending and sigmoid colon was extracted and a resection of colon was performed (Figure 2b). The extent of lymph node dissection was D2 according to the Japanese Classification of Colorectal Carcinoma (8th edition) [3]. End to side anastomosis was performed (Figure 2c). The trocars were removed, and the deep fascias of the ports were closed. Skin incisions were closed by a buried suture. The total time taken for the procedure was 141 min, and the blood loss was 23 ml.
.
Figure 2: Sugical illustrations. (a) Five trocars were placed. (b) The left colonic artery and vein were divided and a resection of colon was performed. (c) End to side anastomosis was performed.
View Figure 2
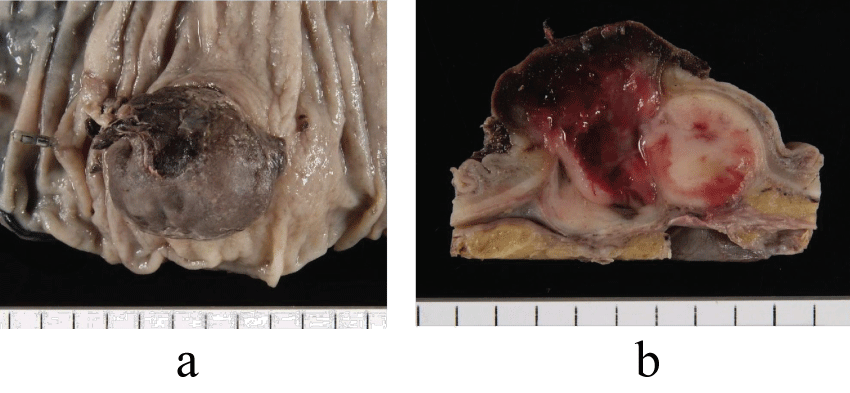
The tumor was a blackish brown and firm nodule projecting into the lumen, measuring 3.5 x 3 cm in diameter (Figure 3a). The cut surface revealed a solid white tumoral tissue with bleeding, which had grown mainly in the muscularis propriae (Figure 3b). The specimen showed proximal margin was negative for tumor, distal margin negative, resected margin negative and 4 lymph nodes negative.
.
Figure 3: Resected specimen. (a) A projecting elastic firm mass was observed. (b) The cut surface revealed a solid tumor with bleeding.
View Figure 3
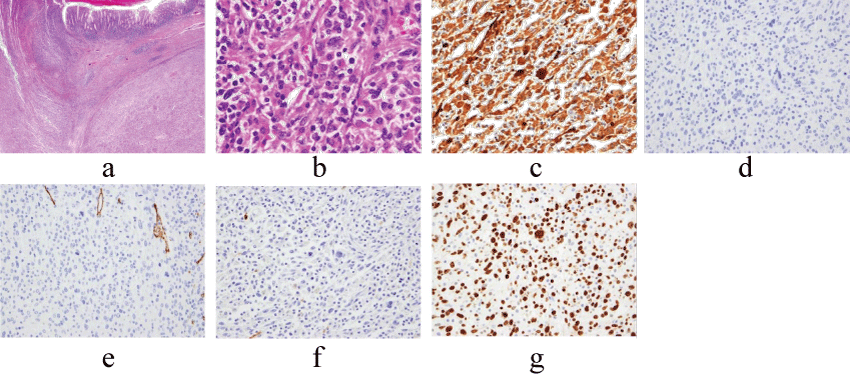
The histopathological report revealed a highly pleomorphic neoplasm composed of large, pleomorphic giant cells as well as spindle cells, arranged in fascicular and storiform pattern mixed with severe inflammatory cells infiltration, including lymphocytes, plasma cells, macrophages, polymorphonuclear leukocytes and multinucleated giant cells (Figure 4a, Figure 4b). Mitotic cells were frequently observed. The lesion was located mainly in the musuclaris propriae spreading to mucosa and subserosa layer. Immunohistochemical (IHC) staining showed positive reaction of tumor cells with CD68, α1-antitrypsin and α1-antichymotrypsin (Figure 4c, Table 1). Cytokeratin, c-kit, desmin, CD34 and S100 were all negative (Figure 4d, Figure 4e, Figure 4f). MIB-1 positive index was 70% (Figure 4g). These findings, in combination with the histomorphology and IHC evaluations, were consistent with MFH of the descending colon. No other primary site was identified (ie, soft tissue) that might render the colon as a metastasis.
.
Figure 4: Histopathological findings. (a,b) The resected specimen revealed that abundant inflammatory cells with histiocyte-like cells(a:hematoxylin and eosin; x12, b: hematoxylin and eosin; x400). (c,d) Immunohistochemical study showed positive reaction for alpha 1-antichymotrypsin(c, x200) but negative for cytokeratin(d, x200), CD34(e, x200) and S100(f, x200). MIB-1 proliferative index was 70% (g, x200).
View Figure 4
![]()
Table 1: Immunohistochemical findings of our case
View Table 1
The patient was treated with no adjuvant chemotherapy. Six months later, at follow-up, no recurrence was detected and surgical incision was clear (Figure 5).
.
Figure 5: A photograph of the patient's abdomen showed a short incision at the navel and 4 trocar incisions, which was taken 6 months after operation.
View Figure 5
Discussion
MFH is a type of soft tissue sarcoma. It was first described in 1963 by Ozzello et al. [4], and later, by O'Brien et al. [5] as a pleomorphic tumor, having both fibroblastic and histiocytic cellular components. WHO classified MFH into undifferentiated pleomorphic sarcoma group [6]. MFH consists of a spectrum of histological appearance and has been classified into the following subtypes: pleomorphic, myxoid, giant cell, and inflammatory [1]. Pleomorphic subtype is the most common, myxoid the second most common. Pathologically, our case was thought to belong to inflammatory type. The most probable differential diagnosis was gastrointestinal stromal tumor (GIST) which was ruled out by negative staining with c-kit. Negative results for cytokeratin also ruled out the poorly differentiated carcinoma and sarcomatoid carcinoma. Roholl et al reported that α1-antitrypsin and α1-antichymotrypsin staining could be used to enhance the pathological diagnosis of MFH [7]. Positive staining with these markers could differentiate between MFH and other soft-tissue tumors (leiomyosarcoma, liposarcoma etc). The immunohistochemical findings in our case are consistent with this study, staining positive with α1-antitrypsin and α1-antichymotrypsin.
On the whole, most MFH cases occur between the ages of 50 and 70; two-thirds of the patients are male, and predominantly Caucasian [2]. MFH typically arises in the extremities(68%), presenting as a painless, enlarging mass [2]. The lower extremity is more frequently involved than the upper extremity. Less commonly, MFH arises in the retroperitoneum (16%), presenting more with constitutional symptoms of weight loss, anorexia and abdominal fullness. It is a high-grade malignancy, with a metastatic rate of 42%, and recurrence rate of 44% [2] . MFH is considered as an aggressive tumor, and treatment with wide local excision or amputation is recommended [2] . Adjuvant chemotherapy or radiation is sometimes employed. At present the role of adjuvant chemotherapy for sarcoma remains unclear. In a study of 200 patients with extraintestinal MFH, 40% died within the first 2 years [2] .
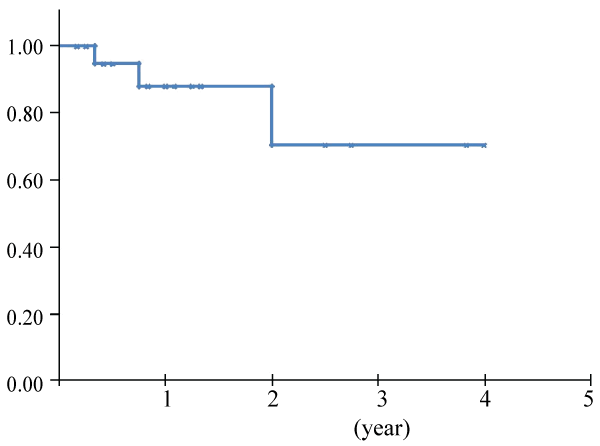
Data about colorectal MFH are yet to be clarified. MFH of the large intestine is extremely rare. Reviewing the available literatures, we found 24 cases of primary colorectal MFH including ours [8-30] (Table 2). The ratio of men to women was 2.4:1, the average age of the patients being 57.4 years, with a rage of 12-84 years. The most common complaints were abdominal pain (42%), bloody stools (17%) and a palpable mass (13%). There were no characteristic complaints as seen with many other cancers. Most of the tumors were large, ranging from 2 to 19 cm in diameter, with an average diameter of 7.6 cm. There were 21 patients with solitary tumors, and 3 with multicentric tumors. The tumors arose from the ascending colon in 6 patients, transverse colon 6, descending colon 4, rectum 4, cecum 2, sigmoid colon 2, and anal canal 2. This distribution is different from that of carcinoma in the large intestine. All the patients, excluding one autopsy case, were treated by surgery, 3 of whom received adjuvant chemotherapy, one radio therapy, and one chemo radiotherapy. Metastases were found at the initial operation in 5 patients; in the peritoneum in 3, the regional lymph nodes in one, and the liver & lymph nodes in one. Only two cases showed lymph node metastasis. After surgery, new recurrence occurred in 4 patients. The distribution of recurrence was found in peritoneum/retroperitoneum in 3 patients, and lung & local in one. No cases showed recurrence of lymph node metastasis. Overall survival was calculated from the data obtained from all references by Kaplan-Meier method (Figure 6). Two year survival rate was 70.4%, which seemed better than that of the former report analyzing mainly MHF of extremity at 60%, reported by Weiss et al. [2] . With respect to the recurrence, MHF of the large intestine and that of extremities might be different with respect to their biological behavior.
.
Figure 6: Overall survival was calculated from the data about colorectal MFH obtained from all references by Kaplan-Meier method.
View Figure 6
![]()
Table 2: Reported cases of MFH
View Table 2
Literally, in MFH of the large intestine, few cases showed lymph node metastasis, and recurrence of peritoneum/retroperitoneum was common (Table 2). In the present case we made a close observation under the laparoscopy, confirmed that the tumor was not invading the adjacent organ and decided to perform the laparoscopic-assisted surgery. Meanwhile the differentiation between MHF and colon cancer was not so easy that D2 dissection of lymph nodes was performed instead of no dissection of them. In MFH cases, D2 dissection is thought to be enough because lymph node metastasis is rare. Actually no lymph node metastasis was observed in the present case. The standard treatment of MFH is a complete surgical resection [9,30]. Judging from the findings of the reported cases and ours, the laparoscopic-assisted surgery would be adapted for colorectal MFH not invading to the adjacent organ.
We referred to MEDLINE searching MFH vs. laparoscopic surgery. Only reports of splenic lesions have been published, but no report about colorectal MFH has been found. Prognosis is difficult to determine due to the lack of available data of colorectal MFH. Intensive follow-up should be performed on patients who underwent surgery for colorectal MFH because recurrence commonly occurs in peritoneum/retroperitoneum.
Conclusions
Primary MFH localized in the large intestine is extremely rare, which requires a surgical resection. Laparoscopic surgery would be an effective treatment for colon MFH. Intensive and careful follow-up should be performed because recurrence commonly occurs in peritoneum/retroperitoneum.
Consent
Written informed consent was obtained from the patient for publication of this case report and any accompanying images. A copy of the written consent is available for review by the Editor-in-Chief of this journal.
Competing Interests
The authors declare that they have no competing interests.
Authors' contributions
KM, SF, RS, and YH diagnosed and performed operation of our patient. The manuscript was prepared by KM under the supervision of YH. All authors read and approved the final manuscript.
References
-
Kempson RL, CD F, HL E (2001) Tumors of the soft tissues. Atlas of tumor pathology, 3rd series: 159-186.
-
Weiss SW, Enzinger FM (1978) Malignant fibrous histiocytoma: an analysis of 200 cases. Cancer 41: 2250-2266.
-
Japanese Society for Cancer of the Colon and Rectum (2013) Japanese Classification of Colorectal Carcinoma, 8th edition: 20.
-
Ozzello L, Stout AP, Murray MR (1963) Cultural characteristics of malignant histiocytomas and fibrous xanthomas. Cancer 16: 331-344.
-
O'Brien JE, Stout AP (1964) Malignant Fibrous Xanthomas. Cancer 17: 1445-1455.
-
Fletcher CD, Bridge JA, Hogendoorn PC (2013) WHO classification of tumors of soft tissue and bone, 4th edition. WHO classification of tumors of soft tissue and bone, 4th edition: 237-238.
-
Rohol PJ, Kleyne J, Elbers H, Van der Vegt MC, Albus-Lutter C, et al. (1985) Characterization of tumour cells in malignant fibrous histiocytomas and other soft tissue tumours in comparison with malignant histiocytes. I. Immunohistochemical study on paraffin sections. J Pathol 147: 87-95.
-
Verma P, Chandra U, Bhatia PS (1979) Malignant histiocytoma of the rectum: report of a case. Dis Colon Rectum 22: 179-182.
-
Sewell R, Levine BA, Harrison GK, Tio F, Schwesinger WH (1980) Primary malignant fibrous histiocytoma of the intestine: intussusception of a rare neoplasm. Dis Colon Rectum 23: 198-201.
-
Levinson MM, Tsang D (1982) Multicentric malignant fibrous histiocytomas of the colon. Report of a case and review of the subject. Dis Colon Rectum 25: 327-331.
-
Rubbini M, Marzola A, Spanedda R, Scalco GB, Zamboni P, et al. (1983) Primary malignant fibrous histiocytoma of the sigmoid colon: a case report. Ital J Surg Sci 13: 299-302.
-
Waxman M, Faegenburg D, Waxman JS, Janelli DE (1983) Malignant fibrous histiocytoma of the colon associated with diverticulitis. Dis Colon Rectum 26: 339-343.
-
Spagnoli LG, Dell'Isola C, Sportelli G, Mauriello A, Rizzo F, et al. (1984) Primary malignant fibrous histiocytoma of storiform-pleomorphic type: a case report of an ano-rectal localization. Tumori 70: 567-570.
-
Kukora JS, Bagnato J, Gatling R (1985) Fibrous histiocytoma of colon and pancreas. Dig Surg 2: 180-184.
-
Baratz M, Ostrzega N, Michowitz M, Messer G (1986) Primary inflammatory malignant fibrous histiocytoma of the colon. Dis Colon Rectum 29: 462-465.
-
Yahagi K, Arai T, Matsumoto J (1986) Malignant fibrous histiocytoma of the colon with marked stenosis. Report of a case. Stomach and Intestine 21: 215-219.
-
Sasaki R, N I, H W (1987) Primary malignant fibrous histiocytoma of the transverse colon. Byoin Byori 5: 62.
-
Satake T, Matsuyama M (1988) Cytologic features of ascites in malignant fibrous histiocytoma of the colon. Acta Pathol Jpn 38: 921-928.
-
Flood HD, Salman AA (1989) Malignant fibrous histiocytoma of the anal canal. Report of a case and review of the literature. Dis Colon Rectum 32: 256-259.
-
Katz RN, Waye JD, Batzel EL, Reiner MA, Freed JS (1990) Malignant fibrous histiocytoma of the gastrointestinal tract in a patient with neurofibromatosis. Am J Gastroenterol 85: 1527-1530.
-
Fukino S, Fukata T, Okano K, Hamasaki T, Inoue A, et al. (1990) [A case of malignant fibrous histiocytoma of the cecum]. Nihon Geka Gakkai Zasshi 91: 1752-1755.
-
Murata I, Makiyama K, Miyazaki K, Kawamoto AS, Yoshida N, et al. (1993) A case of inflammatory malignant fibrous histiocytoma of the colon. Gastroenterol Jpn 28: 554-563.
-
Huang Z, Wei K (1993) Malignant fibrous histiocytoma of the ascending colon in a child. Am J Gastroenterol 88: 972-973.
-
Makino M, Kimura O, Kaibara N (1994) Radiation-induced malignant fibrous histiocytoma of the transverse colon: case report and review of the literature. J Gastroenterol 29: 767-771.
-
Kawashima H, Ikeue S, Takahashi Y, Kashiyama M, Hara T, et al. (1997) Primary malignant fibrous histiocytoma of the descending colon. Surg Today 27: 851-854.
-
Udaka T, Suzuki Y, Kimura H, Miyashita K, Suwaki T, et al. (1999) Primary malignant fibrous histiocytoma of the ascending colon: report of a case. Surg Today 29: 160-164.
-
Singh DR, Aryya NC, Sahi UP, Shukla VK (1999) Malignant fibrous histiocytoma of the rectum. Eur J Surg Oncol 25: 447-448.
-
Okubo H, Ozeki K, Tanaka T, Matsuo T, Mochinaga N (2005) Primary malignant fibrous histiocytoma of the ascending colon: report of a case. Surg Today 35: 323-327.
-
Hagiwara S, Hirayama M, Takahashi T, Meguro T, Morita T, et al. (2008) [Undifferentiated pleomorphic sarcoma detected in the descending colon: report of a case]. Nihon Shokakibyo Gakkai Zasshi 105: 692-698.
-
Azizi R, Mahjoubi B, Shayanfar N, Anaraki F, Zahedi-Shoolami L (2011) Malignant fibrous histiocytoma of rectum: Report of a case. Int J Surg Case Rep 2: 111-113.





