International Journal of Cancer and Clinical Research
Squamous Cell Carcinoma and Squamous Differentiation Histologic Variant of Urothelialcarcinoma of Urinary Bladder: Oncological Outcome Following Definitive Surgical Treatment
Mohamed Wishahi*, HossamElganzoury and Mohamed Badawy
Department of Urology, Theodor Bilharz Research institute, Egypt
*Corresponding author: Mohamed Wishahi, Urology Department, Theodor Bilharz Research Institute, Kornish El-Nil, Embaba, Giza POB 30, Cairo, Egypt, Tel: 2-012-005-56788, E-mail: moh.weshahy@gmail.com
Int J Cancer Clin Res, IJCCR-1-005, (Volume 1, Issue 1), Research Article; ISSN: 2378-3419
Received: August 15, 2014 | Accepted: September 26, 2014 | Published: September 29, 2014
Citation: Wishahi M, Elganzoury H Badawy M (2014) Squamous Cell Carcinoma and Squamous Differentiation Histologic Variant of Urothelialcarcinoma of Urinary Bladder: Oncological Outcome Following Definitive Surgical Treatment. Int J Cancer Clin Res 1:005. 10.23937/2378-3419/1/1/1005
Copyright: © 2014 Wishahi M, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Objectives: To investigate the impact of variant histologic patterns of urothelialcarcinoma of the bladder on oncological outcomes of patients treated with Radical Cystectomy (RC). Three histologic patterns were evaluated: Urothelial Carcinoma (UC), Squamous Cell Carcinoma (SCC), and urothelial carcinoma with Squamous Differentiation (SqD).
Materials and methods: Retrospective archival study included 258 patients treated with RCfor muscle-invasive carcinoma. Data were reviewed in demographic and clinicopathological parameters that included histopathological variant, tumor stage, and nodal status. Overall survival and cancer-specific survival were evaluated. Histologic patterns of these 258 patients were: 90 patients with UC, 112 patients with SCC, and 56 patients with UC with SqD. Archival data were retrieved from 1995 to 2012.
Results: Median followup was 44 months. There were statistically significant differences between survivals in the three groups.Overall survival decreased in UC with SqD. Patients with SCC had a higher risk of local recurrence, while patients with pure UC had a higher risk of metastases.
Conclusion: Squamous differentiation of UC was associated with high tumor stage and high tumor grade. Patients with this histologic variant had less favorable prognosis compared to pure UC and pure SCC. Diagnosis of divert histologic patterns in RC is valuable forplanning a treatment strategy.
Keywords
Carcinoma of the bladder, Urothelial carcinoma, Squamous cell carcinoma, Squamous differentiation, Prognosis, Variant histology, Radical cystectomy.
Abbreviations
UC-Urothelial Carcinoma, SCC-Squamous Cell Carcinoma, UC with SqD-Urothelial Carcinoma with Squamous Differentiation, RC-Radical Cystectomy
Introduction
The most common histological type of carcinoma of the bladder is the urothelialcarcinoma (UC) that comprises 95% of carcinomas. Other cell types of variant histologic pattern are 5% [1]. Approximately 75-85 % of patients hving carcinoma of the bladder with urothelial type present with disease that is confined to the mucosa Ta or CIS, or submucosa T1 [2].
Variant urothelial carcinoma histologies were associated with worse outcome in patients treated with radical cystectomy in univariable analysis [3]. Variant histology of urothelial carcinoma might be a phenotype of high-grade, locally aggressive disease. Variant histology was significantly associated with worse disease specific survival after surgery [4].
Squamous differentiation of urothelial carcinoma showed disease recurrence after radical cystectomy in 64 % of patients compared to 34% in patients with pure UC, Squamous differentiation of UC was an independent prognostic factor for cancer specific survival in patients treated with RC on multivariable analysis [5].
The presence of SqD or SCC with urothelial carcioma in radical cystectomy specimen would indicate an aggressive behavior in the post-operativefollow-up in regard to loco-regional failure and survival [6-8].
The mixed urothelial carcinomas with SqD or SCC were considered an independent factor for recurrence, aggressiveness, and poor survival [9,10].
The different prognosis of mixed tumors of the urinary bladder compared to pureurothelial carcinoma or pure SCC raised awareness for the distinction between pure cell type and mixed primary with SCC or SqD.As this distinction is not detected or not looked for by standard pathological examination of radical cystectomy specimen, led to new immune histochemical technique to detect SqD or SCC [11].
Material and Methods
Patients
The study comprised a retrospective analysis of 258 patients with muscle-invasive carcinoma of the bladder. Patients were treated with radical cystectomy and pelvic lymphadenectomy and urinary diversion. Histopathologigal distribution of the patients was SCC in 90 patients, UC in 112 patients, and UC withSqG in 56 patients. Demographic data is shown in Table 1.
![]()
Table 1: Baseline characteristics and clinicopathologic details of all 258 patients undergoing radical cystectomy for muscle invasive carcinoma of the bladder of three different histologic variant.
View Table 1
Histopathological Evaluation
Archival data were extracted from the affiliated hospital and private hospital archives from 1995 to 2011. Exclusion criteria were the diagnosis of metastatic disease pre-operatively and the histological findings in the cystectomy specimen of positive soft tissue surgical margins, or positive lymph nodes above the common iliac vessels. Inclusion criteria were the pathological diagnosis of the transurethral resection of the bladder tumor of pure UC, pure SCC, or UC with SqD which shows predominance of urothelial carcinoma with varying degrees of cellular differentiation with the presence of keratinized squamous metaplasia, keratinized pearl, intercellular bridges, and formation of small or large nests (Figure 1).
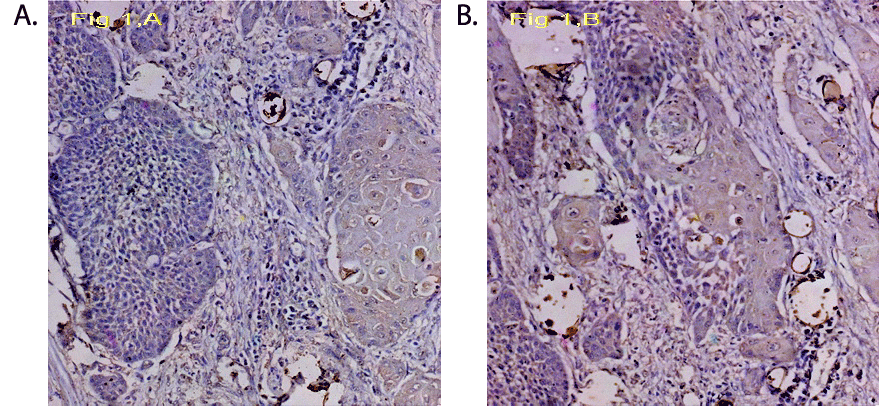
Figure 1a, 1b: Urothelial carcinoma with squamous differentiation showing corneous pearl formation and nest formation. Haematoxylin and eosin stain, magnification x 200.
View Figure 1
Pathologic assessment of the radical cystectomy specimen was done using standard heamatoxylin and eosin staining technique. Examination included mapping of the radical cystectomy specimen by examination of specific sites: Tumour in five sites; adjacent areas; normally looking mucosa; prostatic urethra in men or vaginal stump in women, lower ureters; external iliac lymph nodes on both sides to the bifurcation of the common iliac arteries, perivesical fat of the bladder.
Oncological assessmentFollow up consisted of visits every three months for the first year, visits every 4 months for two years then every 6 months for five years for the survivals, and every year afterwards. Follow- up critical informationwas recurrence-free-survival, cancer specific survival, and pattern of recurrence whether regional, systemic, or regional-systemic.
The recurrence-free-survival period was estimated from the time of surgery to the date of reporting regional or systemic recurrence either clinically, ultrasonographically, CT scan, bone scan, biopsy, or routine urine sample showing malignancy.
Loco-regional recurrence was defined as recurrence in the pelvis, ileal pouch, urethra, pelvic bone including hip bones. Imaging, CT scan, bone scan, or biopsy was used for detecting metastases.
Results
The study comprised 258 patients who had muscle-invasive carcinoma of the bladder and were treated with radical cystectomy, and pelvic lymphadenectomy and urinary diversion. Histologic types of the carcinoma of the bladder in these patients were: pure UC in 90 patients, pure SCC in 112 patients and UC with SqD in 57 patients. Patient's characteristics are shown in (table 1), for gender, age, grade and stage of tomour, and lymph node involvement. It was apparent that the disease is dominant in men in all three histologic variant. Mapping histology of the cystectomy specimen identified 19/56 (34%) cases of UC with SqD. The incidence of lymph nodes involvement was 44.4% in the pure UC group, 46.4% in pure SCC, and 87.5% in the group of UC with SqD.
Urothelial carcinoma with SqD was of high grade in 100% of cases. Pure UC showed high grade in 55.6% of cases, while SCC was of low grade, showing high grade in 19.7% of cases. UC with SqD was of T3-4 tumor stage in 84.7% of cases. SCC was of stage T3-4 in 100% of cases. Pure UC was T3-4 in 72.2% of cases. Follow up following radical cystectomy was up to 5 years. Recurrence rate and metastases are detailed in table 2. At 8 months local recurrence was found in (16.65%) of pure UC cases. Pure SCC was (9.8%), and UC with SqD was 17.8%. At 3 years local recurrence in UC, SCC, and UC with SqD were 27.7%, 24.1%, and 71.4% respectively.
Metastases with or without local recurrence was worse in UC with SqD as it was 84% in 3 years follow-up compared to 33,3% and 40.1% in pure UC and pure SCC respectively.
The overall survival is illustrated in table 2. The decreased overall survival was statistically significant in UC with SqD followed by pure UC and pure SCC. Overall survivals in the three groups of UC, SCC, and UC with SqD in 5 years follow up were 38.8%, 60%, and 10.7 % respectively.
![]()
Table 2: Oncological outcome after radical cystectomy for muscle invasive carcinoma of the bladder in three histologic variant. Recurrence, metastases, and overall survival in 5 years follow up.
View Table 2
Discussion
The spectrum of microscopic forms of UC has been expanded recently to include several histological variants, the recognition of which is important to guide the selection of an appropriate therapeutic approach[11]. The present work emphasizes this statement that proper histological examination of the cystectomy specimen may reveal variant histologic patterns. It shows thatonly mapping histology was able to diagnose the presence of squamous elements in urothelial carcinoma. But also that the presence of squamous differentiation in the urothelial carcinoma has an adverse prognostic factor associated with increased local failure rate and low cancer-specific survival with low overall survival.These findings correlate with the previous reports [4,6,8-10]. The present results show that UC with SqD had a poor cancer- specific- survival and overall survival compared to pure UC. That finding supports previous works [5-7].
Conclusion
There is evidence that cancer specific survival and overall survival are decreasedin patients treated for UC with SqD, rather than in pure UC or pure SCC of the same demographic disease characteristics. Histopathological mapping of radical cystectomy specimen will diagnose the variant histologic pattern of UC with SqD. Immuno histopathological studies may be a clue to the diagnosis of histologic variant.
References
-
Hoskin P, Dubash S (2012) Bladder conservation for muscle-invasive bladder cancer. Expert Rev Anticancer Ther 12: 1015-1020.
-
Babjuk M, Oosterlinck W, Sylvester R, Kaasinen E, Bohle A, et al. (2012) EAU guidelines on non-muscle invasive urothelial carcinoma of the bladder, the 2011 update. Eur Urol 59: 997-1008.
-
Xylinas E, Rink M, Robinson BD, Lotan Y, Babjuk M, et al. (2013) Impact of histological variants on oncological outcomes of patients with urothelial carcinoma of the bladder treated with radical cystectomy. Eur J Cancer 49: 1889-1897.
-
Sakano S, Matsuyama H, Kamiryo Y, Hayashida S, Yamamoto N, et al. (2014) Impact of variant histology on disease aggressiveness and outcome after nephroureterectomy in Japanese patients with upper tract urothelial carcinoma. Int J Clin Oncol .
-
Antunes AA, Nesrallah LJ, Dall'Oglio MF, Maluf CE, Camara C, et al. (2007) The role of squamous differerentiation in patients with transitional cell carcinoma of the bladder treated with radical cystectomy. Int Braz J Urol 33: 339-345.
-
Ehdaie B, Maschino A, Shariat SF, Rioja J, Hamilton RJ, et al. (2012) Comparative outcomes of pure squamous cell carcinoma and urothelial carcinoma with squamous differentiation in patients treated with radical cystectomy. J Urol 187: 74-79.
-
Rogers CG, Palapattu GS, Shariat SF, Karakiewicz PI, Bastian PJ, et al. (2006) Clinical outcomes following radical cystectomy for primary nontransitional cell carcinoma of the bladder compared to transitional cell carcinoma of the bladder. J Urol 175: 2048-2053.
-
Antunes AA, Nesrallah LJ, Dall'Oglio MF, Maluf CE, Camara C, et al. (2007) The role of squamous differentiation in patients with transitional cell carcinoma of the bladder treated with radical cystectomy. Int Braz J Urol 33: 339-345.
-
Wasco MJ, Daignault S, Zhang Y, Kunju LP, Kinnaman M, et al. (2007) Urothelial carcinoma with divert histologic differentiation (mixed histologic features) predict the presence of locally advanced bladder cancer detected at transurethral resection. Urology 70: 69-74.
-
DeGraff DJ, Cates JM, Mauney JR, Clark PE, Matusik RJ, et al. (2013) When urothelial differentiation pathways go wrong: implications for bladder cancer development and progression. Urol Oncol 31: 802-811.
-
Hayashi T, Sentani K, Oue N, Anami K, Sakamoto N, et al. (2011) Desmocollin 2 is a new immunohistochemical marker indicative of squamous differentiation in urothelial carcinoma. Histopathology 59: 710-721.
-
Stenzl A, Cowan NC, De Santis M, Kuczyk MA, Merseburger AS, et al. (2011) Treatment of Muscle-invasive and metastatic bladder cancer: update of the EAU guidelines. EurUrol 59: 1009-1018.





