International Journal of Blood Research and Disorders
The Evaluation of Serum Protein and Serum Immunofixation Electrophoresis Results in Patients with Monoclonal and Polyclonal Gammopathy: A Single Center Experience
Koksal Deveci1, Serdal Korkmaz2*, Enver Sancakdar1, Duygu Acibucu1, Filiz Alkan1, Hatice Terzi3 and Mehmet Sencan3
1Cumhuriyet University Faculty of Medicine, Department of Medical Biochemistry, Sivas, Turkey
2Kayseri Education and Research Hospital, Division of Hematology, Kayseri, Turkey
3Cumhuriyet University Faculty of Medicine, Department of Hematology, Sivas, Turkey
*Corresponding author:
Serdal Korkmaz, Associated Prof. M.D. Kayseri Education and Research Hospital, Division of Hematology, 38100, Kayseri, Turkey, Tel: +90 352 336 88 88, E-mail:
baranserdalkorkmaz@gmail.com
Int J Blood Res Disord, IJBRD-2-017, (Volume 2, Issue 2), Research Article; ISSN: 2469-5696
Received: August 12, 2015 | Accepted: September 15, 2015 | Published: September 17, 2015
Citation: Deveci K, Korkmaz S, Sancakdar E, Acibucu D, Alkan F, et al. (2015) The Evaluation of Serum Protein and Serum Immunofixation Electrophoresis Results in Patients with Monoclonal and Polyclonal Gammopathy: A Single Center Experience. Int J Blood Res Disord 2:017. 10.23937/2469-5696/1410017
Copyright: © 2015 Deveci K, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Objectives: To survey the diagnostic performance of serum protein electrophoresis (SPE) and serum immunofixation electrophoresis (SIFE) in clinical practice of gammopathies.
Methods: One hundered and fifty five patients were evaluated by a retrospective review of our patient records to demostrate the SPE and SIFE results performed by the Clinical Biochemistry. The patients were divided into three groups as monoclonal gammopathy group (MGG), polyclonal gammopathy group (PGG) and biclonal gammopathy group (BGG).
Results: Of the 155 patients, 48 (31%) were in MGG, 104 (67.1%) were in PGG and 3 (1.9%) were in BGG. In totally, there was an M spike in the gamma region in 113 patients, and of these patients, only 49 (43.4%) have been diagnosed with Multiple Myeloma. Of the 49 patients, 38 (79.1%) were in MGG, 8 (7.7%) were in PGG, and 3 (100%) were in BGG. When SPE plus SIFE were the testing panel, 11 (22.5%) patients were missed. The cutoff values were found as 7.25 for total protein (AUC = 0.728; p < 0.05) and 70.5 for erythrocyte sedimentation rate (AUC = 0.692; p < 0.05).
Conclusions: We demostrate that no single clinical laboratory test has sufficient sensitivity for the spectrum of plasma cell disorders. Because of missed 11 (22.5%) patients with myeloma in our study, a SPE be performed at the same time with SIFE results should be interpreted carefully.
Keywords
Serum protein electrophoresis, Serum immunofixation electrophoresis, Monoclonal gammopathy, Biclonal gammopathy, Polyclonal gammopathy
Introduction
A monoclonal gammopathy is defined as the electrophoretically and antigenically homogeneous protein product of a single clone of B lymphocytes and/or plasma cells that has proliferated beyond the constraints of normal control mechanisms [1]. Monoclonal gammopathies are detected in serum and/or urine from individuals with a wide variety of neoplastic, potentially neoplastic, neurological and infectious conditions [2]. It is extremely important to differentiate monoclonal from polyclonal gammopathies. Monoclonal gammopathies are associated with a clonal process that is malignant or potentially malignant. In contrast, polyclonal gammopathies may be caused by any reactive or inflammatory process, and they usually are associated with nonmalignant conditions such as infections, connective tissue diseases, liver diseases and other inflammatory conditions [3].
The diagnostic serologic tests for identifying a monoclonal or a polyclonal gammopathy includes serum and urine protein electrophoresis, serum and urine immunofixation electrophoresis, and quantitative serum free light chain (SFLC) [4]. Serum protein electrophoresis (SPE) is widely used in clinical laboratories, especially for the detection and identification of paraproteins [1]. Whereas SPE can not definitively identify a restriction as an M-protein. For that reason, immunochemical methods together with electrophoresis must be employed. Because of the increased sensitivity for plasma cell disorders, most laboratories now perform SPE plus serum immunofixation electrophoresis (SIFE) assays [5]. The aim of this study is to survey the diagnostic performance and the usefulness of these methods in our clinical practice.
Materials and Methods
One hundered and fifty five patients, whom referred to the Hematology Clinic of the School of Medicine of the Cumhuriyet University (Sivas, Turkey) between January 2006 and April 2013, whose SPE and SIFE tests performed by the Clinical Biochemistry, were included to the study. Patient records were evaluated by a retrospective review to demostrate the age, gender and definitive diagnosis. SPE and SIFE test results were obtained from the biochemistry laboratory archive. SFLC analysis was not available in our clinic at the time of the study. The study has a retrospective design. So, we did not receive the approval of Ethics Committee for Clinical Research of Cumhuriyet University School of Medicine. The study was in accordance with the Declaration of Helsinki.
The patients were divided into three groups according to the SIFE results as monoclonal gammopathy group (MGG), polyclonal gammopathy group (PGG) and biclonal gammopathy group (BGG). Also the patients' admittance serum total protein, serum albumin, C-reactive protein, erythrocyte sedimentation rate (ESR) and white blood cell values were recorded. The albumin/total protein ratio were calculated by dividing serum albumin levels to serum total protein levels.
The SPE assay was performed by agarose gel electrophoresis in a semiautomated SAS-1Plus/SAS-2/Platinum device (Helena Biosciences, Tyne and Wear, England). The bands were found as % ratio and these results were included into one of the three groups, referred to as low, normal and high according to the reference values. The required analitic sensitivity for each band was 0.3 g/L (30 mg/dL) in SPE for visualization a band in SIFE assay. The SIFE assessed migration patterns for IgG, IgA, IgM, kappa (κ), and lambda (λ) immunoglobulin chains in a semiautomated SAS-1Plus/SAS-2/Platinum device (Helena Biosciences, Tyne and Wear, England).
Statistical methods
All statistical analysis were performed with the Statistical Package for the Social Sciences (SPSS) 15.0 Package (SPSS Inc., Chicago, IL, USA). Descriptive statistics were presented as arithmetic mean ± standard deviation. Results which did not follow normal distribution are expressed as median (range) values. For the tests of normality, we used the Kolmogorov-Smirnov test. The significance of the mean differences between groups was assessed by Student's t-test and Mann Whitney U test. Differences were assessed by Chi-squared test for categorical variables. Relationships between variables were tested using Pearson's correlation analysis. ROC Curve graphics were used in the comparison of sensitivity and specificity. P values of less than 0.05 were regarded as significant.
Results
Of the 155 patients, 48 (31%) were in MGG, 104 (67.1%) were in PGG and 3 (1.9%) were in BGG. There was not significant statistical differences between ages of MGG and PGG (66.7 ± 11.3 years vs 63.2 ± 18.0 years, respectively; p = 0.058). And also, we did not obtain significant statistical differences between the gender of MGG and PGG (M/F = 31/19 vs M/F = 53/51, respectively; p = 0.056). The detailed results of SPE between MGG and PGG are outlined in table 1. Also, the detailed SIFE results of each groups were summarized in table 2.
![]()
Table 1: The comparison of results of SPE in MGG and PGG.
View Table 1
![]()
Table 2: The SIFE results of MGG, PGG and BGG.
View Table 2
Since we distinguished the groups as monoclonal, polyclonal and biclonal, then obtained the clinical equivalents of these laboratory results and displayed in table 3. And then we compared the routine laboratory results of the patients in MGG and PGG groups, and obtained significance statistical differences between MGG and PGG in terms of total protein, albumin, albumin/total protein ratio and ESR. All these data are demonstrated in table 4.
![]()
Table 3: The clinical correlation of MGG, PGG and BGG according to the SIFE results.
View Table 3
![]()
Table 4: The comparison of routine laboratory results of MGG and PGG.
View Table 4
To discriminate the patients of MGG and PGG, the performances of total protein, albumin/total protein ratio and ESR tests were determined by ROC analysis. The cutoff values were found as 7.25 for total protein (AUC = 0.728; p < 0.05), 0.5 for albumin/total protein ratio (AUC = 0.165; p > 0.05) and 70.5 for ESR (AUC = 0.692; p < 0.05) (Table 5, Figure 1).
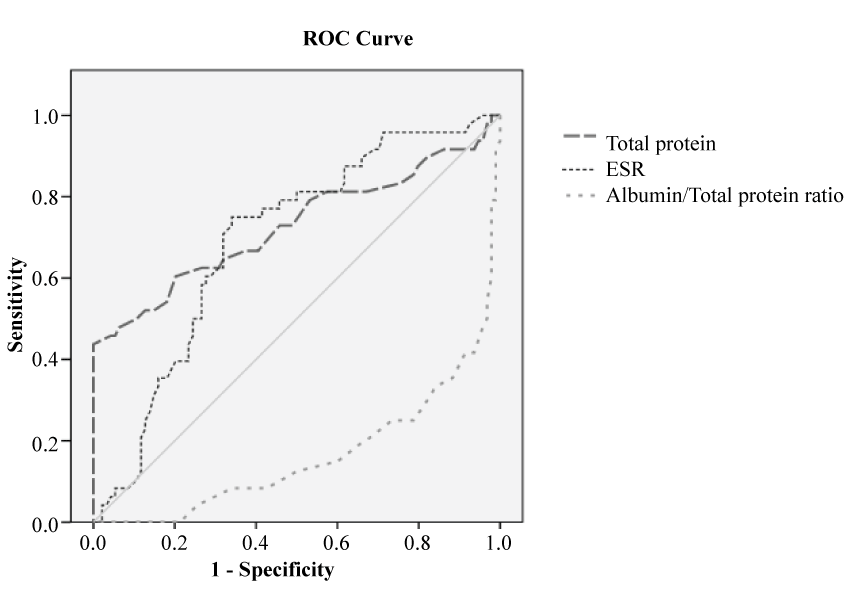
.
Figure 1: The ROC curves showing the performances of total protein, albumin/total proteinratio and ESR.
View Figure 1
![]()
Table 5: The cutoff, Arae Under Curve (AUC), sensitivity and 1-specificity of serum total protein, albumin/total protein ratio and ESR tests.
View Table 5
Discussion
The major structural and functional molecules produced by cells are proteins [6]. Proteins are composed of individual units called amino acids. By altering the pH of an aqueous solution, the charge on an amino acid can be changed [6]. The term 'electrophoresis' refers to the migration of charged particles in an electrical field [7]. It was soon recognized that when tissues responsible for the synthesis or excretion of proteins were altered by disease, the resulting serum would produce distinctive electrophoretic patterns that could be helpful in diagnosis. Detection of monoclonal gammopathies in serum and urine is one of the most important uses of clinical protein electrophoresis.
An M (monoclonal) protein, also called as paraprotein or monoclonal component, is characterized by the presence of a sharp, well-defined band with a single heavy chain and a similar band with a kappa or lambda light chain [8]. A monoclonal gammopathy is defined as the electrophoretically and antigenically homogeneous protein product of a single clone of B lymphocytes and/or plasma cells that has proliferated beyond the constraints of normal control mechanisms [8]. A polyclonal gammopathy is characterized by a broad diffuse band with one or more heavy chains and kappa and lambda light chains [8]. Biclonal gammopathies are a group of disorders characterized by the production of 2 distinct monoclonal proteins. The presence of 2 monoclonal proteins may be because of the proliferation of 2 clones of plasma cells, each producing an unrelated monoclonal immunoglobulin, or it may result from the production of 2 monoclonal proteins by a single clone of plasma cells [9]. In our study group, 48 (31%) patients were in MGG, 104 patients (67.1%) were in PGG and 3 patients (1.9%) were in BGG.
The diagnostic serologic tests for identifying a monoclonal gammopathy includes serum and urine protein electrophoresis, serum and urine immunofixation electrophoresis, and quantitative SFLC [4]. SPE is widely used in clinical laboratories, especially for the detection and identification of paraproteins [1]. SPE is a simple laboratory technique where the serum is applied on a support medium and exposed to an electric current. The different fractions of the serum proteins separate usually into 5 bands, as the albumin, alpha-1 (α1), alpha-2 (α2), beta (β), and the gamma (γ) globulin fractions. Many conditions can cause an increase in the γ region, but those which cause a homogenous spike like a peak in the γ globulin zone which is mainly composed of immunoglobulin, are of special interest. These so called monoclonal gammopathies, result from the proliferation of a single, usually malignant clone of plasma cells which produce either a single class of intact immunoglobulins, heavy chains or light chains or both. The M protein is readily detected as a sharp symmetric spike (M spike) while performing the electrophoresis of serum. In our study population, 42 (84%) of the cases had an M band in the γ region in MGG, and 71 (68%) of the cases had an M band in the γ region in PGG. Chopra et al. [10]. reported that, 84.8% of the cases had an M band in the γ region and that 15.2% cases had an M spike in the β globin region. We could not obtain an M spike in the β globin region. In totally, there was an M spike in the γ region in 113 patients, and of these patients, only 49 (43.4%) have been diagnosed with multiple myeloma (MM). This means that SPE can detect restrictions that resemble monoclonal gammopathies, but it can not definitively identify a restriction as an M protein. SIFE is used to confirm their presence and determine their subtype. For that reason, most laboratories now perform SPE together with SIFE to identify the restriction as an M protein. On the other hand, 38 (79.1%) of the patients were diagnosed with MM in MGG by SIFE results. Namely, SIFE assay did not perform well as a single test, and also when SPE plus SIFE were the testing panel, 11 (22.5%) patients were missed. Because, 8 (7.7%) of the patients were diagnosed with MM in PGG, and 3 (100%) patient were diagnosed with MM in BGG. Katzman et al. [11] suggested that there is not a single clinical laboratory test has sufficient sensitivity for the spectrum of plasma cell disorders. In their study, of the 1877 patients, 26 were negative in all assays. When the omission of SFLC lost 30 patients (6 MM, 23 primary amyloidosis, and 1 light chain deposition disease) and the omission of SIFE lost an additional 58 patients [44 monoclonal gammopathy of undetermined significance (MGUS), 7 POEMS syndrome, 5 primary amyloidosis, 1 smoldering multiple myeloma, and 1 plasmacytoma]. If we could not have performed bone marrow biopsy and have reexamined the clinical features of these patients, the diagnosis of MM would have been bypassed. So, if there is a strong suspicion of a plasma cell disorder, SPE together with SIFE and/or SFLC results should be interpreted carefully.
The monoclonal gammopathies include malignant conditions like plasma cell dyscrasias, chronic lymphatic leukaemias and benign idiopathic forms of unknown significance. They may be associated with the drug treatment (diphenyl hydantoin, sulphonamide and penicillin) [10]. In the great majority of cases, however, monoclonal gammopathy is not accompanied by any physical sign or laboratory abnormality. These cases are defined as MGUS. However, subjects with MGUS do have, on average, a 1% per year lifelong risk of progression to MM or other lymphoproliferative disease [12]. In our study, 2 (4.2%) patients were diagnosed with MGUS.
Once a monoclonal gammopathy is identified, MM must be differentiated from other causes of this type of gammopathy [8,13]. MM is a rare and incurable malignant disorder characterized by uncontrolled proliferation of clonal plasma cells causing a wide variety of complications leading to organ dysfunction and eventually death [14]. These abnormal plasma cells secrete abnormal immunoglobulin that produces a condition called monoclonal gammopathy, which can be detected by the presence of M-protein in serum and urine electrophoresis [15]. Because of increased immunoglobulins, the ESR is high: > 20 mm/h in 85% and > 100 in one third [16]. In our study, the cutoff value of ESR was 70.5 mm/h and there was a significant difference between MGG and PGG (83,9 ± 32,2 mm/h vs 58,5 ± 40.2 mm/h, respectively; p < 0.001). Most frequently, patients with MM have markedly elevated γ globulin regions with a restriction in the migration. MM is the most common cause of paraproteinaemia and an M protein and/or light chain in the serum and/or urine is found in 97% of the patients. About 3% of patients have non-secretory myeloma. MM is classified into different types depending on their immunoglobulin heavy chain and light chain as follows: IgG (52%), IgA (21%), light chain (16%), Bi-clonal (2%), and IgM (0.5%), while IgD and IgE are rare [16]. In our study, types of MM determined by SIFE were as follows: IgG (65%) and IgA (12%). We could not detect isolated IgD, IgE and light chain myeloma because of the lack of related immunoglobulins and SFLC assay.
Since we compared the MGG and PGG in terms of other routine laboratory tests, we obtained significant statistical differences in terms of total protein, albumin, albumin/total protein ratio and ESR. But, the keypoint was the significance level of diagnostic performances of total protein and ESR tests which were analysed by ROC analysis. According to this, if, the cutoff value of total protein is above 7.25 g/dl and the cutoff value of ESR is above 70.5 mm/h, to perform SIFE is useful in combination with SPE for diagnosing a plasma cell disease.
The limitations of our study were; minority of the number of patients in MGG and impossibility of performing SFLC assay. A SFLC assay is useful in detecting patients with no monoclonal bands on SPE and/or SIFE [5]. May be the number of myeloma patients would be higher than the current status, if we could perform SFLC. And also, we could not detect both IgD and IgE myeloma because of the lack of IgD and IgE immunoglobulins in our laboratory. Katzman et al. [17] reported that the use of SPE, SIFE, and SFLC as a diagnostic screen was a sensitive approach for identifying monoclonal gammopathies [17]. So, a SPE be performed at the same time with SIFE should be compared with SFLC can be recommended to improve the identification of M protein [18-20].
In conclusion, SPE is an easy to perform laboratory test which can be used for the detection and the quantification of paraproteinemia. It should be recommended as a preliminary test for the suspected cases of MM. Immunofixation is more sensitive than SPE for detecting the monoclonal immunoglobulins and to identify the heavy or light chain isotype. SFLC assay is useful in detecting patients with no monoclonal bands, but we could not detect any light chain myeloma due to a lack of SFLC in our laboratory. But also, there is a fact that no single clinical laboratory test has sufficient sensitivity for the spectrum of plasma cell disorders. Because of missed 11 (22.5%) patients with myeloma in our study, assays of SPE and SIFE should be interpreted together with correlating the bone marrow biopsy and the clinical features of the suspected patients.
Authors' Contributions
KD and SK, ideated this article and did most of the writing, supported by MS. ES, DA, FA and HT have made substantial contributions to acquisition of data. All authors read and approved the final manuscript.
References
-
Datiles TB, Hunphrey RL (1990) Hypergammaglobulinemia. In: Sheehan C ed. Clinical Immunology. Principles and laboratory diagnosis. JB Lippincott: Philadelphia, 275-289.
-
Kyle RA (2001) Monoclonal gammopathies. In: Rich RR (ed) Clinical Immunology principles and practice. New York: Mosby, 29-37.
-
Dispenzieri A, Gertz MA, Therneau TM, Kyle RA (2001) Retrospective cohort study of 148 patients with polyclonal gammopathy. Mayo Clin Proc 76: 476-487.
-
International Myeloma Working Group (2003) Criteria for the classification of monoclonal gammopathies, multiple myeloma and related disorders: a report of the International Myeloma Working Group. Br J Haematol 121: 749-757.
-
Dispenzieri A, Kyle R, Merlini G, Miguel JS, Ludwig H, et al. (2009) International Myeloma Working Group. International Myeloma Working Group guidelines for serum-free light chain analysis in multiple myeloma and related disorders. Leukemia 23: 215-224.
-
Jones AJS (1993) Analysis of polypeptides and proteins. Adv Drug Del Rev 10: 29-90.
-
Serwer P (1983) Agarose gels: properties and use for electrophoresis. Electrophoresis 4: 375-382.
-
George ED, Sadovsky R (1999) Multiple myeloma: recognition and management. Am Fam Physician 59: 1885-1894.
-
Kyle RA, Robinson RA, Katzmann JA (1981) The clinical aspects of biclonal gammopathies. Review of 57 cases. Am J Med 71: 999-1008.
-
Chopra GS, Gupta PK, Mishra DK (2006) The evaluation of suspected monoclonal gammopathies: the experience in a tertiary care hospital. MJAFI 62: 134-137.
-
Katzmann JA, Kyle RA, Benson J, Larson DR, Snyder MR, et al. (2009) Screening panels for detection of monoclonal gammopathies. Clin Chem 55: 1517-1522.
-
Kyle RA, Therneau TM, Rajkumar SV, Offord JR, Larson DR, et al. (2002) A long-term study of prognosis in monoclonal gammopathy of undetermined significance. N Engl J Med 346: 564-569.
-
Bigos SJ, Richard Bowyer RO, Brown K, Deyo R, Haldeman S, et al. (1994) Acute low back problems in adults. Rockville, Md: U.S. Dept. of Health and Human Services, Public Health Service, Agency for Health Care Policy and Research, 1994; clinical practice guideline no. 14, AHCPR publication no. 95-0642.
-
Al-Farsi K (2013) Multiple myeloma: an update. Oman Med J 28: 3-11.
-
Abdalla IA, Tabbara IA (2002) Nonsecretory multiple myeloma. South Med J 95: 761-764.
-
Kyle RA Gertz MA, Witzig TE, Lust JA, Lacy MQ, et al. (2003) Review of 1027 patients with newly diagnosed multiple myeloma. Mayo Clin Proc 78: 21-33.
-
Katzmann JA, Dispenzieri A, Kyle RA, Snyder MR, Plevak MF, et al. (2006) Elimination of the need for urine studies in the screening algorithm for monoclonal gammopathies by using serum immunofixation and free light chain assays. Mayo Clin Proc 81: 1575-1578.
-
Katzmann JA, Clark RJ, Abraham RS, Bryant S, Lymp JF, et al. (2002) Serum reference intervals and diagnostic ranges for free kappa and free lambda immunoglobulin light chains: relative sensitivity for detection of monoclonal light chains. Clin Chem 48: 1437-1444.
-
Bradwell AR, Carr-Smith HD, Mead GP, Tang LX, Showell PJ, et al. (2001) Highly sensitive, automated immunoassay for immunoglobulin free light chains in serum and urine. Clin Chem 47: 673-680.
-
Tate JR, Gill D, Cobcroft R, Hickman PE (2003) Practical considerations for the measurement of free light chains in serum. Clin Chem 49: 1252-1257.





