International Journal of Blood Research and Disorders
The Comparison between Microhematocrit and Automated Methods for Hematocrit Determination
Gebrewahd Gebretsadkan, Kebede Tessema, Henock Ambachew and Misganaw Birhaneselassie*
Department of Medical Laboratory Sciences, Hawassa University College of Medicine and Health Sciences, Ethiopia
*Corresponding author:
Misganaw Birhaneselassie, Hawassa University College of Medicine and Health Sciences, Department of Medical Laboratory Sciences, Hawassa, Ethiopia, E-mail: misganawbs@gmail.com
Int J Blood Res Disord, IJBRD-2-012, (Volume 2, Issue 1), Research Article; ISSN: 2469-5696
Received: February 25, 2015 | Accepted: March 30, 2015 | Published: April 01, 2015
Citation: Gebretsadkan G, Tessema K, Ambachew H, Birhaneselassie M (2015) The Comparison between Microhematocrit and Automated Methods for Hematocrit Determination. Int J Blood Res Disord 2:012. 10.23937/2469-5696/1410012
Copyright: © 2015 Gebretsadkan G, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Background: Microhematocrit (HCT) method is a gold standard method for hematocrit determination. A comparative cross sectional study was conducted from 28 April to 28 June, 2014 to assess the analytical performance between microhematocrit and automated methods for hematocrit determination.
Methodology: A total of 384 EDTA (Ethyl Diamine Tetra Acetic Acid) collected blood samples from patients attending Yirgalem hospital were analyzed for HCT by both methods. The data for hematocrit value was analyzed by linear regression and Pearson correlation co- efficient. The result of the two methods was expressed as a mean SD (standard deviation).
Result: The correlation coefficient (R=0.95) indicated the strong correlation between manual and automated methods to determine the hematocrit. The manual HCT and automated HCT were significantly different (P< 0.002) at 95% confidence interval. The result indicated higher coefficient of variation (CV) in manual method than automated HCT results, which implicated the percision is good for automated method (mindray 3000 plus) and not good for manual method.
Conclusion: Generally, the study showed the hematocrit value obtained from hematology analyzer (mindray 3000 plus) is different from that of manual, but it is directly proportional in most cases. The automated method can not replace the manual for hematocrit determination though the result of both methods are close to each other.
Keywords
Packed cell volume, Microhematocrit, Automated method
Background
Hematocrit is a test that measures a percentage of blood that is comprised of red blood cell. This is often referred to as packed cell volume (PCV) or erythrocyte volume fraction. It is considered as an integral part of a person�s complete blood count, along with hemoglobin concentration, white blood cell count and platelet counts [1,2].
The measurement of the packed cell volume (PCV) is useful in any hematologic workup and is a main tool in the quality control programs in the hematology laboratory. Incorrectly reported HCT result may bias clinical decision in follow up of patients, blood transfusion decision, and in diagnosis of hematologic diseases such as severe anemia. In spite of its significance it has received far less consideration in research from the standpoint of its reliability than have the measurements of hemoglobin or red cell counts [3,4].
In Ethiopia, HCT (Hematocrit) is one of the most common CBC (Complete Blood Count) parameters routinely used by clinicians and in most parts of the country where automated analyzers are unavailable microhematocrit method is used to determine the HCT value of patients [5].
Microhematocrit method is a gold standard method for hematocrit determination but it associates many problems that may lead to inaccurate and imprecise measurements. Spun hematocrit is 1% to 3% higher than the hematocrit from automated instrument due to plasma that is trapped in erythrocytes. In the normal situation, spun hematocrit, however, may give spuriously higher results (up to 6%) in a number of disorders including polycythemia, macrocytosis, spherocytosis, hypochromic anemia�s, sickle cell anemia, burn patients due to increase trapped plasma compared to the normal condition, of course insufficient centrifugation can also introduce high spun hematocrit [1,6-8].
The interferences that may cause erroneous results of red blood cell (RBC) and mean cell volume (MCV) by automated hematology analyzer could introduce abnormal hematocrit, which include very high white blood cell count, high concentration of platelets, or agglutinated RBCs. From the stand point that the difference in values between the automated and manual methods may impact clinical decision, it would be essential to compare the two methods hematocrit values. Therefore, the study results in the determination of correction, correlation and precision of the two methods; micro hematocrit method and automated hematology (mindray 3000 plus) analyzer. The result of the study is important to reduce ambiguity during hematocrit determination and to get correct hematocrit values.
Methodology
Study area
Yirgalem town is a town in Southern Ethiopia, located 315kms South of Addis Ababa and 40kms South of Hawassa in the Sidama zone of the Southern Nations, Nationalities and peoples region. The town has a latitude and longitude of 6�,45' N and 38�,25'E and elevation of 1776 meters and it is the largest settlement in Dalle Woreda. Based on figures from the central statistical agency in Ethiopia published in 2005, Yirgalem town has an estimated total population of 43,815 of whom 21,840 are men and 21,975 are women (Figure 1).
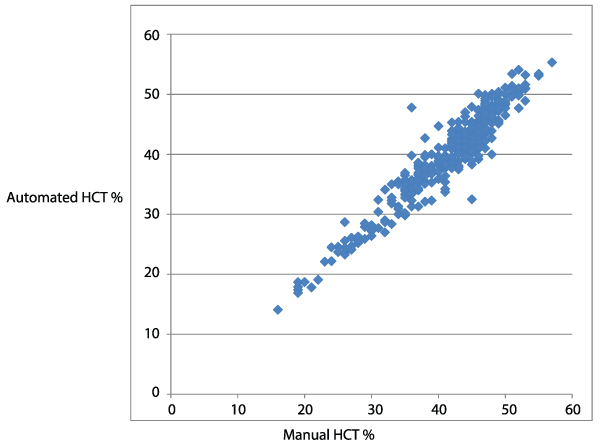
Figure 1: A Scatter plot showing the correlation of automated and manual hematocrit (R=0.95, P< 0.002)
View Figure 1
Study design
A comparative cross sectional study was conducted from 28 April to 28 June, 2014 to assess the analytical performance between microhematocrit and automated methods for hematocrit determination. * Mindray BC- 3000 plus is automated hematology analyzers on which study was done. These machines use impedance method to determine WBCs, RBCs and PLTs. These machines do not directly measure the hematocrit, but rather calculate hematocrit from measurements of individual RBCs sizes and counts. The hematocrit of the original sample is calculated from the number of cells (rbcs) by using the following equation: Hct= number of RBCs X MCV/10.
Venous blood was obtained from patients of both sexes and all age group attending at Yirgalem general hospital during study period. Blood samples which have improper proportion of anticoagulant, strongly lipemic, hyperbilirubinemic, and hemolyzed sample were excluded. Assuming 95% level of confidence, 5% margin of error, and taking P=0.05, since no known information about P-value. The sample size calculated to conduct the study was, 384.
Data collection method
About 2-3 millimeters of blood from vein of selected patients was collected in to a tube containing EDTA anticoagulant. The percentage of packed cell volume was measured manually by filling a capillary tube (plain) and sealing with modeling clay and centrifuging at 3000g for 5 minutes, then the result was read using hematocrit reader. A result from the Mindray automated analyzer was obtained by giving well-mixed EDTA blood to the analyzer. The HCT values of both methods were properly registered for analysis.
Statistical analysis
The results obtained from manual hematocrit and automated hematology analyzers were entered in Excel for analysis. The data for hematorit was analyzed by linear regression and Pearson correlation co- efficient. The result of the two methods was expressed as a mean � SD. Precision was determined using coefficient of variation and the significance of the value was decided based on the p-value [0.05] at 95% confidence
Ethical consideration
Written permission letter was taken from Hawassa University College of medicine and health science (community based education) CBE office and also from Yirgalem general hospital. During the course of study patients� rights was kept and optimal amount of sample was taken. The hematocrit value of patients obtained during the study was automatically reported to the (requesting physician) and concerned supervisors.
Result
A comparison of automated and manual method to determine the hematocrit for 384 patients was shown by mean � SD in Table 1. The correlation coefficients for relationships between the manual and automated (mindray 3000 plus) is calculated by using pearsons correlation coefficient formula, which was R=0.95. The correlation coefficient (R=0.95) indicated the strong positive correlation between manual and automated methods to determine the hematocrit. The mean � SD of HCT result by manual method is 42 � 7.1, whereas that of the automated method is 40.48 � 7.4. This implicated, the manual HCT and automated HCT were significantly different (P< 0.002) which is less than 0.05 at 95% of confidence interval. Five patient�s samples were randomly selected for percision analysis of both manual and automated methods. Each sample was measured five times by both methods to obtain the SD and CV% as shown in Table 2, the result indicated higher coefficient of variation (CV) in manual method than automated HCT results, which implicated the percision is good for automated method (mindray 3000 plus) and not good for manual method.
![]()
Table 1: Mean � SD of hematocrit result by automated and manual methods.
View Table 1
![]()
Table 2: The percision of manual and automated methods for hematocrit determination.
View Table 2
Discussion
This study indicated the manual HCT is higher than automated HCT. It showed the hematocrit values determined by the autohematological analyzer (mindray 3000 plus) can not replace the manual (microhematocrit) results obtained, as the mean difference between both methods is significant (P< 0.002), as also observed in other studies. A study undertaken in Nigeria, using Sysmex KX-21N, revealed statistically sisgnificant difference (P< 0.0001) when the mean and SE values of the two methods (automation and manual) were compared. In another study conducted on canine and feline, there was significant differences between manual and automated HCT (P< 0.05). The results of these studies also indicated that the HCT values from the automated method could not be used to substitute for those of the manual method, though the values of the two methods were accurate and precise [9,10]. Unlike to this study which reported higher value of HCT in manual method, another study reported a higher PCV value from Coulter automated analyzer, eventhough there was no significant association observed [11].
However, the data obtained from this study indicated a strong positive correlation between the two methods (R=0.95). In the study undertaken on dogs and cats, strong correlation using Pearsons correlation coefficient, R, between the two methods were observed (R for HCT of dog=0.96, and cat=0.98) [10] . Another study also reported a comparable positive correlation coefficient (r=0.948) when both methods (manual and automated) were compared [9]. In addition, another study reported the results obtained with Hematology analyzer (Mindray BC: 5500) for PCV were correlated with manual method (R=0.8651) [12].
The automated method is more precise than the manual method due to the higher variation of individual measured samples obtained by the manual method. The imprecision in measurment of PCV by the manual method may result in variations in calculation of RBC indices, such as the MCV and MCHC which are important parameters in classification of anemias.
Conclusion
Generally, the study showed the hematocrit value obtained from hematology analyzer (mindray 3000 plus) is different from that of manual, but it is directly proportional in most cases. The automated method can not replace the manual for hematocrit determination though the result of both methods are close to each other.
Recommendation
This study should be done in large scale. Further study should be done for the evaluation of the two methods including different variables like wbcs, rbcs and platelets. In this case, a specified hematocirt value (1.5%) must be added to the automated result when doing hematocrit if necessary.
Acknowledgement
We acknowledge the Yirgalem hospital laboratory for permitting materials to do the work and to undertake the study in the laboratory.
Author�s contribution
GG and KT: collected the data, data analysis and write up. HA and MB: produced the research idea, supervised data collection, final write up of manuscript.
Conflict of Interest
The authors declared they have no conflict of interest, and all authors have agreed on the submission of the manuscript.
References
-
Kathleen K (2007) The Clinical Laboratory Improvement Act (CLIA) and the Physician�s Office Laboratory. Continuing Medical Education.
-
CLSI (2000) Procedure for Determining Packed Cell Volume by the Microhematocrit Method; Approved Standard (3rd edn) CLSI document H7-A3 [ISBN 1-56238-413-9]. CLSI, 940 West Valley Road, Suite 1400, Wayne, Pennsylvania 19087-1898, USA.
-
Brian SB, Karen LH (2001) Is the Packed Cell Volume (PCV) Reliable? Lab Hematol 7: 191-196
-
Bull BS, Fujimoto K, Houwen B, Klee G, van Hove L, et al. (2003) International Council for Standardization in Haematology (ICSH) Recommendations for �Surrogate Reference� Method for the Packed Cell Volume. Lab Hematol 9: 1-9.
-
Birhaneselassie M, Birhanu A, Gebremedhin A, Tsegaye A (2013) How useful are complete blood count and reticulocyte reports to clinicians in Addis Ababa hospitals, Ethiopia? BMC Hematol 13: 11.
-
Pearson TC, Guthrie DL (1982) Trapped plasma in the microhematocrit. Am J Clin Pathol 78: 770-772.
-
Gotch F, Torres L, Evans M, Keen M, Metzner K, et al. (1991) Comparison of conductivity measured hematocrit to microhematocrit. ASAIO Trans 37: M138-139.
-
Salem M, Chernow B, Burke R, Stacey JA, Slogoff M, et al. (1991) Bedside diagnostic blood testing. Its accuracy, rapidity, and utility in blood conservation. JAMA 266: 382-389.
-
Ike SO, Nubila T, Ukaejiofo EO, Nubila IN, Shu EN, et al. (2010) Comparison of haematological parameters determined by the Sysmex KX - 2IN automated haematology analyzer and the manual counts. BMC Clin Pathol 10: 3.
-
Prihirunkit K, Lekcharoensuk C, Pisetpaisan K (2008) Comparison between Manual and Automated Methods for Determination of Canine and Feline Hematocrit and Hemoglobin Concentration Kasetsart J. Nat. Sci. 42: 655-659.
-
Kakel SJ (2013) The evaluation of traditional and automatic Coulter method in estimation of haematological parameters in adult rats. Beni-Suef University Journal of Basic and Applied Sciences 2: 31- 35.
-
Threeswaran R, Sooriyakumar T, Arasaratnam V (2012) Comparison of the Automated Full Blood Count Results with Manual Method. Proceedings of the Abstracts of Jaffna University International Research Conference (JUICE- 2012).





