International Journal of Allergy Medications
Continuous Albuterol Treatments may Lead to Lactic Acidosis in Children with Status Asthmaticus
Sheel Patel1, Usama Hanhan2*, Caroline Perkowski1 and James Orlowski2
1Lake Erie College of Osteopathic Medicine, Florida, USA
2Department of Pediatrics, Florida Hospital-Tampa, USA
*Corresponding author: Usama Hanhan, Department of Pediatrics, Florida Hospital Tampa, 3100 E Fletcher Ave, Tampa, FL-33613, USA, E-mail: usama.hanhan@ahss.org
Int J Aller Medcations, IJAM-3-023, (Volume 3, Issue 1), Case Report; ISSN: 2572-3308
Received: October 17, 2016 | Accepted: March 18, 2017 | Published: March 21, 2017
Citation: Patel S, Hanhan U, Perkowski C, Orlowski J (2017) Continuous Albuterol Treatments may Lead to Lactic Acidosis in Children with Status Asthmaticus. Int J Aller Medications 3:023.
Copyright: © 2017 Patel S, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Asthma is a chronic inflammatory condition of the airways characterized by airflow obstruction, caused by bronchoconstriction, airway edema, acute inflammation and increased secretions, most commonly treated with albuterol. Albuterol functions by activating beta-2-adrenergic receptors on airway smooth muscles stimulating adenyl cyclase and increasing production of C-AMP causing relaxation of the smooth muscle and bronchodilation. It has been our observation that lactic acid levels tend to increase with the use of continuous albuterol treatments in pediatric patients. This phenomenon is important to recognize as a possible complication of albuterol treatment in patients with status asthmaticus. Not recognizing this association may lead to an escalation of therapy, by misinterpreting the condition as worsening respiratory distress, hypoxemia or volume depletion. Progression of therapy may ultimately lead to prolonged hospitalizations, mechanical ventilation and further increase in morbidity and mortality. In this case report, we discuss three pediatric patients in status asthmaticus, admitted to Florida Hospital Tampa-PICU and placed on continuous albuterol treatments, who in-spite of improvement of airflow exchange, bronchospasm and general condition and in the absence of clinical hypoxemia or hypovolemic shock, went on to develop metabolic acidosis with rising lactic acid levels. We graphed their lactic acid levels throughout their hospital course as a function of continuous albuterol treatments. We observed that weaning the patients off continuous albuterol treatments resulted in quick resolution of acidosis and continued clinical improvement of the patients, resulting in a timely discharge.
Keywords
Albuterol, Lactic acidosis, Children, Asthma
Introduction
Asthma is a chronic inflammatory condition of the airways. The number of children suffering an asthma exacerbation in 2010 was 4.3 million [1]. Albuterol, a beta-2-adrenergic agonist has become the mainstay of bronchodilator therapy in the treatment of asthma and status asthmaticus [1-4]. It has been observed that lactic acid levels increase with the continuous albuterol use in pediatric patients [5,6]. In this case report, we discuss three pediatric patients in status asthmaticus, admitted to FHT and placed on continuous albuterol treatments, who in-spite of improvement of airflow exchange, bronchospasms and general condition and in the absence of clinical hypoxemia or hypovolemic shock went on to develop metabolic acidosis with rising lactic acid levels. Subsequent weaning from continuous albuterol resulted in quick resolution of acidosis [2,7,8].
Case 1
A 17-year-old asthmatic female was admitted to the PICU, with the symptoms of wheezing, tachypnoea and shortness of breath. Her initial saturations were 89% on RA, which improved to 95-99% with oxygen and continuous albuterol treatments. Initial ABG showed pH of 7.33, pCO2 43.4 mmHg, pO2 110 mmHg, bicarbonate 23.1 mEq/L and base excess (BE) of -3 at FiO2 of 0.32. Anion gap was recorded at 11 mEq/L. Patient was treated with IV steroids, continuous albuterol treatments, IVFs and other standard supportive care.
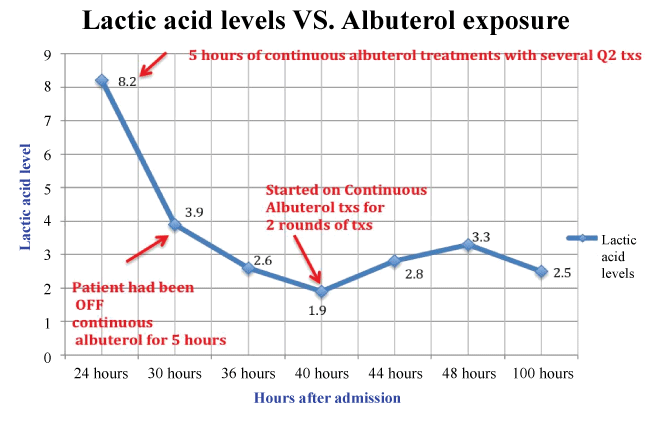
5-hours of continuous albuterol showed pH of 7.29, pCO2 32, pO2 165, HCO3 15.5 and BE of -10.1 (FiO2 of 0.32). The lactic acid level, then, was 8.2 mmol/L (Normal: 0.5-2.2 mmol/L). This was observed in-spite of improved general condition, good hydration and oxygenation with improvement of air exchange in both lungs. Continuous albuterol treatments were then weaned to Q2-4 hour treatments. A follow-up ABG showed pH 7.32, pCO2 33, HCO3 17.1 and BE of -8.1 (FiO2 of 0.21). About 16-hours later ABG normalized and lactic acid levels trended down to 1.9 (Figure 1). The following day, patient's condition declined, requiring her to be placed back on continuous albuterol nebulizer treatments for 8-hours. At that time, lactic acid levels rose to a final value of 3.3, and then declined to 2.5 after weaning her off continuous albuterol treatments. Patient continued to improve and was discharged home after 4 days.
Case 2
A 10-year-old known asthmatic male was admitted from the ED with 1-day history of shortness of breath, wheezing, and cough. He required continuous oxygen supplementation, allowing his oxygen saturation to increase from low 80s to low 90s. Patient's condition warranted immediate intubation. Initial ABG showed respiratory acidosis with pH 7.26, pCO2 53.8, pO2 321.0, BE -3.2 and HCO3 24.0 with FiO2 of 1.0. He continued to be on ventilator support, sedated with versed drip, treated with IV methylprednisolone 1 mg/kg Q6H, continuous albuterol treatments at 0.3 mg/kg/hr, and IVFs.
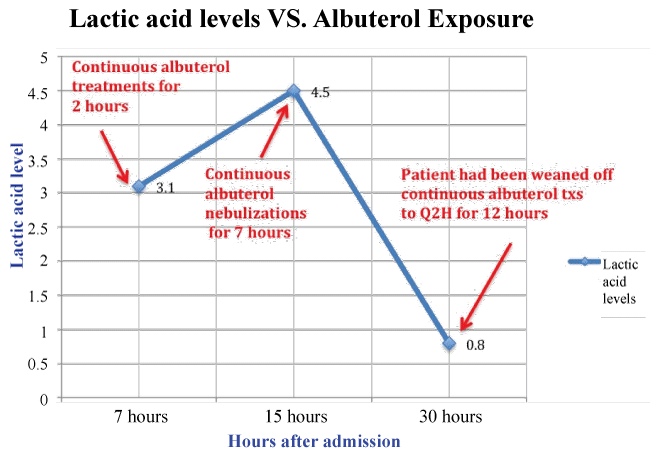
ABG obtained after 7-hours of continuous albuterol treatments showed paradoxical metabolic acidosis in-spite of clinical improvement with pH 7.23, pCO2 41, pO2 79.8, HCO3 17.3 and BE -9.5 (FiO2 of 0.30). Lactic acid level, then, was elevated at 4.5. Patient's oxygen saturation at that time continued to be in high 90s. In addition, patient had no signs to suggest significant changes in his perfusion since initial intubation. Thus, it is less likely that his elevated lactic acid level is due to wash out from improved perfusion. Albuterol treatments were then spaced out to Q2H. Serial lactic acid level measurement decreased to 0.8 (Figure 2). Patient continued to improve and was discharged home on day 5.
Case 3
13-year-old known asthmatic male was admitted from ED after he presented with dyspnoea, cough, shortness of breath and wheezing, in-spite of several albuterol treatments and oral steroids at home. Upon presentation, patient was tachypneic at 28 br/min., with diminished breath sounds on right and some audible wheezing bilaterally. His condition required him to be placed on continuous albuterol treatment and subsequent admission to PICU. In the PICU, patient continued to be on continuous albuterol nebulizations, IV methylprednisolone, and oxygen via high flow NC. Initial ABG done 2-hours after admission, showed pH of 7.42, pCO2 33.6, pO2 73.1 and HCO3 21.9 with a base excess -2.1 at FiO2 of 0.90.
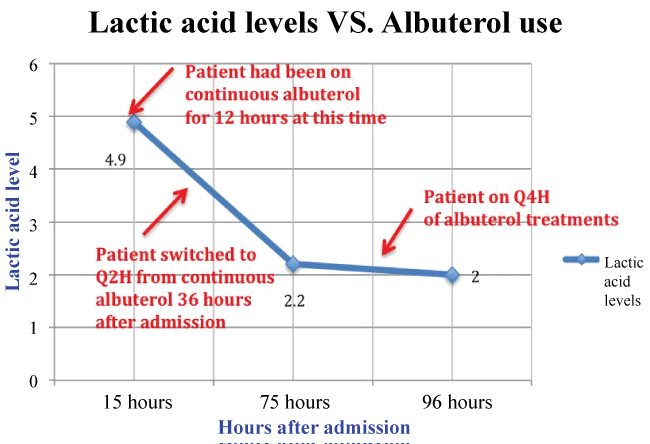
Re-evaluation, later in the day, showed patient to be breathing much easier while still being on continuous albuterol treatments and high flow nasal cannula with adequate oxygenation and perfusion. VBG done after 12-hours of continuous albuterol treatments revealed compensated metabolic acidosis with pH 7.34, pCO2 29.5, pO2 63 and a low HCO3 16 with FiO2 of 0.70. Lactic acid level, at that time was elevated at 4.9 (Figure 3). 36-hours after admission, patient's respiratory status improved with better air exchange and he was weaned to Q2-4 hour treatments. Repeat lactic acid was 2.2. Blood gas normalized with pH 7.43, pCO2 38.3 and HCO3 25. A follow-up lactic acid level before discharge was 2.0.
Discussion
Early in status asthmaticus, respiratory alkalosis from hyperventilation frequently occurs. When the attack persists, CO2 is retained leading to respiratory acidosis. It has been our observation that some patients who receive frequent or continuous albuterol nebulizations will develop a paradoxical metabolic acidosis in the absence of hypovolemia, sepsis or hypoxia [2,9,10]. Figure 1, Figure 2 and Figure 3 show lactic acid levels in relation to continuous albuterol treatments over the course of admission. All our patients were clinically well hydrated while maintaining saturations over 95% throughout.
There are two types of lactic acidosis: Type A and Type B [11,12]. Type A lactic acidosis results from impaired oxygen delivery or consumption, whereas type B lactic acidosis results from altered cellular metabolism of lactic acid in absence of hypoxia or hypoperfusion. B2 agonists have been hypothesized to cause type B lactic acidosis [6,12,13]. Type A and Type B lactic acidosis can be differentiated from each other using the ratio of lactate to pyruvate levels [8,11].
Some postulations have suggested that adrenergic stimulation-exogenous or endogenous, may be associated with increased formation of lactic acid in the body. The suggested mechanism is an increase in metabolic effects including fatty acids and plasma glucose concentrations, glycogenolysis, gluconeogenesis and lipolysis due to stimulation of B-adrenergic receptors [14]. This increase leads to stimulation of glycolysis, which ultimately increases pyruvate. Alongside, a concurrent inhibition of the enzyme pyruvate dehydrogenase, due to hypoxia related to asthma exacerbation and increase in free fatty acids as a result of lipolysis, prevents pyruvate from entering the Kreb's cycle and thus pushing it towards the anaerobic cycle-increasing lactic acid levels [2,4,9,15,16].
Figure 1 represents Case 1. First data point is at 24-hours after admission, when patient had been on 5-hours of continuous albuterol treatments. Here, lactic acid level was 8.2 mmol/L. Weaning to Q2-4 hour treatments for 5-hours regressed the levels to 3.9 and 2.6 at 30 and 36-hours of admission respectively. 42-hours post admission, the levels normalized to 1.9. Placing the patient back on albuterol for other 8-hours raised the lactic acid levels to 2.8 and 3.3, at 4- and 8-hours of treatment respectively. Subsequent weaning to Q2-4 hr lowered the levels to 2.5.
Figure 2 represents Case 2, where the patient in status asthmaticus required intubation. Lactic acid level after 2-hours of continuous albuterol nebulizations was 3.1. Due to his acute condition, patient was placed on 7-hour continuous albuterol. Lactic acid level then peaked to 4.5, represented by the data-point at 15-hours. Weaning down to Q2 hours, allowed his lactic acid levels to drop to 0.8, 27-hours after admission.
Finally, Figure 3 represents Case 3, where patient was placed on 12-hours of continuous albuterol, raising the levels to 4.9, data-point represented at 15-hours. 36-hours after admission, the patient was weaned to Q2-4 hr treatments due to clinical improvement. Consequent lactic acid level measurement was 2.2, about 75-hours after admission. His final lactic acid level before discharge was 2.0. This again represented a drop in lactic acid levels as a result of decrease in frequency of albuterol treatments.
In all cases above, the only change was spacing out the continuous albuterol treatments to a less frequent therapy. Continuous albuterol nebulizations in all the three cases raised the lactic acid levels, whereas weaning the patients led to a gradual decline in the levels. No other treatment changes were made. These findings and cases add to the evidence of possible causal relationship between the administration of continuous albuterol and a rise in lactic acid levels. The next step in establishing a more concrete relationship between lactic acidosis and continuous albuterol treatments is to confirm that albuterol exposure causes Type B Lactic acidosis. For this, we need pyruvate levels. We were unable to draw the levels, in this case report, due to the lack of this laboratory feature at our hospital.
Recognizing this relationship between continuous albuterol nebulization and development of lactic acidosis, with paradoxical metabolic acidosis despite clinical improvement of bronchospasms, is extremely important. If unaware of such a phenomena one can easily confuse metabolic acidosis due to lactic acid accumulation secondary to continuous albuterol treatments, for clinical deterioration and thus inadvertently increasing therapy. Disregarding or misunderstanding this relationship can, therefore, lead to escalation of therapy and other invasive interventions with an intention to better the patient. This is likely to affect the morbidity and mortality of patients, hence discouraging prolonged or escalated treatments in patients with status asthmaticus. We would hope that sharing this information with our colleagues would help them recognize this condition when an ABG read does not correlate with other clinical signs. We suggest de-escalation of therapy in such patients. Weaning the patient off continuous albuterol therapy to intermittent treatments may help resolve the metabolic derangement. It goes without saying that it is important to ensure that the patient is maintaining proper oxygen saturations and is hemodynamically stable before presuming type B lactic acidosis, because withdrawing necessary treatment prematurely can harm the patients.
Acknowledgement
Associates of the library at the Florida Hospital Tampa for helping with the research.
Ethical Statements
This article is based on case reviews that involved three pediatric patients who were admitted to the hospital with asthma exacerbation and underwent treatment with continuous alcohol. No patient information or patient identifiers have been used in the case report.
Contributors Statements
Sheel Patel: Dr. Patel collected the data and hospital records for the patients outlined in the case report, drafted the initial manuscript, and approved the final manuscript as submitted.
Usama Hanhan: Dr. Hanhan provided with the initial and related cases for the case report, revised and reviewed the manuscript, and approved the final manuscript as submitted.
Caroline Perkowski: Dr. Perkowski graphed the data from patient records, reviewed manuscript, and approved the final manuscript as submitted.
James Orlowski: Dr. Orlowski critically reviewed and revised the manuscript several times, and approved the final manuscript as submitted.
References
-
Cohn L, Elias JA, Chupp GL (2004) Asthma Mechanisms of Disease Persistence and Progression. Annual Review of Immunology 22: 789-815.
-
Kovacevic A, Schwahn B, Schuster A (2010) Hyperlactic Acidosis as Metabolic Side-Effect of Albuterol and Theophylline in Acute Severe Asthma. Klin Pädiatr 222: 271-272.
-
Lau E, Mazer J, Carino G (2013) Inhaled β-agonist therapy and respiratory muscle fatigue as under-recognised causes of lactic acidosis. BMJ Case Rep.
-
Manthous CA (2001) Lactic acidosis in Status Asthmaticus: Three Cases and Review of the Literature. Chest 119: 1599-1602.
-
Maury E, Ioos V, Lepecq B, Guidet B, Offenstadt G (1997) A Paradoxical Effect of Bronchodilators. Chest 111: 1766-1767.
-
Walsh SA, Paget RI, Ramnarayan P (2013) Salbutamol Usage and Lactic Acidosis in Acute Severe Asthma. Pediatr Crit Care Med 14: 116-117.
-
Rodrigo GJ, Rodrigo C (2005) Elevated plasma lactate level associated with high dose inhaled albuterol therapy in acute severe asthma. Emerg Med J 22: 404-408.
-
Rodrigo GJ (2014) Serum Lactate Increase During Acute Asthma Treatment: A new piece of the puzzle. Chest 145: 6-7.
-
Claret PG, Bobbia X, Boutin C, Rougier M, de la Coussaye JE (2012) Lactic acidosis as a complication of β-adrenergic aerosols. Am J Emerg Med 30: 1319.e5-1319.e6.
-
Creagh-Brown BC, Ball J (2008) An under-recognized complication of treatment of acute severe asthma. Am J Emerg Med 26: 514.e1-514.e3.
-
Dodda VR, Spiro P (2012) Can Albuterol be blamed for Lactic Acidosis? Respir Care 57: 2115-2118.
-
Prakash S, Mehta S (2002) Lactic Acidosis in asthma: Report of two cases and review of the literature. Can Respir J 9: 203-208.
-
Koul PB, Minarik M, Totapally BR (2007) Lactic acidosis in children with acute exacerbation of severe asthma . Eur J Emerg Med 14: 56-58.
-
Liem EB, Mnookin SC, Mahla ME (2003) Albuterol-induced Lactic Acidosis. Anesthesiology 99: 505-506.
-
Berman SB, Liao JS (2012) Albuterol-Induced Lactic Acidosis: A Case Report. Proceedings of UCLA Healthcare 16.
-
Meert KL, McCaulley L, Sarnaik AP (2012) Mechanism of lactic acidosis in children with acute severe asthma. Pediatr Crit Care Med 13: 28-31.