International Journal of Anesthetics and Anesthesiology
Platelet Count and INR Profile after Hepatic Resection in Cirrhotic Patients: Implications for Epidural Analgesia
Siniscalchi A*, Gamberini L, Carini A, Laici C, Tassinari S and Faenza S
Division of Anesthesiology, Department of Medical and Surgical Sciences - DIMEC, Alma Mater Studiorum - University of Bologna, S.Orsola Hospital, Italy
*Corresponding author: Antonio Siniscalchi, MD, Division of Anesthesiology, Department of Medical and Surgical Sciences - DIMEC, Alma Mater Studiorum University of Bologna, Via Massarenti 9, 40138 Bologna, Italy, Tel: +39 0516363440, E-mail: sinianest@libero.it
Int J Anesthetic Anesthesiol, IJAA-1-009, (Volume 1, Issue 2), Original Article; ISSN: 2377-4630
Received: September 08, 2014 | Accepted: Ocober 13, 2014 | Published: Ocober 15, 2014
Citation: Siniscalchi A, Gamberini L, Carini A, Laici C, Tassinari S, et al. (2014) Platelet Count and INR Profile after Hepatic Resection in Cirrhotic Patients: Implications for Epidural Analgesia. Int J Anesthetic Anesthesiol 1:009. 10.23937/2377-4630/1009
Copyright: © 2014 Siniscalchi A, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Introduction: Since cirrhotic patients undergoing hepatic resection are at increased risk of developing postoperative coagulopathy, epidural catheter placement in this group of patients is still debated. This retrospective study aimed to evaluate postoperative course of coagulation parameters after surgical hepatic resection in cirrhotic patients and their relation to extent of resection and perioperative risk factors.
Methods: Perioperative data from 232 hepatic resections performed in cirrhotic patients were reviewed. We defined postoperative coagulopathy the occurrence of a postoperative platelet count < 100,000/ml and/or INR value = 1.5. Logistic regression was used to assess the association between postoperative coagulopathy and several potential risk factors, while general linear model for repeated measures was used to compare postoperative course of coagulation parameters.
Results: 98 patients (42.24%) showed an abnormal coagulation profile at least once during the first 7 postoperative days. None of the analyzed parameters resulted statistically associated with the development of postoperative coagulopathy. Postoperative INR course was significantly different in patients undergoing minor resections with better values, while platelet count was not.
Conclusions: Postoperative coagulopathy after hepatic resections is a common issue in cirrhotic patients, which may limit the feasibility of invasive procedures such as epidural catheter placement. Multivariate analysis didn't show any association between investigated risk factors and postoperative coagulopathy. Postoperative course of coagulation parameters in cirrhotic patients with normal preoperative coagulation tests undergoing minor liver resection seems to be compatible with epidural catheter placement and management.
Keywords
Liver resection, Liver cirrhosis, Epidural analgesia
Abbreviations
HCC: Hepatocellular Carcinoma; BMI: Body Mass Index; CTP score: Child Turcotte Pugh score; POD: Postoperative Day; IHPBA - International Hepato Pancreato Biliary Association; INR: International normalized ratio; PLT: Platelet Count; MELD: Model of End-Stage Liver Disease; MAC: Minimum Alveolar Concentration; TEG: Thromboelastography; FFP: Fresh Frozen Plasma
Introduction
Hepatocellular Carcinoma (HCC) represents one of the most common tumors worldwide and, in the majority of the cases, occurs in patients with chronic underlying liver disease mainly caused by hepatitis B and C viruses [1]. In cirrhotic patients, resection of HCC requires expert candidate selection, adequate skills in carrying out the surgical procedure, and optimal postoperative management [2].
In addition, liver resection poses many challenges for the anaesthesiologist, including intraoperative haemodynamic instability and coagulopathy even when liver function tests are normal preoperatively [3]. The use of epidural analgesia in this group of patients potentially confers several benefits, including improved intraoperative haemodynamic stability and improved post-operative pain control [4]. However, these advantages have to be balanced against the risks of epidural catheter placement and removal if patients develop postoperative coagulopathy on the basis of liver dysfunction [5,6].
Although most recent studies investigated the changes in postoperative coagulation profile after curative hepatic surgery in mainly non cirrhotic patients [3,7,8], while others examined only healthy donors for donor hepatectomies [9-11], there are currently no literature studies focused exclusively on cirrhotic patients. In most of these previous studies, HCC in liver cirrhosis was not a common surgical diagnosis for hepatic resection; moreover, information about co-existing liver disease was not always given.
The aims of present study were the following:
1. To examine the changes of coagulation parameters after surgical hepatic resection in cirrhotic patients and, in particular, the relationship between postresection coagulation and the extent of hepatectomy.
2. To investigate the relationship between postresection coagulation parameters and intraoperative fluid infusions, Pringle maneuver, length of surgical procedure, BMI, MELD, CTP score and portal hypertension in order to determine possible risk factors for the development of a postoperative coagulopathy.
Materials and Methods
After Hospital Ethics Committee approval (approval number: 282/2013/O/OssN), this retrospective observational study included all cirrhotic patients who underwent laparotomic liver resection between January 2007 and November 2012.
Inclusion criteria were: age > 18 years, histologic-proven liver cirrhosis, open liver resection surgery for HCC. Exclusion criteria were: abnormal preoperative coagulation profile, laparoscopic liver resection.
We defined abnormal coagulation profile the presence of INR values = 1.5 and/or platelet count < 100,000/ml in the first 7 Postoperative Days (POD) [12].
We chose to exclude patients with abnormal preoperative coagulation profile because of the relative contraindication to epidural catheter placement in this sub-group even if patients included in the study did not receive epidural anesthesia.
Major hepatic resection was defined as a resection of three or more hepatic segments, while minor hepatic resection as a resection of two or less hepatic segments with reference to the IHPBA classification; all resections were performed to achieve a tumor-free margin of at least 1 cm based on intraoperative examination and ultrasonography.
Anesthetic management for liver resection at our unit includes: general endotracheal anesthesia, arterial line and central venous catheter placement for fluid infusions, hemodynamic monitoring, and acid-base parameter measurement.
Fluid infusions are kept minimal during hepatic dissection to keep low central venous pressure (= 5 mmHg) in order to reduce bleeding, while red blood cells are transfused when hematocrit is lower than 27% and/or hemoglobin is lower than 8 g/dL. Patient's hypothermia is prevented by warm fluid infusion, forced-air warming and the use of warm water on the surgical field.
Perioperative coagulation alterations are corrected with fresh frozen plasma infusion if INR is > 1.5 and there are clinical signs of active bleeding [13].
Data collected preoperatively included patient characteristics, underlying surgical pathology, aetiology of cirrhosis, Child-Pugh Classification of hepatic disease as well as MELD score, baseline coagulation profile and blood tests.
The analyzed intraoperative data included the type of hepatic resection, duration of sugery, the duration of vascular occlusion techniques (continuous or intermittent Pringle maneuver) and fluid infusions and transfusions of blood products.
The collected postoperative blood tests were platelet count and INR values from POD 1 to POD 7.
Statistical Analysis
Categorical data were expressed as numbers (percentages), continuous variables as mean and standard deviation.
Differences in preoperative data between groups undergoing major and minor liver resection were evaluated with t-test for continuous variables and Chi square test or Fisher exact test for nominal variables.
In order to evaluate risk factors for the development of a postoperative abnormal coagulation profile, univariate and multivariable logistic regression analysis was performed separately for the two conditions INR value = 1.5 and platelet count < 100,000/ml.
The following parameters were considered for analysis:
BMI, portal hypertension, diabetes, CTP and MELD scores, duration of surgery, duration of vascular occlusion technique (Pringle maneuver), transfusion of blood products and fresh frozen plasma and infusion of crystalloids and colloids.
Multivariable logistic regression analysis was conducted on variables that reached p < 0.2 at univariate analysis. Multicollinearity amongst variables was detected by means of Spearman correlation test.
Models building follow a backward-stepwise approach; the test of term significance is the Wald chi-square test with cutoff p value of 0.1 for removal and 0.05 for addition.
Model discrimination was assessed calculating the Area under the Receiver Operator Characteristic (ROC) curve, whereas model calibration has been determined by Hosmer-Lemeshow (H-L) technique.
All p values refer to two-tailed test of significance; p< 0.05 was considered significant.
A General Linear Model for repeated measures was used to compare INR and platelets values during the first postoperative week in patients who underwent minor vs. major hepatic resections.
Statistical analyses were conducted using Stata/SE 12.1 (College Station, TX: StataCorp LP)
Results
A total of 347 cirrhotic patients underwent hepatic resection for HCC at the Department of Surgery and Transplantation of Bologna University between January 2007 and November 2012; 24 of these were not included in statistical analysis for the following reasons: incomplete clinical data (21 patients), early postoperative death due to acute myocardial infarction (2 patients) and pulmonary embolism (1 patient).
Finally, we excluded from the study population 91 patients who showed signs of preoperative coagulopathy. The final study group consisted of 232 patients with HCC and histologic-proven cirrhosis.
Patient characteristics and preoperative data are shown in Table 1. Most patients (97.8 %) were classified as Child-Pugh class A and cirrhosis aetiology was viral in 84.9 %; preoperative INR value and MELD score were significantly higher and platelet count significantly lower in minor resection group.
![]()
Table 1: Patient characteristics and preoperative data.
View Table 1
Table 2 shows intraoperative data: surgery consisted of minor hepatic resection in most cases, patients in major resection group received significantly more intraoperative fresh frozen plasma infusions.
![]()
Table 2: Type of hepatic resection and intraoperative data.Type of hepatic resection and intraoperative data.
View Table 2
In minor resection group, 41% of patients developed a postoperative coagulopaty, while in the major hepatic resection group this event was observed in 52.3 % of cases; however, this difference was not statistically significant.
Considering both minor and major resections, 98 patients (42.24%) showed an abnormal coagulation profile at least once in the first 7 postoperative days, a decrease in platelet count was the most common finding, while at the end of study period, on POD 7, patients had abnormal INR value and platelet count respectively in 5% and 4.3% of cases (data not shown in figures).
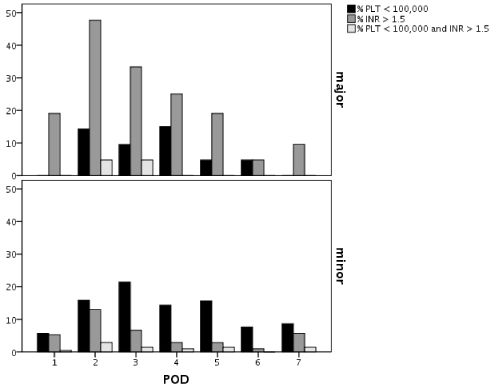
Figure 1 shows the proportion of patients who developed postoperative coagulopathy divided for type of resection and etiology of coagulopathy (INR>1.5, PLT < 100,000/ml or both).
A significant proportion of patients in major liver resection group developed a high INR coagulopathy (47% on POD 2), which gradually decreased up to POD 6, where less than 5% of patients still had a high INR value.
In minor liver resections, low platelet count was the most common cause of coagulopathy, with its maximum incidence on POD 3. 5 patients out of 232 (2.2%) needed postoperative fresh frozen plasma to correct coagulopathy because of significant bleeding; all of these underwent major liver resection, and there was no need for postoperative platelet transfusions in both groups.
Figures 2 and figure 3 show mean INR values and platelet counts observed during first 7 PODs according to the extent of hepatic resection; in both groups, highest INR levels were reached on POD 2.
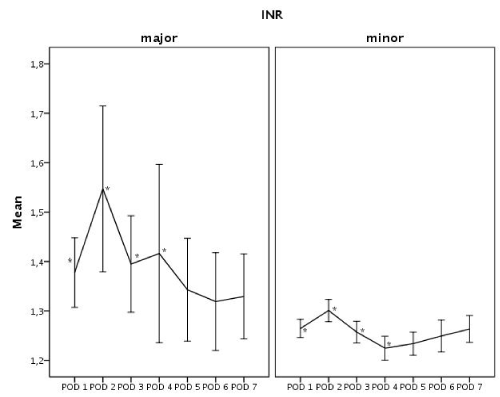
Figure 2: Mean INR values in PODs 1 to 7
POD: Post-Operative Day; INR: International Normalized Ratio. * shows significant (p< 0.05) differences found with General Linear Model for repeated measures, overall difference was significant (p< 0.01). Bars show CI 95%.
View Figure 2
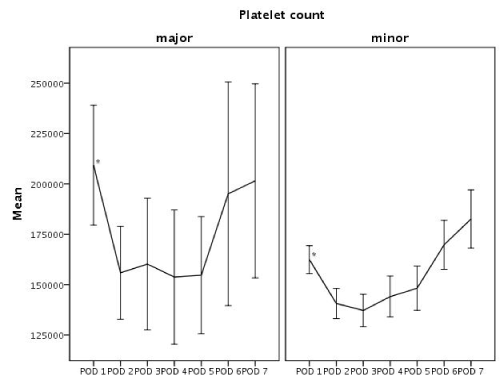
Figure 3: Mean platelet count values in PODs 1 to 7
POD: Post-Operative Day * shows significant (p< 0.05) differences found with General Linear Model for repeated measures, overall difference was not significant (p< 0.055). Bars show CI 95%.
View Figure 3
General linear model analysis for repeated measures resulted in a significant overall difference in INR values between groups (p < 0.01), moreover patients undergoing minor resection presented significantly lower INR values during the first four postoperative days.
Platelet count reached its nadir on POD 2 in major resection group and on POD 3 in minor resection group. General linear model analysis didn't report a significant overall difference (p = 0.055) and platelet count was significantly lower in minor resection group only during the first postoperative day.
Table 3 shows uni- and multivariate logistic regression analysis considering platelet count < 100,000/ml the dependent variable. No statistically significant association was found between any of the parameters and the development of a postoperative low platelet count.
![]()
Table 3: Logistic regression analysis: PLT < 100000 ml-1 as dependent variable.
View Table 3
As shown in Table 4, multivariable analysis didn't highlight any statistically significant association between postoperative INR value = 1.5 and any of the studied parameters.
![]()
Table 4: Logistic regression analysis: INR = 1,5 as dependent variable.
View Table 4
Discussion
Our study included only histologic-proven cirrhotic patients suitable for epidural catheter placement on the basis of preoperative coagulation profile and showed that significant alterations in INR and platelet values were observed in 98 out of 232 cirrhotic patients who underwent uncomplicated liver resection for HCC (42%).
In minor resection group, INR value >1.5 and platelet count < 100,000/ml were present respectively in less than 3% and 6% of patients on postoperative day 6 (Figure 1); moreover this group had an INR profile that seems compatible with epidural catheters management (Figure 2); low platelet count (Figure 3) was the most common problem, and the significant difference in platelet count between the two groups found on POD 1 could be explained by a significantly lower preoperative count in patients undergoing minor resection (Table 1).
Previous studies have shown that underlying hepatic disease [6,8] and the extent of hepatic resection [7,8,14] contribute to the development of coagulopathy; in other studies, risk factors such as low BMI [14], duration of vascular occlusion used to limit blood loss during parenchymal dissection [8], length of surgical procedure [3,14], the volumes of fluids administered [5,10,15,16] and intraoperative blood loss [8] were associated to coagulopathy.
In contrast with other studies, we focused only on patients with chronic liver disease undergoing hepatic resections and we found that these risk factors were not significantly associated with an abnormal postoperative coagulation profile in this category.
Limitations of this study are related to its retrospective nature and the unavoidable selection bias between patients undergoing major resection and minor one. In fact major resection requires a better baseline hepatic function than minor resection because of the larger amount of parenchyma removed. However, despite significantly better preoperative MELD score and platelet count, patients undergoing major resection had worse postoperative coagulation parameters.
Another limitation of the study is the difference in sample size between major and minor resection groups. However we have to state that major resection surgery in cirrhotic patients isn't a frequent intervention at our unit and requires meticulous candidate selection due to the high risk of postoperative liver failure.
At our unit, epidural catheter placement for liver resection has been implemented in the last year together with the development of fast track liver surgery protocols, blended anesthesia is proposed to both cirrhotic and non cirrhotic patients with preoperative normal coagulation profile undergoing minor laparotomic hepatic resections.
For blended anesthesia in liver resection, we usually put a T8-T9 epidural catheter and, after an initial bolus of L-bupivacaine 7.5-10 mg, we start a continuous infusion at 5 to 7 mL/h of L-bupivacaine 2.5 mg/mL, we also add epidural sufentanyl boluses of 5 to 10 mcg; sedation is maintained with Sevoflurane at a concentration of 0.5-0.7 MAC.
In 2013, 254 patients underwent hepatic resection at our unit, 66 of these had an epidural catheter placed, 70 out of 254 were cirrhotic undergoing HCC resection and 13 of these received a blended anesthesia. The results were the following: epidural catheters were usually removed between postoperative day 3 and 5, no complications related to epidural catheter were recorded and we never needed to correct coagulation parameters with fresh frozen plasma or platelets infusion in this case series. The most important causes of the non applicability of epidural anesthesia were: extension of resection, preoperative coagulopathy, laparoscopic liver resection and anesthesiologist's poor confidence with this technique.
Liver cirrhosis increases the risk of an abnormal postoperative coagulation profile after hepatectomies, [6,8] hence the anesthesiologist should exercise caution in this subgroup when considering epidural catheter placement, due to the risks associated with the removal of the catheter during the postoperative period.
Operative time, entity of blood losses, usage of intermittent Pringle maneuver instead of continuous technique, and managing patients in high-dependency unit setting could strongly influence postoperative coagulopathy incidence and therefore epidural catheter safety even for the same resected volume of parenchyma.
There is still significant variability between centers in terms of surgical and anesthesiological experience in this field, so that postoperative coagulopathy could have a much higher variability (longer operative times and pedicle clamping, bleeding, postoperative liver failure); we suggest that this procedure should be performed only by experienced operators in a safe surgical setting.
For those patients considered to be at high risk of postoperative coagulopathy, or in case of inexperienced teams (where the liver resection is not a surgical procedure that it is often performed), when epidural catheter placement is considered too hazardous, possible alternatives for the management of postoperative pain may be represented by continuous local anesthetic wound infiltration [17], intercostal nerve blocks [18], transversus abdominis plane block or the use of intrathecal morphine, [19,20] although these approaches are not without potential problems (e.g. the risk of late respiratory depression with intrathecal morphine). It would also be interesting to compare epidural approach to these latter even in well compensated cirrhotic patients in order to evaluate safety and efficacy: prospective studies investigating these aspects are needed.
It is important to remember that, in presence of cirrhosis, we observe a rebalanced global hemostasis that could increase the risk of thrombotic events [21] and that the reliability of standard tests of hemostasis such as INR value and platelet count has been questioned in favor of Thromboelastography (TEG) which may provide a better assessment of bleeding risk [22].
Thus, patients who, according to standard tests of hemostasis, appear to be at increased bleeding risk after invasive procedures like epidural analgesia, may actually be eligible for such procedures. However, more comprehensive clinical studies are required comparing the effectiveness and safety of perioperative TEG use versus standard tests of hemostasis.
Because of the paucity of data about epidural anesthesia safety in cirrhotic patients it is actually impossible to express indications in this field. However, we suggest that centers approaching epidural catheter management in cirrhotic patients should primarily review their data about postoperative coagulopathy in liver resected patients to assess the safety of this approach and also to adopt a case by case discussion with their surgical team about potential entity of resection, estimated residual liver function, expected technical difficulties and risk for intraoperative bleeding.
Conclusions
Cirrhotic patients undergoing hepatic resection are at high risk of postoperative coagulopathy: our study shows that this is a common event and may limit the feasibility of invasive procedures such as placement of an epidural catheter for postoperative pain management.
Risk factors for postoperative coagulopathy found in other studies on hepatic resections in non cirrhotic or mixed patients weren't confirmed in our study.
Postoperative course of coagulation parameters is significantly different between patients who underwent minor and major resection.
Patients undergoing minor resection present a favourable profile which seems to be compatible with epidural catheter placement and management. However, the safety of this approach needs to be confirmed in this particular population with larger studies.
Ethical Approval
This study was conducted after Institutional Review Board approval (approval number: 282/2013/O/OssN).
References
-
Bosch FX, Ribes J, Borras J (1999) Epidemiology of primary liver cancer. Semin Liver Dis 19: 271-285.
-
Belghiti J, Regimbeau JM, Durand F, Kianmanesh AR, Dondero F, et al. (2002) Resection of hepatocellular carcinoma: a European experience on 328 cases. Hepatogastroenterology 49: 41-46.
-
Weinberg L, Scurrah N, Gunning K, McNicol L (2006) Postoperative changes in prothrombin time following hepatic resection: implications for perioperative analgesia. Anaesth Intensive Care 34: 438-443.
-
Tzimas P, Prout J, Papadopoulos G, Mallett SV (2013) Epidural anaesthesia and analgesia for liver resection. Anaesthesia 68: 628-635.
-
Matot I, Scheinin O, Eid A, Jurim O (2002) Epidural anesthesia and analgesia in liver resection. Anesth Analg 95: 1179-1181, table of contents.
-
Takita K, Uchida Y, Hase T, Kamiyama T, Morimoto Y (2014) Co-existing liver disease increases the risk of postoperative thrombocytopenia in patients undergoing hepatic resection: implications for the risk of epidural hematoma associated with the removal of an epidural catheter. J Anesth 28: 554-558.
-
Stamenkovic DM, Jankovic ZB, Toogood GJ, Lodge JPA, Bellamy MC (2011) Epidural analgesia and liver resection: postoperative coagulation disorders and epidural catheter removal. Minerva Anestesiol 77: 671-679.
-
Yuan FS, Ng SY, Ho KY, Lee SY, Chung AY, et al. (2012) Abnormal coagulation profile after hepatic resection: the effect of chronic hepatic disease and implications for epidural analgesia. J Clin Anesth 24: 398-403.
-
Siniscalchi A, Begliomini B, De Pietri L, Braglia V, Gazzi M, et al. (2004) Increased prothrombin time and platelet counts in living donor right hepatectomy: implications for epidural anesthesia. Liver Transpl 10: 1144-1149.
-
Borromeo CJ, Stix MS, Lally A, Pomfret EA (2000) Epidural catheter and increased prothrombin time after right lobe hepatectomy for living donor transplantation. Anesth Analg 91: 1139-1141.
-
Schumann R, Zabala L, Angelis M, Bonney I, Tighiouart H, et al. (2004) Altered hematologic profiles following donor right hepatectomy and implications for perioperative analgesic management. Liver Transpl 10: 363-368.
-
Horlocker TT, Wedel DJ, Benzon H, Brown DL, Enneking FK, et al. (2003) Regional anesthesia in the anticoagulated patient: defining the risks (the second ASRA Consensus Conference on Neuraxial Anesthesia and Anticoagulation). Reg Anesth Pain Med 28: 172-197.
-
Liumbruno G, Bennardello F, Lattanzio A, Piccoli P, Rossetti G; Italian Society of Transfusion Medicine and Immunohaematology (SIMTI) Work Group (2009) Recommendations for the transfusion of plasma and platelets. Blood Transfus 7: 132-150.
-
Kim YK, Shin WJ, Song JG, Jun IG, Kim HY, et al. (2010) Factors associated with changes in coagulation profiles after living donor hepatectomy. Transplant Proc 42: 2430-2435.
-
Shontz R, Karuparthy V, Temple R, Brennan TJ (2009) Prevalence and risk factors predisposing to coagulopathy in patients receiving epidural analgesia for hepatic surgery. Reg Anesth Pain Med 34: 308-311.
-
Wheatley RG, Schug SA, Watson D (2001) Safety and efficacy of postoperative epidural analgesia. Br J Anaesth 87: 47-61.
-
Ventham NT, Hughes M, O'Neill S, Johns N, Brady RR, et al. (2013) Systematic review and meta-analysis of continuous local anaesthetic wound infiltration versus epidural analgesia for postoperative pain following abdominal surgery. Br J Surg 100: 1280-1289.
-
Finnerty O, Carney J, McDonnell JG (2010) Trunk blocks for abdominal surgery. Anaesthesia 65 Suppl 1: 76-83.
-
Kasivisvanathan R, Abbassi-Ghadi N, Prout J, Clevenger B, Fusai GK, et al. (2014) A prospective cohort study of intrathecal versus epidural analgesia for patients undergoing hepatic resection. HPB (Oxford) 16: 768-775.
-
De Pietri L, Siniscalchi A, Reggiani A, Masetti M, Begliomini B, et al. (2006) The use of intrathecal morphine for postoperative pain relief after liver resection: a comparison with epidural analgesia. Anesth Analg 102: 1157-1163.
-
Tripodi A, Primignani M, Lemma L, Chantarangkul V, Dell'Era A, et al. (2010) Detection of the imbalance of procoagulant versus anticoagulant factors in cirrhosis by a simple laboratory method. Hepatology 52: 249-255.
-
Stravitz RT (2012) Potential applications of thromboelastography in patients with acute and chronic liver disease. Gastroenterol Hepatol (N Y) 8: 513-520.