International Archives of Clinical Pharmacology
Mass Dose Effects of Carfentanil and Raclopride on Venous Plasma Cortisol and Prolactin after Tobacco Smoking during PET Scanning
Edward F Domino*, Lisong Ni and Mika Hirasawa-Fujita
Department of Pharmacology, University of Michigan, USA
*Corresponding author:
Edward F Domino, Department of Pharmacology, University of Michigan, 1301 MSRB III, Ann Arbor, MI 48109, USA, Tel: 734-971-7363, Fax: 734-763-6672, E-mail: efdabcde@umich.edu
Int Arch Clin Pharmacol, IACP-2-005, (Volume 2, Issue 1), Original Article
Received: March 22, 2016: Accepted: May 05, 2016: Published: May 09, 2016
Citation: Domino EF, Ni L, Hirasawa-Fujita M (2016) Mass Dose Effects of Carfentanil and Raclopride on Venous Plasma Cortisol and Prolactin after Tobacco Smoking during PET Scanning. Int Arch Clin Pharmacol 2:005.
Copyright: © 2016 Domino EF, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Mass dose effects of the PET radioligands [11C] carfentanil and [11C] raclopride were determined on venous plasma cortisol and prolactin levels before and after tobacco smoking in overnight abstinent healthy male smokers.
Methods:The total [11C] labeled and unlabeled doses of each ligand were used to assess the association between the doses of ligands and the hormone levels by linear regression analyses.
Results:Peak cortisol levels after average nicotine (avnic) tobacco smoking were decreased with greater carfentanil doses (p = 0.03). The change in plasma cortisol levels after avnic smoking compared to before avnic smoking was also decreased with greater carfentanil doses (p = 0.003). Unexpectedly the changes in cortisol levels after denicitinized (denic) tobacco smoking compared to before denic smoking were increased with greater carfentanil doses (p = 0.04). No dose effects of raclopride were found on cortisol levels. Peak prolactin levels after avnic smoking were increased with greater raclopride doses (p = 0.003). The changes in prolactin levels after avnic smoking compared to before were also increased with greater doses of raclopride (p < 0.001). There was no effect of carfentanil on plasma prolactin.
Conclusion:No correlations between the doses of the radioligands and the basal hormone levels were found. However in the presence of nicotine tobacco smoking the PET ligands had significant subtle clinical pharmacological effects on both cortisol and prolactin.
Keywords
PET, Carfentanil, Raclopride, Mass dose, Endocrine
Introduction
[11C] Carfentanil and [11C] raclopride are selective PET tracers for mu opioid receptors and dopamine D2/D3 receptors with agonist or antagonist effects. PET tracers must be given to humans in mass doses that have no obvious clinical pharmacological effects. An important basic science issue is whether such tiny mass doses have any subtle pharmacologic effects. The present study involves the use of nicotine in tobacco smoking to increase venous plasma cortisol and prolactin during PET imaging. The prototype classic mu opioid receptor agonist is morphine. In humans acute morphine reduces venous cortisol levels [1] and increases prolactin levels [2,3]. Hence the very potent mu agonist carfentanil should also decrease cortisol and increase prolactin levels with increasing doses. Amphetamine induces mesolimbic brain dopamine and plasma cortisol releases [4]. On the other hand dopamine receptor agonists inhibit prolactin release [5]. Raclopride is a specific antagonist for dopamine D2/D3 receptors [6] and may increase plasma prolactin in large doses.
The typical PET doses of total carfentanil are approximately 1 to < 10 μg with 3 to 20 mCi [7-9]. The typical PET doses of raclopride are commonly reported only in radioactivity such as 3 to 10 mCi [6,10]. Although many articles regarding PET scans using these radiotracers have been published, only a few reports mention any pharmacological effects of the tracers. Wand et al. [11] reported no agonist effects of carfentanil < 40 ng/kg. Scott et al. [12] concluded no pharmacological effect with 48.0 ± 37.0 ng/kg of carfentanil and 89.0 ± 47.0 ng/kg of raclopride. In this study the doses of carfentanil and raclopride were 3.5 to 34.4 ng/kg (mean ± SD 16.7 ± 9.8 ng/kg, 10-15 mCi) and 0.8 to 45.1 ng/kg (mean ± SD 12.8 ± 10.8 ng/kg, 10-15 mCi) respectively. This present study provides evidence that even the very small mass doses of carfentanil and raclopride used in PET studies have subtle effects on tobacco/nicotine endocrine actions.
Materials and Methods
Twenty four healthy American male 20-36 years old smokers were recruited for this study. Their average weight was 88.8 ± 12.8 kg. They smoked 15-40 cigarettes per day for at least 1 year. The detailed subject criteria and the procedures of PET scans used for this study were same as previous studies [13,14]. Two different research tobacco cigarettes, average nicotine (avnic) and denicotinized (denic) were used. The avnic and denic cigarette included 1.01 and 0.08 mg of nicotine and 9.5 and 9.1 mg of tar respectively. Both cigarettes had identical filter tips and no flavors added. Rate of puff and depth of inspiration were similar in both cigarettes. The denic smoking produced a very minor increase in venous plasma nicotine compared to avnic smoking which increased plasma nicotine levels > 10 ng/ml. Several subjects were excluded due to lack of hormonal, PET scan, or radioligand data. For cortisol analyses, N =17 and 20 for carfentanil and raclopride. For prolactin analysis, N = 13 and 14 for carfentanil and raclopride respectively.
Two PET scans were subsequently completed with a 15 min break between the scan sessions. In the first scan session started from around 8:30 am, one of the PET ligands was administrated via i.v. in the PET scanner and the subjects smoked denic cigarettes. In the second scan session, the other PET ligand was administrated and the subjects smoked avnic cigarettes. On another day, the subjects had other two PET scans with counterbalanced tracer-cigarette combinations. In all sessions, the time of radiotracer administration was considered as 0 min. The subjects inhaled smoke of two denic cigarettes from an enclosed gallon bottle at 43-53 min of the first session and the subjects inhaled smoke of two avnic cigarettes from an enclosed gallon bottle at 43-53 min of the second session. Each PET scan lasted for about 95 min. The administrated mass doses of carfentanil and raclopride were 3.5 - 34.4 ng/kg (mean ± SD 16.7 ± 9.8 ng/kg) and 0.8 - 45.1 ng/kg (mean ± SD 12.8 ± 10.8 ng/kg). Cortisol and prolactin data were measured at 0, 15, 30, 43, 49, 59, 65, 75, and 95 min during each. The hormone levels measured before either denic or avnic smoking are labeled as before. The highest hormone levels after either denic or avnic smoking are labeled as peak. The changes (delta) in hormonal levels were calculated as peak minus before. Venous blood samples were sent to the University of Michigan CLASS lab for plasma hormonal analyses. Correlations between each hormone level and either carfentanil or raclopride mass dose were determined by linear regression analyses with using the EZR version 1.2121 [15]. The p value was considered as significant if < 0.05.
Results
Radiotracer mass dose effects on cortisol levels
Seventeen of 20 healthy smokers were studied for carfentanil mass dose effects on plasma cortisol. After carfentanil administration, the cortisol levels before smoking (before), peak after smoking (peak) and changes (delta) were 10.3 ± 4.0, 8.4 ± 3.7 and -1.9 ± 0.7 μg/ml (mean ± SD) with denic, and 7.5 ± 3.6, 11.2 ± 4.5 and 3.7 ± 3.7 μg/ml with avnic smoking. The delta cortisol levels after denic (F(1,15) = 5.99, p = 0.03), peak cortisol levels after avnic (F(1.15) = 13.3, p = 0.002) and delta cortisol levels after avnic (F(1.15) = 4.96, p = 0.04) were significantly correlated with carfentanil mass dose (Figure 1).
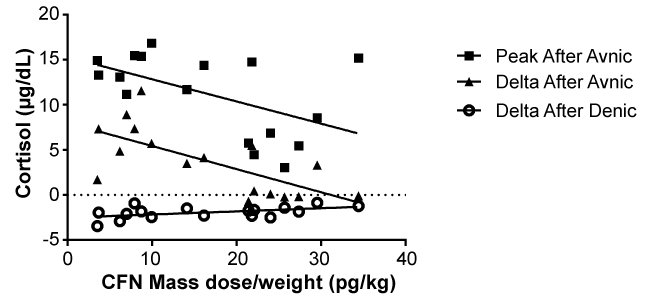
.
Figure 1: Venous plasma cortisol levels and carfentanil mass doses. Peak and delta cortisol levels after avnic smoking and carfemtanil mass doses were significantly negatively correlated (p = 0.002 for peak, 0.04 for delta). Delta cortisol levels after denic smoking and the mass doses were significantly positively correlated (p = 0.03).
View Figure 1
All 20 subjects were studied for raclopride mass dose effects on cortisol. After raclopride administration, the cortisol levels of before, peak and delta were 11.3 ± 4.1, 11.5 ± 4.2 and 0.3 ± 1.8 μg/ml with denic, and 6.1 ± 4.4, 12.7 ± 6.2 and 6.6 ± 5.2 μg/ml with avnic. No correlation between raclopride mass doses and cortisol levels were found.
Radiotracer mass dose effects on prolactin levels
Thirteen of 20 subjects were studied for carfentanil mass dose effects on prolactin. After carfentanil administration, the prolactin levels of before, peak and delta were 6.4 ± 2.1, 7.3 ± 2.1 and 0.9 ± 0.8 ng/ml with denic, and 6.3 ± 2.4, 9.2 ± 4.6 and 2.8 ± 3.4 ng/ml with avnic smoking. There was no correlation was found between the mass dose and the prolactin levels.
Fourteen of 20 subjects were studied for raclopride mass dose effects on prolactin. After raclopride administration, the prolactin levels 5.7 ± 1.8, 7.3 ± 2.5 and 1.6 ± 1.4 ng/ml with denic, and 5.3 ± 1.7, 7.6 ± 2.9 and 2.3 ± 0.5 ng/ml with avnic. The peak prolactin levels after avnic (F(1,12)=14.15, p = 0.003) and delta prolactin levels after avnic (F(1.12) = 34.02, p < 0.001) were significantly correlated with raclopride mass dose (Figure 2).
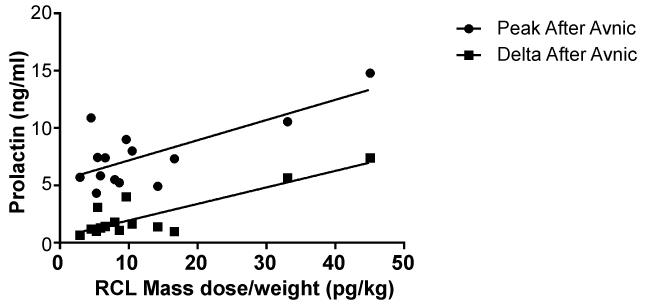
.
Figure 2: Venous plasma prolactin levels and raclopride mass doses. Peak and delta prolactin levels after avnic smoking and raclopride mass doses were significantly positively correlated (p = 0.003 for peak, p < 0.001 for delta).
View Figure 2
Discussion
Plasma cortisol and prolactin levels were chosen to be measured as possible indicators of very subtle PET ligand clinical and pharmacological actions. The initial hypothesis for this study was that the PET tracer doses of carfentanil and raclopride had no effects on tobacco smoked cortisol and prolactin levels because the doses are clinically subtheraputic. Nicotine is well-known to increase cortisol and prolactin levels [16,17]. To exclude the nicotine effects, the hormone levels before smoking were studied first. No significant correlations between the basal hormone levels and the mass doses were found. However, their mass doses correlated mainly with the peak and delta hormone levels after avnic smoking. This indicates that radiotracer mass dose effects are real but not clinically significant without an exogenous trigger such as nicotine/tobacco smoking.
The analytic variations in cortisol and prolactin blood levels were small compared to the effects by nicotine inhalation in the total group. The associations between the hormone levels and tracer mass doses were determined by linear regression study. The effects of mu opioid receptor agonists on glucocorticoids release are very different in humans compared to animals. In rats, morphine and fentanyl increase plasma corticosterone levels [18]. Pascoe et al. [19] found no cortisol changes with fentanyl (0.0003 - 0.02 mg/kg) in monkeys. In humans, morphine and other opioid agonists [20,21] inhibit the HPA axis and result in decreases in plasma cortisol levels [22]. Therefore carfentanil as a potent mu opioid receptor agonist decreases cortisol levels with larger mass dose with exogenous nicotine/tobacco smoking in this study. However the cortisol levels after denic smoking were increased with carfentanil mass dose. Although this result can be explained as significant cortisol release induced by PET scan related physical and psychological stress, more studies are needed to determine the effects of mass dose and denic smoking. The peak and delta prolactin levels were significantly positively correlated with raclopride mass doses with only avnic smoking. Denic smoking does not change cortisol and prolactin levels [23]. No correlations between heart rate and the tracers were found in the present study (data not shown).
One of the major basic as well as clinical issues in pharmacology is the number of receptors occupied by an agonist or antagonist when given in therapeutic doses. In the case of tobacco smoking, only 1 to 2 puffs of a nicotine containing cigarette results in only in a 0.87 ng/ml of nicotine venous plasma concentration. Surprisingly this very small increase in nicotine plasma levels results in 50% of α4β2 nicotinic acetylcholine receptors occupancy for 3.1 hours [24]. Perhaps the fact that in the present study far more α4β2 nicotinic acetylcholine receptors were occupied by nicotine permitted the minor pharmacological effects of mass doses of these two PET ligands to be measured.
The total carfentanil doses in this study were in the range of the doses commonly used for humans which is 26 to 30 ng/kg and are less than 15% of the therapeutic dose for conscious sedation [9,25-28]. Compared to the carfentanil in vitro EC50 = 1.30 ± 0.15 nM for mu opioid receptors [29], the tracer dose of carfentanil of 30 ng/ml in a 70 kg human body equals about 1.0 nM which may be significant even if non-specific binding is taken account.
Regarding raclopride, the doses used in this study (0.8 to 45.1 ng/kg) were similar to the doses in other studies which were 10 to 30 ng/kg [30,31]. The raclopride Kd for dopamine D2 receptors in humans is 10 nM [32]. Farde et al. [33] clearly stated the total tracer dose should be less 1 μg in total for human PET studies. Furthermore the same group [34] reported that raclopride increased plasma prolactin in a pilot study with mg order doses. Radioligand doses even if it is very small may have subtle pharmacological effects in the presence of an exogenous trigger such as nicotine/tobacco smoking. It should be noted that this research study is not clinically relevant because the subjects were smoking tobacco cigarettes during the PET scanning. Obviously smoking is not permitted during such procedure. Smoking was used as a trigger to enhance cortisol and prolactin levels to observe any differences with radiotracer injection.
Conclusions
1. PET imaging is a very important medical technique widely used and generally radiotracers are thought to have no obvious clinical pharmacological effects.
2. While this is true the present study reminds us all that subtle clinical and pharmacological actions are produced by the two radioactive ligands, [11C] raclopride and [11C] carfentanil.
Acknowledgements
We thank the members of the University of Michigan Hospital PET Facility. The authors also would like thank Drs. Catherine L. Evans, Robert A. Koeppe for their help in analyzing some of the PET imaging data. In addition, Dr. Kathleen B. Welch, statistician Staff Specialist in the University of Michigan Center for Statistical Consultation and Research was very helpful with statistical analyses.
Ethical Statement
All procedures performed in this study involving human participants were in accordance with the ethical standards of the institutional (University of Michigan Institutional Review Board) and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Informed consent was obtained from all individual participants included in the study.
Financial Support
This work was supported in part by the Department of Pharmacology Psychopharmacology Research Fund C361024, Education and Research Development Fund 276157, the National Institute of Health Grant R01 DA 016423, and the Smoking Research Foundation (Japan).
References
-
Zis AP, Haskett RF, Albala AA, Carroll BJ (1984) Morphine inhibits cortisol and stimulates prolactin secretion in man. Psychoneuroendocrinology 9: 423-427.
-
Delitala G, Grossman A, Besser GM (1983) The participation of hypothalamic dopamine in morphine-induced prolactin release in man. Clin Endocrinol (Oxf) 19: 437-444.
-
Vuong C, Van Uum SH, O'Dell LE, Lutfy K, Friedman TC (2010) The effects of opioids and opioid analogs on animal and human endocrine systems. Endocr Rev 31: 98-132.
-
Oswald LM, Wong DF, McCaul M, Zhou Y, Kuwabara H, et al. (2005) Relationships Among Ventral Striatal Dopamine Release, Cortisol Secretion, and Subjective Responses to Amphetamine. Neuropsychopharmacology 30: 821-832.
-
Lu K-H, Meites J (1971) Inhibition by L-Dopa and Monoamine Oxidase Inhibitors of Pituitary Prolactin Release; Stimulation by Methyldopa and d-Amphetamine. Exp Biol Med 137: 480-483.
-
Farde L, Ehrin E, Eriksson L, Greitz T, Hall H, et al. (1985) Substituted benzamides as ligands for visualization of dopamine receptor binding in the human brain by positron emission tomography. Proc Natl Acad Sci USA 82: 3863-3867.
-
Frost JJ, Douglass KH, Mayberg HS, Dannals RF, Links JM, et al. (1989) Multicompartmental analysis of [11C]-carfentanil binding to opiate receptors in humans measured by positron emission tomography. J Cereb Blood Flow Metab 9: 398-409.
-
Kuwabara H, Heishman SJ, Brasic JR, Contoreggi C, Cascella N, et al. (2014) Mu Opioid Receptor Binding Correlates with Nicotine Dependence and Reward in Smokers. PLoS One 9: e113694.
-
Ray R, Ruparel K, Newberg A, Wileyto EP, Loughead JW, et al. (2011) Human Mu Opioid Receptor (OPRM1 A118G) polymorphism is associated with brain mu-opioid receptor binding potential in smokers. Proc Natl Acad Sci U S A 108: 9268-9273.
-
Volkow ND, Wang GJ, Smith L, Fowler JS, Telang F, et al. (2015) Recovery of dopamine transporters with methamphetamine detoxification is not linked to changes in dopamine release. Neuroimage 121: 20-28.
-
Wand GS, Weerts EM, Kuwabara H, Frost JJ, Xu X, et al. (2011) Naloxone-induced cortisol predicts mu opioid receptor binding potential in specific brain regions of healthy subjects. Psychoneuroendocrinology 36: 1453-1459.
-
Scott DJ, Stohler CS, Koeppe RA, Zubieta JK (2007) Time-course of change in [11C]carfentanil and [11C]raclopride binding potential after a nonpharmacological challenge. Synapse 61: 707-714.
-
Domino EF, Evans CL, Ni L, Guthrie SK, Koeppe RA, et al. (2012) Tobacco smoking produces greater striatal dopamine release in G-allele carriers with mu opioid receptor A118G polymorphism. Prog Neuropsychopharmacol Biol Psychiatry 38: 236-240.
-
Domino EF, Hirasawa-Fujita M, Ni L, Guthrie SK, Zubieta JK (2015) Regional brain [(11)C]carfentanil binding following tobacco smoking. Prog Neuropsychopharmacol Biol Psychiatry 59: 100-104.
-
Kanda Y (2013) Investigation of the freely available easy-to-use software 'EZR' for medical statistics. Bone Marrow Transplant 48: 452-458.
-
Mendelson JH, Goletiani N, Sholar MB, Siegel AJ, Mello NK (2008) Effects of smoking successive low- and high-nicotine cigarettes on hypothalamic-pituitary-adrenal axis hormones and mood in men. Neuropsychopharmacology 33: 749-760.
-
Wilkins JN, Carlson HE, Van Vunakis H, Hill MA, Gritz E, et al. (1982) Nicotine from cigarette smoking increases circulating levels of cortisol, growth hormone, and prolactin in male chronic smokers. Psychopharmacology (Berl) 78: 305-308.
-
Hayes AG, Stewart BR (1985) Effect of mu and kappa opioid receptor agonists on rat plasma corticosterone levels. Eur J Pharmacol 116: 75-79.
-
Pascoe JE, Williams KL, Mukhopadhyay P, Rice KC, Woods JH, et al. (2008) Effects of mu, kappa, and delta opioid receptor agonists on the function of hypothalamic-pituitary-adrenal axis in monkeys. Psychoneuroendocrinology 33: 478-486.
-
Bouloux PM, Grossman A, Allolio B, Delitala G, Besser GM (1986) Effect of adrenergic antagonism on the growth hormone, prolactin and cortisol response to a synthetic opioid agonist in man. Horm Res 23: 83-90.
-
Giusti M, Delitala G, Marini G, Uggias B, Sessarego P, et al. (1992) The effect of a met-enkephalin analogue on growth hormone, prolactin, gonadotropins, cortisol and thyroid stimulating hormone in healthy elderly men. Acta Endocrinol (Copenh) 127: 205-209.
-
Pechnick RN (1993) Effects of opioids on the hypothalamo-pituitary-adrenal axis. Annu Rev Pharmacol Toxicol 33: 353-382.
-
Xue Y, Morris M, Ni L, Guthrie SK, Zubieta JK, et al. (2010) Venous plasma nicotine correlates of hormonal effects of tobacco smoking. Pharmacol Biochem Behav 95: 209-215.
-
Brody AL, Mandelkern MA, London ED, Olmstead RE, Farahi J, et al. (2006) Cigarette smoking saturates brain alpha 4 beta 2 nicotinic acetylcholine receptors. Arch Gen Psychiatry 63: 907-915.
-
Colasanti A, Searle GE, Long CJ, Hill SP, Reiley RR, et al. (2012) Endogenous opioid release in the human brain reward system induced by acute amphetamine administration. Biol Psychiatry 72: 371-377.
-
Cortínez LI, Muñoz HR, De la Fuente R, Acuña D, Dagnino JA (2005) Target-controlled infusion of remifentanil or fentanyl during extra-corporeal shock-wave lithotripsy. Eur J Anaesthesiol 22: 56-61.
-
Goodman LS, Brunton LL, Blumenthal DK, Murri N, Hilal-Dandan R (2011) Goodman & Gilman's The pharmacological basis of therapeutics. New York: McGraw-Hill Medical.
-
Wager TD, Scott DJ, Zubieta JK (2007) Placebo effects on human mu-opioid activity during pain. Proc Natl Acad Sci U S A 104: 11056-11061.
-
Feldman PL, James MK, Brackeen MF, Bilotta JM, Schuster SV, et al. (1991) Design, synthesis, and pharmacological evaluation of ultrashort- to long-acting opioid analgetics. J Med Chem 34: 2202-2208.
-
Aalto S, Ingman K, Alakurtti K, Kaasinen V, Virkkala J, et al. (2015) Intravenous ethanol increases dopamine release in the ventral striatum in humans: PET study using bolus-plus-infusion administration of [(11)C]raclopride. J Cereb Blood Flow Metab 35: 424-431.
-
Martikainen IK, Nuechterlein EB, Peciña M, Love TM, Cummiford CM, et al. (2015) Chronic Back Pain Is Associated with Alterations in Dopamine Neurotransmission in the Ventral Striatum. J Neurosci 35: 9957-9965.
-
Farde L, Eriksson L, Blomquist G, Halldin C (1989) Kinetic analysis of central [11C]raclopride binding to D2-dopamine receptors studied by PET--a comparison to the equilibrium analysis. J Cereb Blood Flow Metab 9: 696-708.
-
Farde L, Wiesel FA, Hall H, Halldin C, Stone-Elander S, et al. (1987) No D2 receptor increase in PET study of schizophrenia. Arch Gen Psychiatry 44: 671-672.
-
Farde L, von Bahr C, Wahlen A, Nilsson L, Widman M (1989) The new selective D2-dopamine receptor antagonist raclopride--pharmacokinetics, safety and tolerability in healthy males. Int Clin Psychopharmacol 4: 115-126.





