International Archives of Addiction Research and Medicine
Flaxseed Oil Decreases Craving for Chocolate: Preliminary Results
Pedro Luis Prior1, Anna Carolina Ramos2, Jaqueline Kalleian Eserian2, Juçara Zaparoli2 and José Carlos Fernandes Galduróz2*
1Department of Medicine, Universidade Federal de São Paulo, Brazil
2Department of Psychobiology, Universidade Federal de São Paulo, Brazil
*Corresponding author:
José Carlos Fernandes Galduróz, Adjunct Professor Department of Psychobiology, Universidade Federal de São Paulo R. Napoleão de Barros, 925. Vila Clementino. Zip code: 04024-002. São Paulo, SP. Brazil, E-mail: galduroz@unifesp.br
Int Arch Addict Res Med, IAARM-1-010, (Volume 1, Issue 2), Brief Communication; ISSN: 2474-3631
Received: September 16, 2015 | Accepted: October 20, 2015 | Published: October 22, 2015
Citation: Prior PL, Ramos AC, Eserian JK, Zaparoli J, Galdurózc JCF (2015) Flaxseed Oil Decreases Craving for Chocolate: Preliminary Results. Int Arch Addict Res Med 1:010. 10.23937/2474-3631/1510010
Copyright: © 2015 Prior PL, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Chocolate is recognized as a type of food that contains many psychoactive components. Many studies have shown that some people may present compulsive symptoms such as the difficulty in resisting the urge to consume chocolate. On the other hand, there is evidence that omega 3 deficiency results in a dopaminergic hypofunctioning of the mesocortical and mesolimbic pathways related to reinforcing effects. Therefore, the objective of this study was to determine the effects of flaxseed oil (rich in omega 3) on chocolate consumption and craving. We conducted a double blind, placebo-controlled, randomized trial and assigned volunteers to two groups: placebo, which received capsules of paraffin plus dye (1000 mg), and omega 3, which received capsules containing flaxseed oil (1000 mg). The Binge Eating Scale (BES) was applied before supplementation and at the end of the study (two months) in order to determine if there was any change in chocolate consumption. After the treatment, there was a significant difference between the BES scores only in the omega 3 group before and after the intervention. The Newman-Keuls post-hoc test showed an increase of DHA in the omega 3 group after supplementation (p < 0.05). Hence, our results suggest that omega 3 fatty acids may be important in mitigating chocolate craving and consumption.
Keywords
Chocolate, Omega 3, Flaxseed oil, Craving, Binge eating scale
Introduction
Chocolate is recognized as a type of food that contains many psychoactive components (cannabinoid-like molecules, methylxanthines, long-chain carboxylic acids, and other factors) which activate the serotonergic system of the prefrontal cortex and, more importantly, the dopaminergic system, for short periods of time [1]. Many studies have shown that some people may present compulsive symptoms such as difficulty in resisting the urge to consume chocolate [2]. There is, therefore, similarity between this condition and the craving that happens in substance dependence.
Many neural pathways are involved in craving, especially serotoninergic, dopaminergic and opioidergic, derived from several cortical areas (prefrontal cortex, amygdala and hippocampus) [3,4]. There are still no specific treatments for these symptoms.
Modern theories profess that lack of balance of polyunsaturated fatty acids (PUFAs) in diets may be the cause of deleterious changes in the neuronal physiology, which may cause or worsen pathological conditions, such as depression, mood instability, and drug craving [5-8]. Some studies have shown the association of more frequent drug relapse and lower levels of PUFAs in cocaine addicts [6]. Another recent study has shown that oral administration of omega 3 fatty acids lowered cigarette craving in addicted patients [9]. To the best of our knowledge, there are no studies on the effect of PUFAs on chocolate craving.
There are two molecular families in the PUFAs classification: omega 3 and omega 6 fatty acids. Through a series of chemical reactions, alpha linolenic acid is transformed into either docosahexaenoic acid (DHA) or eicosapentaenoic (EPA) - both omega 3 fatty acids. Another PUFA is the linoleic acid that generates arachidonic acid (AA) - omega 6 fatty acids. Omega 6 fatty acids is pro-inflammatory, pro-thrombotic and seems to contribute to drug addiction and relapse, and omega 3 fatty acids inhibit these same processes, acting as antagonists of omega 6 fatty acids [5].
Flaxseed oil is rich in alpha-linolenic acid, the main precursor of omega 3 fatty acids and linoleic acid, rich in omega 6. This combination determined our choice to administer flaxseed oil, since it contains both omega 6 and omega 3, in the ratio 4:1, even though several studies suggest the ideal ingestion ranging from 3:1 up to 5:1 [10].
Therefore, the objective of this study was to determine if daily dietary supplementation with flaxseed oil might successfully mitigate chocolate consumption and craving.
Methodology
This is a double blind, placebo-controlled, randomized trial. The inclusion criteria were: both genders; age between 18 and 45 years; BMI between 18.5 and 25, and binge eating scale (BES) score greater than 18. The exclusion criteria were: volunteers diagnosed with any psychiatric disorder (according to DSM-IV-TR criteria) and those taking any psychoactive medication, or using any recreational drugs. The Beck Depression Inventory and the Beck Anxiety Inventory [11] were also applied. Any volunteer classified as moderately depressed or anxious according to their score in those instruments was excluded.
Our initial sample consisted of 29 females (mean age 30 years old). Four volunteers dropped out of the study for personal reasons, not related to the experiment itself, such as side effects of the flaxseed oil or any other difficulties. Therefore, our final sample was of 25 volunteers (N = 25).
The volunteers considered eligible filled the BES, informed the amount of chocolate (g/day) ingested before and after two months of treatment. Additionally, they provided blood samples on the same occasions.
We assigned them to two groups: the placebo group received capsules containing a formula of paraffin and # 2 dye, simulating the visual aspect of fatty acids, and the experimental group received capsules containing 1000 mg of flaxseed oil, in the proportion of 4:1 of omega 6 and omega 3, respectively. The intervention lasted two months. Volunteers were instructed to take one capsule three times a day, and not to modify their regular diet during the study.
In order to obtain objective data about craving symptoms, we used the binge eating scale (BES), originally developed by Gormally et al. [12] and translated into Portuguese by Freitas et al. [13]. The instrument evaluated three aspects: typical behavioral manifestations (shame and social isolation), subjective feelings in regard to daily consumption of chocolate, and frequency and amount of chocolate consumption. Every question was multiple-choice and self-reported, presenting 3 to 4 different alternatives each. Total scores evaluated different states: scores lower than 17 were correlated with minimal or non-existent chocolate craving; scores between 18 and 26-regarded moderate craving, and scores higher than 27 indicated severe craving.
In order to assess the PUFAs (EPA, DHA, AA and Linoleic acid) levels of participants, we utilized the high performance liquid chromatography (HPLC) technique, dosed at baseline in blood samples of the volunteers and after two months of supplementation. The quantification protocol of PUFAs dosage was performed according to Abushufa in 1994 [14] using the HPLC. Briefly, total lipids were extracted from plasma, using water-chloroform-methanol [15]. Fatty acids were prepared with 4-bromomethyl-7-coumarin and then separated on a Shimadzu LC-10A high performance liquid chromatograph using an octa-decylsilica column (25 cm × 4.6 mm i.d.; particle size 5 mm). Fatty acids were resolved using a mobile phase of acetonitrile-water (85:15 vol/vol) at a flow rate of 1.5 ml/min. Fatty acid derivatives were detected by fluorescence (325-nm excitation; 395-nm emission).
The project was approved by the committee of ethics in research under registration number 0695/10. We obtained informed consent form from all participants.
Statistical Analyses
We analyzed data with ANOVA for repeated measures and Newman-Keuls post-hoc test (for measurements in the omega 3 group - BES score between both groups at the beginning and end of study). Data from the laboratorial analyses were paired with BES scores using Pearson's correlation. The other data concerning group characteristics - initial BMI, Beck anxiety and depression scores, Chocolate daily consumption - between the groups in both measurements were compared using t Student test. The level of significance was p < 0.05 for all analyses.
Results
Table 1 shows the comparison between the placebo (n = 14) and the experimental (n = 11) groups, before and after the treatment. There were no statistically significant differences, before the intervention, between the placebo and experimental groups concerning the following parameters: educational level, body mass index, anxiety, depression and initial BES scores.
![]()
Table 1: Characteristics of the sample (age and BMI), anxiety, daily consumption of chocolate in grams and BES score, before and after treatment.
View Table 1
After the two-month trial period, there was a significant difference between the BES scores of the experimental and placebo groups (ANOVA for repeated measures: F1,24 = 111.878; p < 0.001). In the experimental group, there was a significant difference between the BES scores before and after treatment (post hoc Newman-Keuls p < 0.001) (Figure 1).
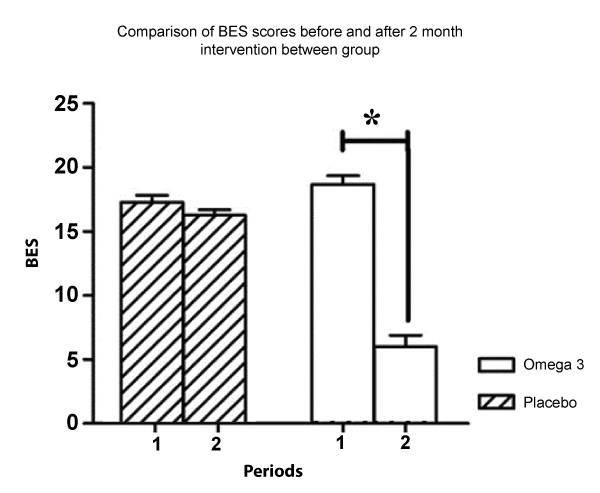
.
Figure 1: BES scores before (column 1) and after (column 2) the two months intervention in both groups.
View Figure 1
The consumption of chocolate was also different between groups after treatment. The ANOVA pointed to a significant effect of time (F1,24 = 15.207; p < 0.001), and group (F1,24 = 15.589; p < 0.001). According to the post-hoc test, the experimental group significantly reduced chocolate consumption after treatment (p < 0.001), while the placebo group kept a similar mean consumption (p > 0.05) (Table 2).
![]()
Table 2: Pearson's correlations between dosages of plasmatic fatty acids and BES, in the placebo group, before (1) and after (2) treatment.
View Table 2
The fatty acid profile established by the HPLC method was analyzed, and there was a significant difference in the DHA levels between groups (F1,16 = 5.4963; p < 0.05). The Newman-Keuls post-hoc test showed an increase of DHA in the experimental group after supplementation for two months (p < 0.05). The mean percentage of DHA in the experimental group increased from 3.45% in the pre-treatment to 4.75% in the post-treatment. The other dosages did not show significant variations in either group (Table 3).
![]()
Table 3: Pearson's correlations between dosages of plasmatic fatty acids and BES, in the experimental group, before (1) and after (2) treatment.
View Table 3
When data from BES scores were paired with the fatty acid fractions obtained, a strong negative correlation was observed between the amounts of DHA in the experimental group (DHA 1) in the beginning of the experiment and the initial BES score (BES 1). The results showed that the participants most affected were the ones who presented lower concentrations of DHA fatty acids (Pearson correlation = -0.66). This correlation was practically eliminated in the analysis after two months of intervention (DHA 2 and BES 2), thus indicating the participants were no longer characterized as persons craving for chocolate. Additionally, fatty acid levels were normalized (Pearson correlation = 0,17). There was a negative correlation between the increasing EPA levels and the BES scores after the intervention in the experimental group (Pearson correlation = -0.57), suggesting that this component also helps reduce craving symptoms. There were no clear significant correlations in the placebo group.
Discussion
The results of this study showed that the participants who ingested flaxseed oil (3,000 mg/day) for two months presented lower scores in the binge eating scale (BES) after intervention. One possible explanation for these findings could be the role of omega 3 in regulating the functioning of the dopaminergic systems [16]. Some studies show that omega 3 deficiency results in a dopaminergic hypofunctioning of the mesocortical and mesolimbic pathways related to reinforcing effects [5,6]. Therefore, our results would suggest that there was a regulation of dopamine neurotransmission and the consequent reduction of craving for chocolate. However, we should bear in mind that flaxseed oil contains two forms of PUFAs (Omega 6 and Omega 3) in a ratio close to that recommended for a healthy diet which, according to several studies, should be 3:1 and 5:1, respectively [10].
Studies employing functional magnetic resonance imaging (fMRI) detected that stimulation by chocolate enhances the brain activities of the reward systems comprised by areas such as the striatum, the medial orbitofrontal cortex and the cingulate gyrus [17].
Several researches demonstrated that those brain areas can also be activated by a number of highly palatable foods [18-21]. The same might happen with stimuli of other natures, notably music, painting and monetary rewards [22-25].
It is widely recognized that the brain regions related to reward mechanisms are mediated by dopaminergic pathways through the release of dopamine from the ventral tegmental area into their terminals in the nucleus accumbens and striatum [26].
Another aspect to be highlighted regards the correlation between the reduction in the BES and the increase in the blood levels of DHA. In other words, the supplementation with omega 3 played an important role in reducing craving for chocolate. Some studies assert that this craving increases when people stop ingesting that omega, remaining for about two weeks [27]. Increased levels of anxiety are also observed under those circumstances [28,29]. They can increase even further when the individuals are exposed to smell or images of chocolate [30,31].
Finally, high performance liquid chromatography (HPLC) detected higher concentrations of DHA-fraction of omega 3 only in the group which received flaxseed oil. Consequently, we can conclude that the experimental group ingested the capsules regularly, and that the placebo group followed the instruction not to change their diet during the period of the study. Even though our sample was small, the results seem to be promising to mitigate chocolate craving and consumption.
Acknowledgments
Fundacao de Amparo a Pesquisa do Estado de Sao Paulo (FAPESP), Associacao Fundo de Incentivo a Pesquisa (AFIP) and Conselho Nacional de Pesquisa (CNPq).
Clinical Trial Information: Ethical approval: # 0695/10; Clinical Trials # NCT01391624.
References
-
Bruinsma K, Taren DL (1999) Chocolate: food or drug? J Am Diet Assoc 99: 1249-1256.
-
Frings D, Eskisan G, Spada MM, Albery IP (2015) Levels of craving influence psychological challenge and physiological reactivity. Appetite 84: 161-165.
-
Heinz A, Beck A, Grusser SM, Grace AA, Wrase J (2009) Identifying the neural circuitry of alcohol craving and relapse vulnerability. Addict Biol 14: 108-118.
-
Weiss F, Ciccocioppo R, Parsons LH, Katner S, Liu X, et al. (2001) Compulsive drug-seeking behavior and relapse. Neuroadaptation, stress, and conditioning factors. Ann N Y Acad Sci 937: 1-26.
-
Haag M (2003) Essential fatty acids and the brain. Can J Psychiatry 48: 195-203.
-
Buydens-Branchey L, Branchey M, McMakin DL, Hibbeln JR (2003) Polyunsaturated fatty acid status and relapse vulnerability in cocaine addicts. Psychiatry Res 120: 29-35.
-
UN Das (2003) Long-chain polyunsaturated fatty acids in the growth and development of the brain and memory. Nutrition 19: 62-65.
-
Richardson AJ (2004) Long-chain polyunsaturated fatty acids in childhood developmental and psychiatric disorders. Lipids 39: 1215-1222.
-
Rabinovitz S (2014) Effects of omega-3 fatty acids on tobacco craving in cigarette smokers: A double-blind, randomized, placebo-controlled pilot study. J Psychopharmacol 28: 804-809.
-
Simopoulos AP (2004) Omega-6/Omega-3 essential fatty acid ratio and chronic diseases. Food Rev Inter 20: 77-90.
-
Beck AT, Ward CH, Mendelson M, Mock J, Erbaugh J (1961) An inventory for measuring depression. Arch Gen Psychiatry 4: 561-571.
-
Gormally J, Black S, Daston S, Rardin D (1982) The assessment of binge eating severity among obese persons. Addict Behav 7: 47-55.
-
S Freitas, C Lopes, W Coutinho, JC Appolinario (2001)Traducao e adaptacao para o portugues da Escala de Compulsao Alimentar Periodica. Rev Bras Psiquiatr 23: 215-220.
-
Abushufa R, Reed P, Weinkove C (1994) Fatty acids in erythrocytes measured by isocratic HPLC. Clin Chem 40: 1707-1712.
-
Folch J, Lees M, Sloane Stanley GH (1957) A simple method for the isolation and purification of total lipides from animal tissues. J Biol Chem 226: 497-509.
-
Dyall SC, Michael-Titus AT (2008) Neurological benefits of omega-3 fatty acids. Neuromolecular Med 10: 219-235.
-
Rolls ET, McCabe C (2007) Enhanced affective brain representations of chocolate in cravers vs. non-cravers. Eur J Neurosci 26: 1067-1076.
-
Burger KS, Stice E (2011) Relation of dietary restraint scores to activation of reward-related brain regions in response to food intake, anticipated intake, and food pictures. NeuroImage 55: 233-239.
-
Kringelbach ML (2005) The human orbitofrontal cortex: linking reward to hedonic experience. Nat Rev Neurosci 6: 691-702.
-
O'Doherty JP, Deichmann R, Critchley HD, Dolan RJ (2002) Neural responses during anticipation of a primary taste reward. Neuron 33: 815-826.
-
Small DM, Veldhuizen MG, Felsted J, Mak YE, McGlone F (2008) Separable substrates for anticipatory and consummatory food chemosensation. Neuron 57: 786-797.
-
Blood AJ, Zatorre RJ (2001) Intensely pleasurable responses to music correlate with activity in brain regions implicated in reward and emotion. Proc Natl Acad Sci USA 98: 11818-11823.
-
Kahnt T, Heinzle J, Park SQ, Haynes JD (2010) The neural code of reward anticipation in human orbitofrontal cortex. Proc Natl Acad Sci USA 107: 6010-6015.
-
Kringelbach ML, O'Doherty J, Rolls ET, Andrews C (2003) Activation of the human orbitofrontal cortex to a liquid food stimulus is correlated with its subjective pleasantness. Cereb Cortex 13: 1064-1071.
-
Lane RD, Reiman EM, Ahern GL, Schwartz GE, Davidson RJ (1997) Neuroanatomical correlates of happiness, sadness, and disgust. Am J Psychiatry 154: 926-933.
-
Bjorklund A, Dunnett SB (2007) Dopamine neuron systems in the brain: an update. Trends Neurosci 30: 194-202.
-
Polivy J, Coleman J, Herman CP (2005) The effect of deprivation on food cravings and eating behavior in restrained and unrestrained eaters. Int J Eat Disord 38: 301-309.
-
Davis C, Strachan S, Berkson M (2004) Sensitivity to reward: implications for overeating and overweight. Appetite 42: 131-138.
-
Franken IH, Muris P (2005) Individual differences in reward sensitivity are related to food craving and relative body weight in healthy women. Appetite 45: 198-201.
-
Rodriguez, S Fernandez MC, Cepeda-Benito A, Vila J (2005) Subjective and physiological reactivity to chocolate images in high and low chocolate cravers. Biol Psychol 70: 9-18.
-
Moreno-Dominguez S, Rodriguez-Ruiz S, Martin M, Warren CS (2012) Experimental effects of chocolate deprivation on cravings, mood, and consumption in high and low chocolate-cravers. Appetite 58: 111-116.





