Clinical Medical
Reviews and Case Reports
Ruptured Intracranial Teratoma: A Case Report and Literature Review
Luis R. Romero1 , Betty Y. Chen2 , Miguel A. Guzman2 , Yihua Zhou3 , Jin-Ping Lai2 and Fangxiang Chen1,4*
1Department of Neurosurgery, Saint Louis University, USA
2Department of Pathology, Saint Louis University, USA
3Department of Radiology, Saint Louis University, USA
4St Anthony's Medical Center, Saint Louis, USA
*Corresponding author:
Fangxiang Chen, Department of Neurosurgery, Saint Louis University, 3635 Vista Ave, Saint Louis, MO 63104, USA, Tel: 314-577-8715, Fax: 314-577-8720, E-mail: fchen10@slu.edu
Clin Med Rev Case Rep, CMRCR-2-029,, (Volume 2, Issue 5), Case Report; ISSN: 2378-3656
Received: March 16, 2015 | Accepted: May 18, 2015 | Published: May 20, 2015
Citation: Romero LR, Chen BY, Guzman MA, Zhou Y, Jin-Ping L, et al. (2015) Ruptured Intracranial Teratoma: A Case Report and Literature Review. Clin Med Rev Case Rep 2:029. 10.23937/2378-3656/1410029
Copyright: © 2015 Romero LR, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Teratomas are germinal cell tumors originating from all three germ cell layers. When intracranial in location, they typically occur in the pineal or suprasellar regions. Ruptured intracranial teratomas are exceedingly rare. The authors report a case of a 34-year-old woman with a ruptured fourth ventricular mature teratoma that was treated with open surgery. The patient initially presented complaints of headache, nausea, and dizziness. CT scan demonstrated a mixed density lesion in the fourth ventricle and hypodensity lesions in bilateral lateral ventricles. MRI imaging revealed a complex fourth ventricular lesion and high T1 signal bilaterally in the lateral ventricles. A ruptured fourth ventricular teratoma was suspected and the patient was taken for surgery. Histopathology confirmed the diagnosis. Due to CSF leaking from her wound she ultimately underwent a ventriculoperitoneal shunt procedure several days later for persistent pseudomeningocele and hydrocephalus. A review of the literature is performed. Spontaneous rupture of intracranial mature teratomas is seldom encountered. The later atypical presentation may be attributed to the tumor's unusual location, precluding the more typical visual or endocrine abnormalities which may have otherwise resulted in an earlier diagnosis.
Keywords
Teratoma, Intraventricular tumor, Ruptured teratoma, Fourth ventricular tumor, Germ cell tumor
Introduction
Intracranial teratomas are very rare mass lesions. Though there are reports of ruptured extracranial teratomas in the literature [1-3], spontaneous rupture of intracranial mature teratomas is seldom encountered. The authors report a ruptured fourth ventricular mature teratoma with its content migrating superiorly into the bilateral lateral ventricles.
Case Report
History and presentation
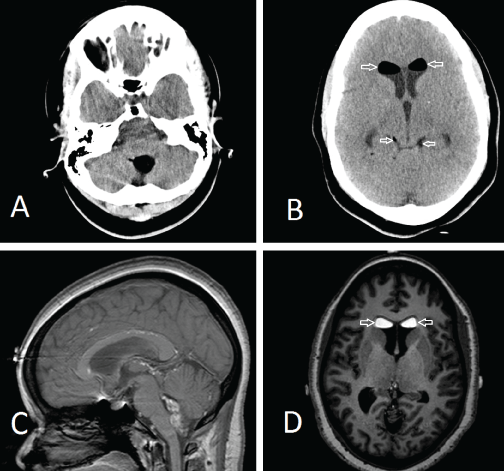
A 34-year-old female with a 10-year history of migraines presented with acute onset nausea, headache, and dizziness that occurred while sitting at work. After seeking medical attention, she was found to be neurologically intact on physical exam. CT scan of the head was performed and demonstrated a mixed density mass in the fourth ventricle with peripheral calcifications, septations, and soft tissue components. Hypodense lesions were seen in the lateral ventricles, third ventricle, quadric geminal cistern, subarachnoid space, and interhemispheric fissure. The lesions in the lateral ventricles demonstrated hypo attenuations as low as -160 Hounsfield Units, consistent with fat. Ventriculomegaly was also evident, due outlet obstruction of the fourth ventricle (Figure 1A,1B). MRI also confirmed the presence of a fat-containing mass in the fourth ventricle, with scattered fat droplets in the same places seen on CT (Figure 1C,1D).
.
Figure 1: Imaging study of the ruptured intracranial teratoma. A. Non-contrast CT, low density lesion in the fourth ventricle; B. Non-contrast CT, enlarged ventricles with hypodense lesions (arrows) in the lateral ventricles; C. Sagittal contrasted T1 MRI demonstrating fourth ventricular lesion; and D. Axial non-contrast T1 MRI demonstrating increased signal (arrows) in the lateral ventricles.
View Figure 1
Operation
The patient was taken to the operating room where she was placed in the prone position with her head secured in a Mayfield head holder. A suboccipital craniotomy was performed including resection of the posterior C1 arch. Once the dura was opened, the mass could be seen protruding in the midline from the foramen of Magendie. A telovelar approach to the fourth ventricle was performed, allowing adequate access for a gross total resection. A very small portion of tumor capsule adherent to the floor of the fourth ventricle was left behind given the potential for significant complication. The dura was closed in a watertight fashion using a suturable dural substitute and tissue sealant glue. The patient tolerated the procedure well without any intraoperative complication.
Pathological findings
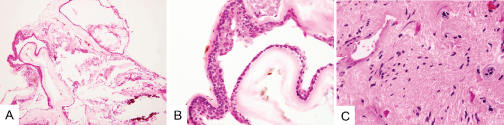
Histopathological examination revealed a multi-cystic mass comprised of tissues from all three embryonal cell layers: epidermis and cutaneous appendages (ectoderm); fat, bone, and muscle (mesoderm); cavities lined with respiratory type ciliated epithelium and mucinous epithelium (endoderm). Additionally, one specimen contained a 2 mm x 5 mm bony structure. The final pathology report confirmed the diagnosis of a mature cystic teratoma (Figure 2A-2C).
.
Figure 2: Histopathology of the intracranial teratoma. A-B. A representative section of the tumor showing a cystic lesion (arrows) lined focally by stratified squamous epithelium (H&E, A, 100 x; B, 400x); and C. Surrounding brain parenchyma showing gliosis and Rosenthal fibers (arrows) (H&E, 400 x).
View Figure 2
Postoperative course
She remained neurologically intact following the procedure, the patient was discharged to inpatient rehabilitation at physical therapy's recommendation. She returned several weeks later with a pseudomeningocele and CSF leaking from her wound. She was taken back to the operating room for duraplasty, however ultimately underwent a ventriculoperitoneal shunt procedure several days later for persistent pseudomeningocele and hydrocephalus.
Discussion
As one of six subtypes of germ cell tumors classified by the World Health Organization, teratomas derive from all three of the germ cell layers [4]. Although usually found in a mature benign form, they can also take the form of immature fetal appearing tissue and have the potential for malignant transformation [5]. Histologically, they often contain cystic and solid elements, containing fully differentiated tissue organized in a fashion resembling its associated organ. Frequently found elements include fat, bone, cartilage, teeth, and hair. They have been reported to occur in the lateral ventricles, pineal region, suprasellar regions, and the posterior fossa, while teratomas arising within the fourth ventricle have been rarely reported [6-12]. Patients with these lesions typically present in the first two decades of life [9]. For tumors originating in the pineal region, clinical presentation is usually that of hydrocephalus or Parinaud's Syndrome [13]. Those located in the suprasellar region are more likely to present with endocrine dysfunction or visual disturbances [5].
The pathogenesis of germ cell tumors remains a topic of debate. While the most widely accepted theory considers these tumors a result of a primordial totipotent germ cell, Sano published an alternative theory. He proposed that embryonic cells in various stages of development may become misplaced during primitive streak formation and ultimately become inappropriately enfolded into the brain during neural tube formation [14].
In this case, we report a patient who presents with a ruptured intracranial teratoma originating from the fourth ventricle. Although well described in the pineal and suprasellar regions, infratentorial teratomas originating in the fourth ventricle have only rarely been described [6-12]. In 2010, Xuzhu et al. reported a ruptured teratoma with a large posterior fossa component and a smaller suprasellar component after a patient presented with cranial nerve dysfunction [15].
An intraventricular or subarachnoid low density lesion should raise suspicion for a ruptured teratoma. CT scans of teratomas usually demonstrate fat densities, which can be diagnostic. In this case, we obtained a measurement of -160 Hounsfield Units in the lateral ventricles. MRI imaging can readily identify intraventricular free fat and distinguish it from other pathology such as pneumocephalus or pneumoventricle based on a high T1 signal and fat suppression. The primary lesion is typically lobulated, with prominent enhancement of the solid portion or a thick wall as key features in differentiating mature versus malignant teratomas [16]. Fat suppression MRI and prone position MRI may be useful in distinguishing tumor from free fat [17].
Although the CT scan did suggest posterior fossa pathology in this particular case, initial imaging may not always identify the tumor's primary location. Their primary location may not be intracranial despite the initial intracranial findings. Once ruptured, spinal teratomas may release fatty contents which may ascend into the intracranial subarachnoid and ventricular space [18]. Oh et al. described a case of a lumbar spinal teratoma with rupture into the central canal with subsequent rostral migration of the lipid-laden contents into the intracranial subarachnoid and ventricular space [19].
Intracranial teratomas occur frequently; however, ruptured teratomas are rarely reported. Various mechanisms of rupture have been proposed, including physical injury, ischemic necrosis, and enzymatic degradation [1]. Nevertheless, the pathophysiology of this event within the cranial compartment continues to remains unclear. Once ruptured, a patient may rapidly deteriorate, presenting with hydrocephalus-like symptoms including headache, nausea, vomiting, and papilledema. Early recognition of a ruptured teratoma can be critical for successful and optimal management of patients.
Although a rare tumor in adults, teratoma is the most commonly found intracranial tumor at birth, and rupture in the perinatal period has also been documented. Bolat et al. reported a 25 week gestational age fetus with a massive intracranial teratoma discovered on initial prenatal ultrasound [20]. Fetal demise was also apparent and the diagnosis was confirmed on autopsy. Likewise, Washburne et al. reported a fetus with severe hydrocephalus delivered by caesarian section at 30 weeks gestation which suffered a spontaneous skull rupture at delivery [21]. In the setting of abnormally rapid growth, prenatal ultrasound may reveal polyhydramnios, cranial enlargement, and other intracranial abnormalities [22]. Fetal hydrops may also be observed and likely results from high-output cardiac failure resulting from arteriovenous shunting in a massive tumor [23]. When not diagnosed in utero, a congenital intracranial teratoma may present itself in early infancy. Sinha et al. reported a 2 month old presenting with macrocephaly, seizures and failure to thrive who found to have a large teratoma in the temporal horn of the left lateral ventricle [24]. In a case series and review of intracranial teratomas, Nanda et al. categorized congenital teratomas into 3 types: massive intracranial teratoma replacing the intracranial contents of a neonate, smaller intracranial teratomas producing hydrocephalus, and intracranial teratomas extending into the head or neck.
Conclusion
Intracranial teratomas are very rare and spontaneous rupture of intracranial mature teratomas is exceedingly rare. We report a 34 year old woman with a ruptured mature cystic teratoma of the fourth ventricle. Her long history of migraine headaches may potentially stem from what is likely chronic hydrocephalus. Additionally, her acute symptomatology just prior to presentation likely represents rupture of her teratoma. The later atypical presentation may be attributed to the tumor's unusual location, precluding the more typical visual or endocrine abnormalities which may have otherwise resulted in an earlier diagnosis.
References
-
Choi SJ, Lee JS, Song KS, Lim TH (1998) Mediastinal teratoma: CT differentiation of ruptured and unruptured tumors. AJR Am J Roentgenol 171: 591-594.
-
Madhusudhan KS, Sharma R, Gadodia A, Kumar A (2012) Spontaneous rupture of benign mediastinal teratoma: A report of two cases. Indian J Med Paediatr Oncol 33: 123-125.
-
Serraj M, Lakranbi M, Ghalimi J, Ouadnouni Y, Smahi M (2013) Mediastinal mature teratoma with complex rupture into the lung, bronchus and skin: a case report. World J Surg Oncol 11: 125.
-
Louis DN, Ohgaki H, Wiestler OD, Cavenee WK, Burger PC, et al. (2007) The 2007 WHO classification of tumours of the central nervous system. Acta Neuropathol 114: 97-109.
-
Jennings MT, Gelman R, Hochberg F (1985) Intracranial germ-cell tumors: natural history and pathogenesis. J Neurosurg 63: 155-167.
-
Desai K, Nadkarni T, Muzumdar D, Goel A (2001) Midline posterior fossa teratoma--case report. Neurol Med Chir (Tokyo) 41: 94-96.
-
Drapkin AJ, Rose WS, Pellmar MB (1987) Mature teratoma in the fourth ventricle of an adult: case report and review of the literature. Neurosurgery 21: 404-410.
-
Furtado SV, Ghosal N, Rokade VB, Hegde AS (2011) Fourth-ventricular immature teratoma. J Clin Neurosci 18: 296-298.
-
Harada K, Okamoto H, Fujioka Y, Kiya K, Mukada K, et al. (1984) Teratoma in the fourth ventricle of an elderly adult--case report. Neurol Med Chir (Tokyo) 24: 499-503.
-
Iwasaki I, Horie H, Yu TJ, Kokubo Y, Kato M, et al. (1984) Intracranial teratoma with a prominent rhabdomyogenic element and germinoma in the fourth ventricle. Neurol Med Chir (Tokyo) 24: 51-55.
-
Morelli RJ (1973) Teratoma of the fourth ventricle. Case report. J Neurosurg 38: 355-357.
-
Srinivasan US, Joseph T (1999) Intra-fourth-ventricle teratoma. Pediatr Neurosurg 30: 200-202.
-
Packer RJ, Cohen BH, Cooney K (2000) Intracranial germ cell tumors. Oncologist 5: 312-320.
-
Sano K (1999) Pathogenesis of intracranial germ cell tumors reconsidered. J Neurosurg 90: 258-264.
-
Xuzhu C, Tao J, Junmei W, Jianping D (2010) Spontaneous rupture of an intracranial teratoma. Neurol India 58: 166-167.
-
Liu Z, Lv X, Wang W, An J, Duan F, et al. (2014) Imaging characteristics of primary intracranial teratoma. Acta Radiol 55: 874-881.
-
Hashiguchi K, Inamura T, Nishio S, Nakamizo A, Inoha S, et al. (2001) Mobile intracranial oily substances from a ruptured teratoma. J Clin Neurosci 8: 567-569.
-
Scearce TA, Shaw CM, Bronstein AD, Swanson PD (1993) Intraventricular fat from a ruptured sacral dermoid cyst: clinical, radiographic, and pathological correlation. Case report. J Neurosurg 78: 666-668.
-
Oh JS, Im SB, Kim BT, Shin WH (2009) Surgical findings of a lumbar mature teratoma accompanying the preoperative intracranial dissemination of Fatty droplets. J Korean Neurosurg Soc 46:409-412.
-
Bolat F, Kayaselcuk F, Tarim E, Kilicdag E, Bal N (2008) Congenital intracranial teratoma with massive macrocephaly and skull rupture. Fetal Diagn Ther 23: 1-4.
-
Washburne JF, Magann EF, Chauhan SP, Fratkin JD, Morrison JC (1995) Massive congenital intracranial teratoma with skull rupture at delivery. Am J Obstet Gynecol 173: 226-228.
-
Lipman SP, Pretorius DH, Rumack CM, Manco-Johnson ML (1985) Fetal intracranial teratoma: US diagnosis of three cases and a review of the literature. Radiology 157: 491-494.
-
Sherer DM, Abramowicz JS, Eggers PC, Metlay LA, Sinkin RA, et al. (1993) Prenatal ultrasonographic diagnosis of intracranial teratoma and massive craniomegaly with associated high-output cardiac failure. Am J Obstet Gynecol 168: 97-99.
-
Sinha VD, Dharker SR, Pandey CL (2001) Congenital intracranial teratoma of the lateral ventricle. Neurol India 49: 170-173.





