Clinical Medical
Reviews and Case Reports
Application of Multiple Novel Imaging Modalities for Guidance and Treatment of the Patient with Subaortic Stenosis
Xiaomei Lin, Howard Axelrod, Bruno Dantas, Patrick O'Beirne, Kenneth Khaw, Edward Wrobleski, John Saia, Gerald Ukrainski, Lannae Ewing, Chad Bousanti, Lawrence J Wehner and Jingsheng Zheng*
Division of Cardiology, Atlanticare Regional Medical Center, USA
*Corresponding author:
Jingsheng Zheng, MD, PhD, FACC, Division of Cardiology, Atlanticare Regional Medical Center, 65 Jimmie Leeds Road, Pomona, NJ08205, USA, Tel: 609-561-8500, Fax: 609-567-0432, E-mail: jszheng@comcast.net
Clin Med Rev Case Rep, CMRCR-2-028, (Volume 2, Issue 4), Case Report; ISSN: 2378-3656
Received: March 05, 2015 | Accepted: April 27, 2015 | Published: April 30, 2015
Citation: Lin X, Axelrod H, Dantas B, O'Beirne P, Khaw K et al. (2015) Application of Multiple Novel Imaging Modalities for Guidance and Treatment of the Patient with Subaortic Stenosis. Clin Med Rev Case Rep 2:028. 10.23937/2378-3656/1410028
Copyright: © 2015 Lin X, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Discrete Subvalvar Aortic Stenosis (DSS) is an uncommon acquired cause of left ventricular outflow tract obstruction (LVOT) and often found during Transthoracic Echocardiography (TTE). The membranes are located adjacent to the Aortic Valve (AV) or extended to the anterior leaflet of the mitral valve. The high-velocity systolic jet collides and damages the AV leaflets making the valve more likely to fail and more susceptible to clot and vegetation formation. There is an increased risk of moderate to severe Aortic Insufficiency (AI) when the peak LVOT gradient reaches >50mmHg. The clinical course of subvalvar aortic stenosis is generally progressive with increasing obstruction and progression of AI. Surgical resection of the fibrous ridge with myomectomy is the treatment of choice.
The DSS was discovered unexpectedly in this 42 year-old white male with a past medical history of ascending aortic aneurysm, diabetes, hypertension and hyperlipidemia. He was admitted with chest pain, shortness of breath and syncope. TTE showed normal left ventricle with calcified and thickened AV. Peak LVOT gradient was 48mmHg. Due to poor acoustic window, Transesophageal Echocardiogram (TEE) with real time 3-dimensional imaging (RT-3D-TEE) was performed. TEE revealed a membranous structure located in the LVOT below a severely calcified AV. Doppler study revealed peak LVOT gradient of 57mmHg with a mean gradient of 38mmHg. Severe AI was also noted. Patient received a #25mm porcine valve with resection of the subaortic membrane and septal myomectomy. Pathology report revealed rubbery, fibromembranous tissue, consistent with DSS. Patient did well after surgery and was discharged home in 4 days. Application of multiple novel imaging modalities, such as TEE and RT-3D-TEE allowed us to better characterize the structure of the DSS and provide guidance for better surgical outcome.
Keywords
Subaortic stenosis, 2D echocardiogram, Transesophageal echocardiogram, Real-time 3-dimensional echocardiogram
Introduction
Subaortic Stenosis (SAS) is a form of LVOT obstruction, which usually presents after the first decade of life. There is unsolved speculation and controversy on its pathogenesis. A number of theories have been proposed to explain its occurrence. Congenital and acquired factors may contribute to the formation of SAS. SAS may be due to thin membrane, thick fibromuscular ridge, or diffuse tunnel-like obstruction. The discrete form of fibromuscular subvalvar aortic stenosis is the most frequent encountered (90%) [1]. The prevalence of Discrete Subaortic Stenosis (DSS) in adults with congenital heart disease is 6.5% with male predominance (2:1) [2]. DSS is often associated with other cardiac congenital malformations. The most common associated malformations include: Patent Ductus Arteriosus (PDA), Ventricular Septal Defect (VSD), Bicuspid Aortic Valve (BAV), Aortic Coarctation and Pulmonic Stenosis (PS) [3].
LVOT obstruction is often found incidentally when evaluating for systolic murmur. TTE and TEE are widely used for identifying and quantifying the severity of DSS. DSS can cause increased stricture of the LVOT below the AV. This may be enough to generate high velocity with pulsatile turbulent flow directed at the leaflets to cause fibrosis and retraction, which in turn, causes AI [4]. In one series, moderate to severe AI was present in 20% of the patients with primary operation and 60% patients with re-do operation [5].
Clinical course of SAS is generally progressive with increasing obstruction and progression of the AI. The most common symptom in patients with DSS is diminished exercise tolerance. These patients may also experience angina pectoris and syncope. Many patients are asymptomatic, even in the presence of significant LVOT gradients that indicate surgery [5]. Asymptomatic patients with a peak LVOT gradient of >64mmHg (4m/sec) are likely to develop symptoms within 5 years [6]. Surgical resection is the intervention of choice for treatment of DSS. Definitive therapy for subvalvar AS consists of surgical correction of the obstruction with excision of the fibrous ridge with septal myomectomy [7].
Case Presentation
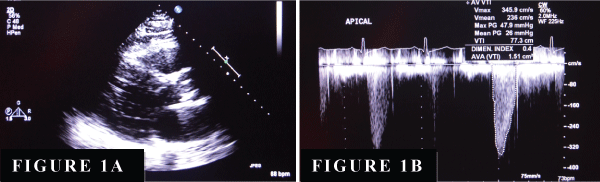
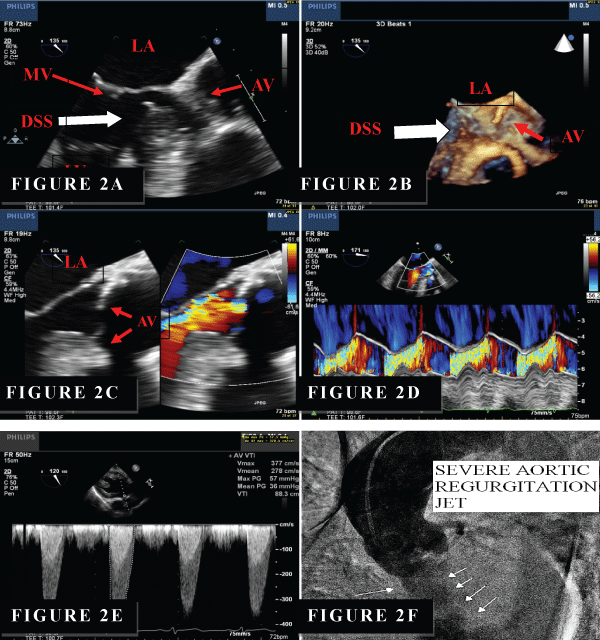
Our patient was a 42-year-old white male with past medical history of ascending aortic aneurysm, diabetes mellitus and hypertension. He presented to the hospital with a chief complaints of mild shortness of breath, chest pain and one episode of syncope. Physical examination revealed a harsh systolic murmur at the base and 3/6 diastolic murmur at the left sternal border. He underwent TTE examination. TTE revealed normal Left Ventricular Systolic Function (LVEF), AV was calcified and thickened (Figure 1A). Peak velocity for aortic valve was 3.5m/sec with peak and mean gradient of 48/26mmHg (Figure 1B). There was moderate AS with aortic valve area of 1.5cm2 and moderate AI. However, due to the poor acoustic window, TEE and RT-3D-TEE was performed. TEE revealed a fibromembranous structure located proximal to the junction of the aortic root and the intra-ventricular septum (Figure 2A). RT-3D-TEE clearly demonstrated a fibromembranous ridge located in the LVOT below the calcified aortic valve (Figure 2B). There was a significant LVOT obstruction with peak and mean trans-aortic gradient of 57/36mmHg, which was consistent with severe subaortic stenosis (Figure 3). The AV was thickened and moderately calcified but widely patent. There was severe AI by color Doppler and color M-mode (Figure 2C,2D). Cardiac catheterization (Figure 2F) revealed minimal coronary artery disease, severe AI and significant LVOT obstruction with peak and mean gradient of 42/38mmHg. No gradients were noted across the AV. There is no evidence of coarctation.
.
Figure 1: (A) Long axis view of 2D echocardiogram revealed heavily calcified AV with restrictive opening during mid-systole. (B) Doppler of aortic valve at apical view revealed peak velocity for aortic valve is 345cm/sec with peak and mean trans aortic gradient of 47/26mmHg.
View Figure 1
.
Figure 2: (A) A membranous structure (white arrow) is located beneath the AV at LOVT and with (B) real-time 3D TEE (RT-3D TEE) Note: 3D TEE was performed on the same setting as the 2D TEE using Philips iE33 xMATRIX with X7-2t probe in this study . TEE, mid-epigastric view. (C) Color comparison at 135 degree view reveals severe AI. (D) Color M-mode reveals severe AI. (E) Transgastric view of TEE: significant gradient is noted of LVOT with continuous Doppler with peak and mean gradient of 57/36mmHg. (F) Aortic root angiogram revealed severe AI and mild ascending aorta aneurysm.
View Figure 2
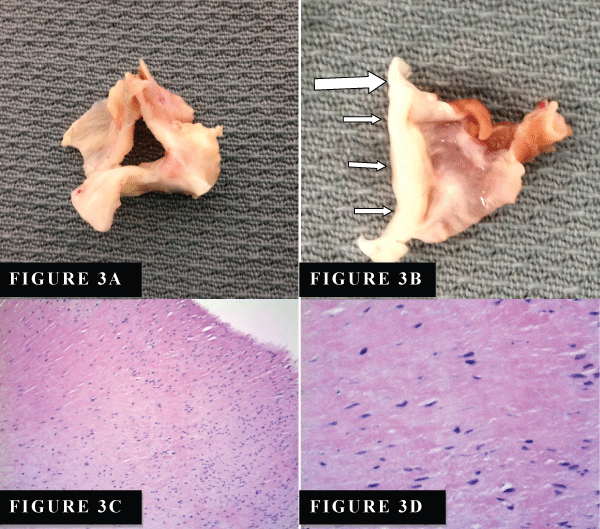
Patient underwent aortic valve replacement with #25mm porcine valve and resection of subaortic stenosis and septal myomectomy. Resected AV appeared to be heavily calcified but tricuspid (Figure 3A). The subaortic membrane is noted as the white fibrous tissues at the top of the myomectomy specimen (Figure 3B). Histological examination in hematoxylin-and-eosin revealed fibrous membranous tissue with mild to moderate chronic inflammatory infiltrates and eosinophilic fibrosis with some spindle-shape fibroblasts fibrin tissue and collagen (Figure 3 C,3D). He did well after surgery and was discharged home in 4 days.
.
Figure 3: (A) Gross photography of resection aortic valve showing significant subaortic irregular fibrous thickening. (B) Subaortic membrane (large arrow, which is corresponding to the mobile fibromuscular membrane of Figure 1 A and 1B) is white fibrous tissues (small white arrows) at the top of the myomectomy specimen. (C) Low power view of aortic valve showing fibrous thickening with mild to moderate chronic inflammatory infiltrates (Hematoxylin and Eosin stain, magnification x100). (D) High power view of aortic valve showing eosinophilic fibrosis with some spindle-shape fibroblasts (Hematoxylin &Eosin stain, magnification x400).
View Figure 3
Discussion
The case is unusual given the patient's age and advanced calcifications associated with AS ads AI. TTE revealed thickened and calcified tri-leaflet AV with poor axial 2D-imaging along the LVOT. These findings are often seen in elderly patients. Our patient presented with classic symptoms of AS. In order to fully assess the structure and develop a treatment plan, RT-3D-TEE was performed. RT-3D-TEE revealed a fibromembranous structure located proximal to the junction of the aortic root and the intra-ventricular septum causing his severe subvalvar aortic stenosis. Subvalvar aortic stenosis can be misdiagnosed as AS or hypertrophic cardiomyopathy (HOCM) with obstructive physiology. Multiple imaging modalities are often needed to distinguish DSS from AS and HOCM with obstruction.
AI is the most common acquired lesion of the aortic valve in DSS. DSS caused by abnormal LVOT architecture leads to LVOT turbulence and fibrosis of the LVOT. High velocity jets often cause trauma of aortic leaflets, which in turn cause fibrosis and retraction of the aortic leaflets. McMahon et al. [8] reported that in a series of 220 patients with subaortic stenosis, peak LVOT gradient >50mmHg was a strong predictor for at least moderate AI. However, two recent studies showed a majority of patients (58-68%) had mild AI and most of the patients did not progress over time [9]. Significant AI appeared to be more common in patients following balloon dilatation [2,7,8]. Surprisingly, valvular AS is also common finding in patients with DSS. Surgical interventions for AS occurred more frequently than interventions for AI [10].
Precise anatomical analysis of the lesions of LVOT in the DSS is challenging due to its complex nature. DSS is often associated with other congenital lesions. Multiple imaging modalities are often needed to fully characterize DSS. Cardiac catheterization provides hemodynamic information, such as LVOT gradient, especially when multiple levels of obstruction are suspected. It is also used to evaluate the degree of AI and other associated lesions, such as VSD, PDA, aortic aneurysm and coarctation. Continuous Wave (CW) Doppler is often used to evaluate the LVOT gradient and to assess the degree of the obstruction across the LVOT. The measurement of the maximal instantaneous gradient by CW Doppler is often higher than the peak-to-peak gradient from the cardiac catheterization. Novel imaging modalities, such as TEE and RT-3D-TEE may provide more detailed and important information about this complicated disease and may provide guidance during the operation and follow-up.
Cardiovascular Magnetic Resonance Imaging (CMR) has emerged as a novel 3- dimensional tomographic imaging modality, providing images of the heart at high spatial and temporal resolution. It has no ionizing radiation. In selected cases where echocardiographic images are suboptimal, CMR is very helpful in providing detailed morphological information regarding LVOT obstruction, particularly in patients with multi-level LVOT obstruction (such as mitral stenosis, BAV, aortic aneurysm and coarctation) [11]. Cardiac Multi Detector Computed Tomography (CT) and ECG-gated CT angiography provide novel opportunity for evaluation of cardiac morphology and function, LVOT obstruction as well as the coronary arteries [12,13]. In comparison with CMR, multidetector CT has higher spatial resolution. It provides high-quality multiplanar reconstructions in any desired image orientation. It is very useful in those patients who need to exclude coronary artery disease or in those patients who have contraindications for CMR, such as pacemaker or defibrillator.
Definitive treatment for DSS with significant LVOT obstruction is surgical resection and myomectomy. For patients with diffuse narrow LVOT, a Konno procedure involving incision of the ventricular septum and patch augmentation of LVOT may be needed. However, optimal timing for surgery remains debatable. Early aggressive approach should be weighed against significant incidence of recurrence of DSS. The rate of recurrent obstruction can be as high as 26%- 33% [9,14]. In this large multi-center study, median follow-up is 12.9 years. All patients had adequate relief of LVOT obstruction. Mean gradient reduced from 76mmHg preoperatively to 15mmHg postoperatively. There is a 1.3mmHg increase in LVOT gradient postoperative per year. However, if surgery was performed when the peak LVOT gradient of was < 40mmHg, the rate of reoperation was significantly lower (0.9 vs 6.5% per year in patients with higher gradient) [15]. Geva et al. [16] reported that if the lesion is very close to the AV (< 6mm), or peak LVOT gradient >60mmHg or there is intraoperative peeling of the membrane from the aortic or mitral valve, these are associated with higher incidence of reoperation. The presence of an immediate postoperative gradient of >10mmHg also predicts recurrent DSS in 75% of the patients [1]. Progressive AI may also develop despite resection of DSS. Persistent turbulent flow after surgical resection of DSS can still cause the damage of the aortic valve. Brauner et al. [15] reported that a higher preoperative LVOT gradient predicted progression of the AI in the later course. It is recommended [7] that when the peak LVOT gradient is >50mmHg, patients should undergo surgical intervention. Surgery should be also considered in patients with low LVOT gradient (< 50mmHg) if the patient has left ventricular dysfunction, moderate to severe AI or VSD. Asymptomatic patients who plan to become pregnant or wishing to participate in competitive sports should be considered for surgical resection if peak LVOT gradient is >30mmHg. Surgical treatment consists of resection of the DSS and septal myomectomy. Surgical mortality is low and complications are usually minimal.
Infective endocarditis occurs in 12% of the patients with SAS [17]. Incidence of endocarditis was significant reduced after surgery when LVOT gradient < 80mmHg. Antibiotics prophylaxis for infective endocarditis is no longer recommended in patient with SAS, except patient with previous history of infective endocarditis or with a repair prosthetic material or device. In the later cases, antibiotic prophylaxis is recommended for the first 6 months after repair unless a residual defect is present.
In the present study, novel imaging modalities, such as TEE, RT-3D-TEE and digital subtracted angiography were used to provide comprehensive anatomical and functional assessment of malformed AV and to guide surgical intervention. The patient was done well after resection of the discrete subaortic membrane and myomectomy.
Conclusion
DSS is a rare cause of LVOT obstruction that can mimic AS and HOCM. It should be suspected in young patients with AS. TTE has been the initial imaging study performed for evaluation of AS. If clinical presentation suggests other possible causes for LVOT obstruction, TEE and RT-3D-TEE should be used to obtain comprehensive anatomical and functional assessment of the LVOT and AV. Treatment for DSS is usually surgical. The common approach is resection of discrete membrane with resection of the septal myocardium.
References
-
Aboulhosn J, Child JS (2006) Left ventricular outflow obstruction: subaortic stenosis, bicuspid aortic valve, supravalvar aortic stenosis, and coarctation of the aorta. Circulation 114: 2412-2422.
-
Oliver JM, Gonz�lez A, Gallego P, S�nchez-Recalde A, Benito F, et al. (2001) Discrete subaortic stenosis in adults: increased prevalence and slow rate of progression of the obstruction and aortic regurgitation. J Am Coll Cardiol 38: 835-842.
-
Leichter DA, Sullivan I, Gersony WM (1989) "Acquired" discrete subvalvular aortic stenosis: natural history and hemodynamics. J Am Coll Cardiol 14: 1539-1544.
-
Barkhordarian R, Uemura H, Rigby ML, Sethia B, Shore D, et al. (2007) A retrospective review in 50 patients with subaortic stenosis and intact ventricular septum: 5-year surgical experience. Interact Cardiovasc Thorac Surg 6: 35-38.
-
Darcin OT, Yagdi T, Atay Y, Engin C, Levent E, et al. (2003) Discrete subaortic stenosis: surgical outcomes and follow-up results. Tex Heart Inst J 30: 286-292.
-
Pellikka PA, Sarano ME, Nishimura RA, Malouf JF, Bailey KR, et al. Outcome of 622 adults with asymptomatic, hemodynamically significant aortic stenosis during prolonged follow-up. Circulation 111: 3290-3295.
-
Gersony WM (2001) Natural history of discrete subvalvar aortic stenosis: management implications. J Am Coll Cardiol 38: 843-845.
-
McMahon CJ, Gauvreau K, Edwards JC, Geva T (2004) Risk factors for aortic valve dysfunction in children with discrete subvalvar aortic stenosis. Am J Cardiol 94: 459-464.
-
van der Linde D, Takkenberg JJ, Rizopoulos D, Heuvelman HJ, Budts W, et al. (2013) Natural history of discrete subaortic stenosisin adults: a multicentre study. Eur Heart J 34: 1548-1556.
-
Laksman ZW, Silversides CK, Sedlak T, Samman AM, Williams WG, et al. (2011) Valvular aortic stenosis as a major sequelae in patients with pre-existing subaortic stenosis changing spectrum of outcomes. J Am Coll Cardiol 58: 962-965.
-
Nagueh SF, Bierig M, Budoff MJ, Desai M, Dilsizian V, et al. (2011) American society of echocardiography clinical recommendations for multimodality cardiovascular imaging of patients with hypertrophic cardiomyopathy. J Am Soc Echocardiogr 24: 472-498.
-
Chun EJ, Choi SI, Jin KN, Kwag HJ, Kim YJ, et al. (2010) Hypertrophic cardiomyopathy: assessment with MR imaging and multidetector CT. Radiographics 30: 1309-1328.
-
Halpern EJ, Gupta S, Halpern DJ, Wiener DH, Owen AN (2012) Characterization and normal measurements of the left ventricular outflow tract by ECG-gated cardiac CT: implications for disorders of the outflow tract and aortic valve. Acad Radiol 19: 1252-1259.
-
Brown DW, Keane JF (2013) Subvalvar aortic stenosis. In: Up To Date Kim MS (Ed) Waltham, MA.
-
Brauner R, Laks H, Drinkwater DC Jr, Shvarts O, Eghbali K, et al. (1997) Benefits of early surgical repair in fixed subaortic stenosis. J Am Coll Cardiol 30: 1835-1842.
-
Geva A, McMahon CJ, Gauvreau K, Mohammed L, del Nido PJ, et al. (2007) Risk factors for reoperation after repair of discrete subaortic stenosis in children. J Am Coll Cardiol 50: 1498-1504.
-
Wright GB, Keane JF, Nadas AS, Bernhard WF, Castaneda AR (1983) Fixed subaortic stenosis in the young: medical and surgical course in 83 patients. Am J Cardiol 52: 830-835.





