Clinical Medical Reviews and Case Reports
Monitoring of the Left Recurrent Laryngeal Nerve during Mediastinoscopy is Feasible and Safe
Wolfram Karenovics1*, Sebastien Guigard1, Besa Zenelaj1, Marc Licker2 and Federic Triponez1
1Thoracic and Endocrine Surgery Unit, University Hospitals of Geneva, Switzerland
2Anaesthesiology Department, University Hospitals of Geneva, Switzerland
*Corresponding author: Wolfram Karenovics, Thoracic and Endocrine Surgery, University Hospitals of Geneva, Rue Gabrielle-Perret-Gentil 4, 1211 Geneva, Switzerland, Tel: +41223727873; Fax: +41223727880; E-mail: Wolfram.Karenovics@hcuge.ch
Clin Med Rev Case Rep, CMRCR-1-012, (Volume 1, Issue 2), Research Article; ISSN: 2378-3656
Received: November 25, 2014 | Accepted: December 26, 2014 | Published: December 29, 2014
Citation: Karenovics W, Guigard S, Zenelaj B, Licker M, Triponez F (2014) Monitoring of the Left Recurrent Laryngeal Nerve during Mediastinoscopy is Feasible and Safe. Clin Med Rev Case Rep 1:012. 10.23937/2378-3656/1410012
Copyright: © 2014 Karenovics W, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Objective: Left Recurrent Laryngeal Nerve (RLN) palsy is a well known complication of cervical mediastinoscopy and is not infrequent if specifically looked for. Electro-physiological monitoring of the RLN is common practice in thyroid surgery and has greatly improved outcomes. We applied the same technique during cervical video-mediastinoscopy.
Patients and methods: Between October 2012 and October 2013 patients undergoing mediastinoscopy were enrolled prospectively for intra-operative monitoring of the left RLN. A standardized protocol for the use of the neuromonitor was followed. At least 3 lymph node stations were systematically sampled in all patients including station 4L in all cases.
Results: Data were obtained from 12 patients (8 males); median age was 60 years. Indication for mediastinoscopy was staging for lung cancer in 6 patients and biopsy of undiagnosed mediastinal lesions in the remaining 6 patients. The RLN could be clearly identified and a good signal on the neuromonitor was obtained at the beginning and at the end of the operation in all cases. No abnormal trajectory of the RLN was noted. There was no case of postoperative hoarseness.
Conclusion: Intraoperative electrophysiological monitoring to identify the left RLN was feasible and safe in the 12 patients analysed in this study. Whether the use of the neuromonitor can reduce the rate of recurrent laryngeal nerve palsies remains to be shown.
Keywords
Mediastinoscopy, Neuromonitoring, Recurrent laryngeal nerve
Introduction
Since the advent of EBUS and EUS the frequency of cervical mediastinoscopy has greatly diminished. It is, however, still an important tool in the staging for non small cell lung cancer (NSCLC) and in the diagnosis of mediastinal lymphadenopathies and other mediastinal lesions. Damage to the left Recurrent Laryngeal Nerve (RLN) during mediastinoscopy is a well known complication although the literature on its frequency is quite scarce and the mechanism not always well understood. Electrophysiologic monitoring of the RLN has proven to be a valuable aid in thyroid and parathyroid surgery where it has gained widespread acceptance. The same technology can be used during cervical mediastinoscopy to help identifying with certainty the left RLN and, hopefully, to help preserving it.
The aim of this preliminary study was to evaluate the feasibility and safety of using intra-operative neuromonitoring during mediastinoscopy to identify the left RLN and to report pitfalls and difficulties in using this technique.
Methods
We used the Medtronic NIM-Response 3.0� intraoperative nerve monitoring system during cervical video-mediastinoscopy to monitor the left RLN. This requires special endotracheal tubes with electrodes above the tracheal cuff facing the vocal cords (NIM� EMG Tube). We used the same protocol that we described for thyroid surgery [1]: General intravenous anaesthesia was mainly based on short-acting hypnotics (propofol) and analgesics (sufentanyl) with a single dose of myorelaxant allowing recovery of neuromuscular function within 15-20 minutes after tracheal intubation. Importantly, mechanical lung ventilation without additional myorelaxants was facilitated by a satisfactory level of general anaesthesia as monitored by the bispectral index of electrical brain activity. Correct function of the system, including correct positioning of the endotracheal tube was confirmed by an initial test on the left vagus nerve with a stimulant current of 1mA. The left recurrent nerve was then tested as soon as it was discovered in the left paratracheal region (Figure 1,2). It was tested again at the end of the operation and finally the left vagus nerve was tested again to confirm the integrity of the nerve in its entire length. The strength of the signal in micro Volts (�V) at the beginning and the end of the operation were compared. The final signal was considered good if it had similar strength as the initial signal and was above the threshold of 175�V. The neuromonitoring was not used systematically in all patients undergoing a mediastinoscopy during the study period. Reasons for not using the neuromonitoring were unavailability of the neuromonitoring device or cases where there was no need to go in a region close to the left RLN. There were also 3 cases with bulky tumours, where dissection and visualisation of the left recurrent nerve was not possible. These were not included.
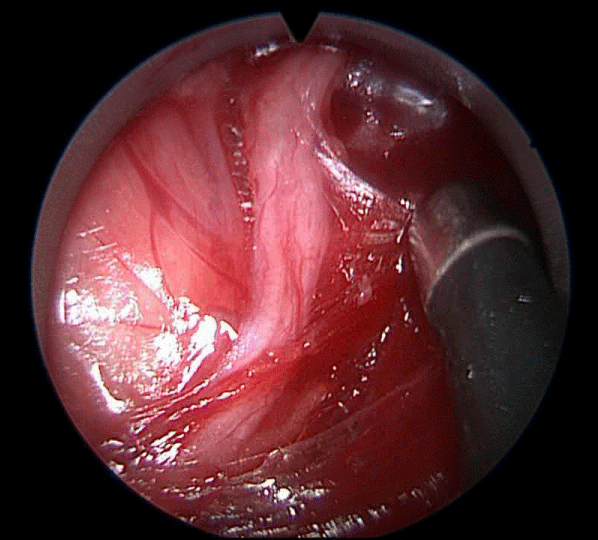
Figure 1: Visualisation of the left RLN
View Figure 1
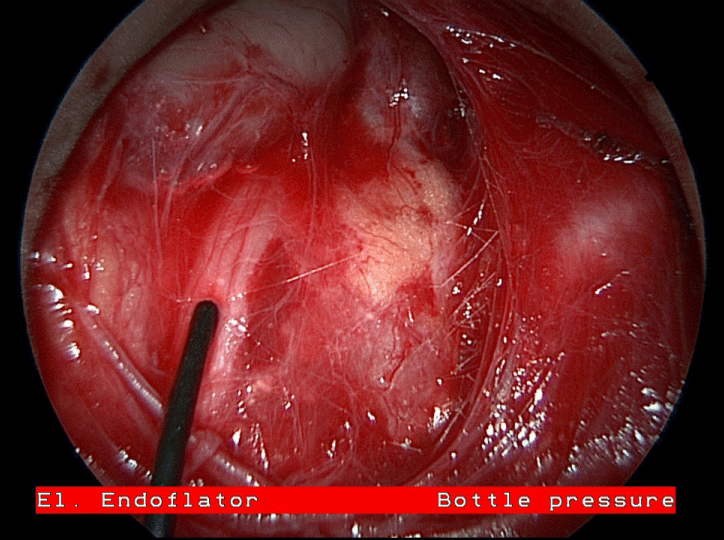 Figure 2:The stimulater probe on the RLN. The trachea is visible on the right side of the picture. In the top left corner a station 4L lymph node
View Figure 2
Figure 2:The stimulater probe on the RLN. The trachea is visible on the right side of the picture. In the top left corner a station 4L lymph node
View Figure 2
Results
Between October 2012 and October 2013 we prospectively included 12 patients on whom we performed a video-mediastinoscopy with intraoperative nerve monitoring. Median age was 60 years (range: 39 � 78 years); 8 patients were males. Indications for mediastinoscopy were staging for NSCLC in 6 patients, suspected lymphoma in 3 patients, suspected sarcoidosis in 1 and mediastinal lymphadenopathies of unknown origin in 2 patients (Table 1). The left RLN could be clearly identified in the region of the 4L station. We obtained a good signal from the neuromonitor on the left RLN and the left vagus nerve at the end of the intervention in all patients. No abnormal position or trajectory was identified in this small group. The position of the left RLN was, in the contrary, very predictable and stable. We noted, however, that it could run above or below the more lateral station 4 nodes. The vagus nerve was identified and tested between left common carotid artery and left internal jugular vein at the height of the neck incision. This part of the procedure was challenging as the nerve is very deep and lateral and the incision small, sometimes making visualisation difficult. In all patients, biopsy or lymphadenectomy were performed at least in 3 lymph node stations, including 4L in all cases. There was no case of postoperative hoarseness or vocal cord palsy. Histology showed no mediastinal nodal metastasis in 3 cases, presence of mediastinal involvement in 3 cases, granulomatous inflammation in three patients, lymphoma in one patient, tuberculosis in one patient and reactive lymph nodes in one patient.
![]()
Table 1: Indications and histopathology
View Table 1
Discussion
Cervical mediastinoscopy is the gold standard for diagnosis and staging of mediastinal metastases in patients with NSCLC. It is also an important tool for the diagnosis of non-NSCLC mediastinal lymphadenopathies and tumours. It has a false negative rate between 2 and 9%; sensitivity and specificity are 78 to 94 and 100% respectively [2] for the staging of lung cancer. Since its beginnings in 1959, the complication rate has reportedly been low, generally less than 3 % [3-8]. Complications include bleeding from central greater vessels (azygos vein, superior vena cava, innominate vein and artery, pulmonary artery, aorta) tracheal and oesophageal lesions, infection, pneumothorax and vocal cord palsy [9,10]. This latter complication is in most series less than 1% in some, even large series, even 0%. It is probably underreported or under diagnosed. If specifically looked for with a postoperative laryngoscopy, incidence of vocal cord palsy may increase to 6 % [11]. A recent report by Walles et al showed a similar rate of vocal cord palsy or hoarseness of about 6.5 % [12]. Approximately the same rate, i.e. 5.5% of prolonged hoarseness of the voice, was confirmed by Chabowski et al in their series of 54 consecutive patients [13].
The reported occurrence of recurrent laryngeal nerve palsy in thyroid surgery is 2 to 3 times higher when this complication is specifically looked for, either by routine post-operative laryngoscopy or by systematic use of neuromonitoring of the RLN [1]. The main reason for this is that the dysphonia can be very subtle or even absent in many patients with vocal cord palsy if the paralysed vocal cord is in a median or paramedian position. Paralysis of the RLN can lead to dysphonia which can greatly affect professional or recreational activities. However and perhaps more importantly in thoracic surgical patients, it can lead to bronchoaspiration and to inefficient cough. The vocal cords do not close properly during deglutition and do not protect the airways. In these patients, Valsalva manoeuvre is not possible and the efficacy of the cough to clear the airways is reduced. Prevention of this underrecognized complication is therefore important.
In our small series, none of the patients developed postoperative hoarseness or vocal cord palsy. This was, however, not verified by laryngoscopy. In the presence of a good nerve signal on the vagus nerve at the end of the operation, this did not seem necessary or useful. It has been shown that the neuromonitoring reliably predicts postoperative function [14,15] and the risk of postoperative RLN palsy is less than 0.1% - 1% [1,16-18] if there is a good signal. On the other hand, not in all cases of loss or marked diminution of the signal will there be a RLN palsy. It is, however, extremely important to follow a standardized and validated protocol to avoid potential errors in the utilization of the neuromonitoring system leading to erroneous results [19]. Errors may result from surgical technique (quality of nerve exposure etc.), problems related to technical aspects of the neuromonitor (strength of current, event threshold, electrode placement etc.), equipment malfunction, as well as from problems on the anesthesia front, like tube size and placement, type of anaesthesia, secretions around the electrodes etc. Communication with the anesthesiologist is therefore crucial. Intravenous anaesthesia and mechanical ventilation without additional myorelaxant agents facilitated the operating conditions and allowed us to apply correctly the nerve monitoring. Technically the most challenging part was the stimulation of the vagus nerve through the low and small neck incision. Clear visual identification of the vagus nerve was not always possible. A good signal could still be obtained by stimulating deeply between left common carotid artery and internal jugular vein after gentle dissection.
The use of mediastinoscopy has been decreasing steadily over the last few years [20]. This is due mainly to the increased use of endoscopic ultra-sound guided fine needle aspiration (EBUS and EUS). These techniques are highly reliable with sensitivities and specificities of about 92% and 100% respectively [21]. They also compare favourably with respect to invasiveness, patient comfort and morbidity [22]. It therefore seems highly likely that they will gain further territory and mediastinoscopy become even less frequent. For young surgeons, it is thus becoming more and more difficult to learn the technique of mediastinoscopy and to feel comfortable with a rarely performed procedure. On the other hand, for the time being, mediastinoscopy is still necessary in order to clarify the status of lymph nodes in lung cancer if there is discordance between PET and EBUS [2,23] as well as in the diagnosis of lymphoma [22], where cytology is often not sufficient and in the diagnosis of other mediastinal lesions. The neuromonitoring helps to clearly identify the left recurrent nerve and to ascertain its functional integrity thus facilitating confident removal or biopsy of lymph nodes in its proximity. Obviously this helps to prevent direct injury to the nerve but would not prevent damage by other mechanisms like traction as described by Roberts et al. [6]. We did not encounter this problem in our small series, however, and obtained a good signal on the vagus nerve at the end of the procedure in all cases. We also feel that the nerve in this region is very free and mobile unless it is encased by tumour, making it less prone to traction injury than closer to its entry in the larynx where it is relatively fixed, as in thyroid surgery. We also regularly observe during thyroid surgery that, for instance, cephalad traction on the thyroid while working on its inferior poles generates increased activity on the neuromonitor and may cause coughing. This seems to be related to patient arousal (no muscle relaxation) in response to the increased stimulation caused by this traction and is by no means predictive of recurrent laryngeal nerve injury. That said, it is possible that blunt finger dissection may cause harm to the left RLN especially when pushing the aorta forward [24].
Many institutions currently use a neuromonitoring system for thyroid procedures, so the device and know-how is available. The cost of disposable material (endotracheal tube and stimulation probe) is variable from one country to another and from one institution to another. In a study by Dionigi et al. on thyroid surgery in Italy, the added cost of neuromonitoring was evaluated at 215.5 Euros in institutions with a high volume and 278.8 Euros in institutions with low volume. The cost of the non-disposable equipment is also included in this analysis [25].
In conclusion, we demonstrate in this preliminary data that it is feasible and safe to identify the left recurrent laryngeal nerve intraoperatively. Further studies are needed to examine whether the use of neuromonitoring of the recurrent laryngeal nerve during mediastinoscopy reduces the rate of recurrent laryngeal nerve palsy.
References
-
Sadowski SM, Soardo P, Leuchter I, Robert JH, Triponez F (2013) Systematic use of recurrent laryngeal nerve neuromonitoring changes the operative strategy in planned bilateral thyroidectomy. Thyroid 23: 329-333.
-
Detterbeck FC, Lewis SZ, Diekemper R, Addrizzo-Harris D, Alberts WM (2013) Executive Summary: Diagnosis and management of lung cancer, 3rd ed: American College of Chest Physicians evidence-based clinical practice guidelines. Chest 143: 7S-37S.
-
Carlens E, Hambraeus GM (1967) Mediastinoscopy. Indications and limitations. Scand J Respir Dis 48: 1-10.
-
Karfis EA, Roustanis E, Beis J, Kakadellis J (2008) Video-assisted cervical mediastinoscopy: our seven-year experience. Interact Cardiovasc Thorac Surg 7: 1015-1018.
-
Kiser AC, Detterbeck FC (2011) General aspects of surgical treatment. In: Detterbeck FC, Rivera MP, Socinski MA, editors. Diagnosis and Treatment of Lung Cancer: An Evidence-based Guide for the Practicing Clinician. W.B. Saunders Company: 133-147.
-
Roberts JR, Wadsworth J (2007) Recurrent laryngeal nerve monitoring during mediastinoscopy: predictors of injury. Ann Thorac Surg 83: 388-391.
-
Venissac N, Alifano M, Mouroux J (2003) Video-assisted mediastinoscopy: experience from 240 consecutive cases. Ann Thorac Surg 76: 208-212.
-
Hammoud ZT, Anderson RC, Meyers BF, Guthrie TJ, Roper CL, et al. (1999) The current role of mediastinoscopy in the evaluation of thoracic disease. J Thorac Cardiovasc Surg 118: 894-899.
-
Lemaire A, Nikolic I, Petersen T, Haney JC, Toloza EM, et al. (2006) Nine-year single center experience with cervical mediastinoscopy: complications and false negative rate. Ann Thorac Surg 82: 1185-1189.
-
Pearson FG (1993) Staging of the mediastinum. Role of mediastinoscopy and computed tomography. Chest 103: 346S-348S.
-
Widstr�m A (1975) Palsy of the recurrent nerve following mediastinoscopy. Chest 67: 365-366.
-
Walles T, Friedel G, Stegherr T, Steger V (2013) Learning mediastinoscopy: the need for education, experience and modern techniques--interdependency of the applied technique and surgeon's training level. Interactive Cardiovasc Thorac Surg 16: 450-454.
-
Chabowski M, Szymanska-Chabowska A, Skotarczak J, Janczak D Jr, Pawlowski L, et al. (2014) The Role of Mediastinoscopy in the Diagnosis of Thoracic Disease: One-Year Single Center Experience. Adv Exp Med Biol .
-
Pardal-Refoyo JL, Cuello-Azcarate JJ, Ochoa-Sangrador C (2013) Contribution of neuromonitoring to the safety of tracheal extubation after total thyroidectomy. Prospective study with needle electrodes. Revista espanola de anestesiologia y reanimacion 60: 563-670.
-
Cernea CR, Brand�o LG, Hojaij FC, De Carlucci D Jr, Brand�o J, et al. (2012) Negative and positive predictive values of nerve monitoring in thyroidectomy. Head Neck 34: 175-179.
-
Dralle H, Sekulla C, Haerting J, Timmermann W, Neumann HJ, et al. (2004) Risk factors of paralysis and functional outcome after recurrent laryngeal nerve monitoring in thyroid surgery. Surgery 136: 1310-1322.
-
Barczynski M, Konturek A, Cichon S (2009) Randomized clinical trial of visualization versus neuromonitoring of recurrent laryngeal nerves during thyroidectomy. Br J Surg 96: 240-246.
-
Dionigi G, Boni L, Rovera F, Rausei S, Castelnuovo P, et al. (2010) Postoperative laryngoscopy in thyroid surgery: proper timing to detect recurrent laryngeal nerve injury. Langenbeck's Arch 395: 327-331.
-
Randolph GW, Dralle H, International Intraoperative Monitoring Study G, Abdullah H, Barczynski M, et al. (2011) Electrophysiologic recurrent laryngeal nerve monitoring during thyroid and parathyroid surgery: international standards guideline statement. Laryngoscope 121: S1-S16.
-
Vyas KS1, Davenport DL, Ferraris VA, Saha SP (2013) Mediastinoscopy: trends and practice patterns in the United States. South Med J 106: 539-544.
-
Chandra S, Nehra M, Agarwal D, Mohan A (2012) Diagnostic accuracy of endobronchial ultrasound-guided transbronchial needle biopsy in mediastinal lymphadenopathy: a systematic review and meta-analysis. Respir Care 57: 384-391.
-
Varela-Lema L, Fernandez-Villar A, Ruano-Ravina A (2009) Effectiveness and safety of endobronchial ultrasound-transbronchial needle aspiration: a systematic review. Eur Respir J 33: 1156-1164.
-
De Leyn P, Dooms C, Kuzdzal J, Lardinois D, Passlick B, et al. (2014) Revised ESTS guidelines for preoperative mediastinal lymph node staging for non-small-cell lung cancer. European Journal of Cardio-Thoracic Surgery 45: 787-798.
-
Benouaich V, Marcheix B, Carfagna L, Brouchet L, Guitard J (2009) Anatomical bases of left recurrent nerve lesions during mediastinoscopy. Surg Radiol Anat 31: 295-299.
-
Dionigi G, Bacuzzi A, Boni L, Rausei S, Rovera F, et al. (2012) Visualization versus neuromonitoring of recurrent laryngeal nerves during thyroidectomy: what about the costs? World J Surg 36: 748-754.





