Clinical Medical Reviews and Case Reports
Ocular, Nasal and Aural Myiasis in an Intoxicated Patient: A Case Report
Giorgio Berlot* and Cristina Calderan
Department of Anesthesia and Intensive Care, University of Trieste, Italy
*Corresponding author: Prof. Giorgio Berlot, Department of Anesthesia and Intensive Care, University of Trieste, Cattinara Hospital, Strada di Fiume 447, 34149 Trieste, Italy, Tel: +390403994540, Fax: +39040912278, E-mail: berlot@inwind.it
Clin Med Rev Case Rep, CMRCR-4-156, (Volume 4, Issue 2), Case Report; ISSN: 2378-3656
Received: January 10, 2017 | Accepted: February 22, 2017 | Published: February 25, 2017
Citation: Berlot G, Calderan C (2017) Ocular, Nasal and Aural Myiasis in an Intoxicated Patient: A Case Report. Clin Med Rev Case Rep 4:156. 10.23937/2378-3656/1410156
Copyright: © 2017 Berlot G, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
The authors describe a case of myiasis occurred in a self-intoxicated patient lying outdoor in a reduced state of consciousness for more than two days. The causes and the favoring circumstances of the infestation are described and discussed.
Keywords
Myiasis, Self-intoxication, Lucilia sericata
Introduction
The word Myiasis indicates the infestation of alive vertebrates, including humans, by larvae of different species of Diptera [1,2]. Its occurrence is facilitated either by environmental factors, such as warm and wet climates, poor hygiene and water sanitation and by host-related factors, including skin wounds, infections, burns and tumors. Independently from the involved species, the initial step of infestation consists in the deposition of several hundreds of eggs by each mature female followed by the maturation of the larvae until they fall on the ground and pupate. According to their biology, larvae can be subdivided in (a) obligate parasites, which need a host for their development; (b) facultative parasites, which do not need a host for their entire life-cycle; and (c) accidental parasites, which are accidentally ingested but not killed in the digestive tract [3]. In each case the larvae feed on mammalian dead or living tissues and fluids and can cause different pathologic conditions according to the location, the interaction with the host and the possible bacterial contamination [2].
In Western Europe myiasis occurs mainly in subjects with soil-contaminated wounds, infected or necrotic skin areas or returning from regions where it is endemic.
Here we report the case of a patient who suffered from nasal, ocular and aural myiasis occurred during a more-than 2-days long outdoor exposition after the ingestion of benzodiazepines and paroxetin.
Case Description
A 55-year-old man with a history of major depression was admitted to the Emergency Department (ED) due to the ingestion of an undetermined amount of benzodiazepines and paroxetin occurred in the preceding two days. In the early afternoon of 5/27/2016 the patient did not return home from work and in the evening of the same day a Search and Rescue operation was launched. On 5/29/2016 at 8 PM, he was found lying in a wood of the hills surrounding the city of Trieste, in North-Eastern Italy; in this time interval the temperature and the air humidity ranged from 10 °C to 21 °C and from 66% to 72%, respectively [4]. The rescuers discovered several empty packages of diazepam and paroxetin aside the patient, who was sleepy but arousable and wore thick sweater and trousers. The patient's exposed surface and clothes were covered by a huge number of larvae of flies and ticks and he was brought to the Emergency Department wrapped in a thermal blanket. At admission, the patient was fully alert and the body temperature (BT) was 34.2 °C; other vital signs and the blood chemistries were normal. At the examination, many maggots were observed escaping from his eyelids, ears and nostrils (Figure 1) and crawling on his trunk; an erythema was present over his chest. The patient was actively warmed with heated intravenous fluids and the antitetanic prophylaxis was provided; the ticks and the larvae were removed and his eyes, ears and nostrils were flushed with warm water till the complete disappearance of the parasites; an antibiotic eye lotion was applied on his eyes. Once in the Intensive Care Unit (ICU), the patient was given antibiotic for the possible skin infection and for the prophylaxis of Lyme disease; a bronchoscopy showed that larvae were not present in the upper airways; he was transferred to the Department of Infectious Disease two days after the ICU admission. According to an entomologist, the larvae belonged to the Lucilia sericata species, of the Calliphoridae family, which is also known as green bottle blowfly.
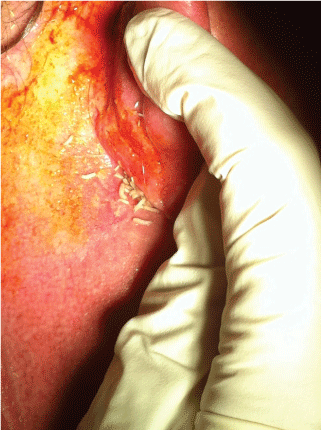
.
Figure 1: Larvae crawling behind the right ear.The picture has been taken 12 hours after the ICU admission.
View Figure 1
Discussion
Although myiasis is not limited to developing countries, in Western Europe it is uncommon in patients free of risk factors such as skin wounds, dermatologic infections and/or tumors [5]; as a matter of fact, the infestation by larvae of Calliphorae, including those belonging to Lucilia sericata species, is extremely uncommon in living healthy humans without the above mentioned predisposing conditions: actually, according to a recent review of published cases, in Italy almost all (10/11) cases of myiasis due to Calliphoridae and Sarchophagidae families have been described in subjects with preexisting skin lesions which were absent in our patient [6]. Then, it is likely that some other interacting circumstances account for its occurrence in this otherwise healthy patient.
First, the infestation has been facilitated either by the long period of immobility caused by the ingested drugs and by the low BT mimicking the presence of a cadaver and the facial natural orifices constituted the only possible access for the egg deposition due to the clothes covering the rest of the body: a similar mechanism has been described in patients admitted to the ICU in a hospital following mouse infestation whose carcasses constituted the primary site of parasites maturation [7]. As far as the thoracic erythema is concerned, it can be ascribed to the many bacterial species carried by the larvae spreading downward from the head, because the thick garments worn by the patient likely hampered their penetration [8].
Second, the persisting reduction of consciousness prevented the patient from perceiving the symptoms associated with the different sites of infestations, including tinnitus, ocular and ear pain and the sensation of foreign body: in the only described case of external ocular myiasis caused by Lucilia Sericata occurred without predisposing factors [9] as well in other cases of adult aural myiasis the patients complained of extremely disturbing symptoms [10,11]. The same neurological conditions impeded the self-removal of the larvae, whose number was so elevated that an awake patient could not have overlooked them.
Finally, the environmental factors likely played a role because the relatively mild air temperature as well as the elevated humidity constituted an ideal setting for the initial infestation and subsequent maturation of the maggots. The same applies for ticks, which are endemic in the areas where the patient was found.
Conclusions
In developed countries, the infestation of living humans by Diptera larvae is associated with the presence of favoring factors, including wounds, skin infections or tumors. In the described case, in whom these predisposing circumstances were absent, different host and environmental factors likely contributed to its occurrence, including the prolonged reduction of consciousness, the decreased BT and the relatively mild air temperature, which favored the egg deposition and the development of the larvae in the facial orifices.
A more and more people are involved in open-air and in wilderness leisure activities, myiasis should be suspected in patients lacking self-protection capabilities exposed to a prolonged outdoor permanence.
References
-
Francesconi F, Lupi O (2012) Myiasis. Clin Microbiol Rev 25: 79-105.
-
Singh A, Singh Z (2015) Incidence of myiasis among humans-a review. Parasitol Res 114: 3183-3199.
-
Mc Graw TA, Turianski (2008) Cutaneous Myiasis. J Am Acad Dermatol 58: 907-926.
-
www.ilmeteo.it
-
Sesterhenn AM, Pfützner W, Braulke DM, Wiegand S, Werner S, et al. (2009) Cutaneous manifestations of myiasis in malignant wounds of the head and the neck. Eur J Dermatol 19: 64-68.
-
Zammarchi, Giorni A, Gabrielli S, Strohmeyer M, Cancrini G, et al. (2014) Human oestriasis acquired in Florence and review of human myiasis in Italy. Parasitol Res 113: 2379-2385.
-
Beckendorf R, Klotz SA, Hinkle N, Bartholomew W (2002) Nasal myiasis in an intensive care unit linked to hospital-wide mouse infestation. Arch Intern med 162: 638-640.
-
Villwock JA, Harris TM (2014) Head and neck myiasis, cutaneous malignancy, and infections: a case series and review of the literature. J Emerg Med 47: e37-e41.
-
Choi W, Kim GA, Park SH, Shin SE, Park JH, et al. (2015) First report of external ophtalmomyiasis caused by Lucilia sericata Meigen in a healthy patientwithout predisposing risk factors. Parasitol Int 64: 281-283.
-
Hatten K, Gulleth Y, Mejer T, Eisenman DJ (2010) Myiasis of the external and middle ear. Ann Otol Rhinol Laryngol 119: 436-438.
-
Magliulo G, Gagliardi M, D'Amico R (2000) Human aural myiasis. Otolaryngol Head Neck Surg 122: 777.





