Clinical Medical
Reviews and Case Reports
Acute Disseminated Encephalomyelitis (ADEM) Secondary to Neurosyphilis
Ryan Kahn1*#, Christopher Dermarkarian1#, Elizabeth Crocco2, Gabrielle Yeaney3 and Marcia Williams4
1Miller School of Medicine, University of Miami, USA
2Department of Psychiatry & Behavioral Sciences, University of Miami, USA
3Division of Neuropathology, RT Pathology & Laboratory Medicine Institute, USA
4Miami Veterans Affairs Healthcare System, USA
#These authors contributed equally to the manuscript
*Corresponding author: Ryan Kahn, Miller School of Medicine, University of Miami, 55 SE 6th St. #2607 Miami, FL 33131, USA, Tel: 954-558-6376, E-mail: RKahn16@med.miami.edu
Clin Med Rev Case Rep, CMRCR-3-143, (Volume 3, Issue 11), Review Article; ISSN: 2378-3656
Received: September 24, 2016 | Accepted: November 23, 2016 | Published: November 25, 2016
Citation: Kahn R, Dermarkarian C, Crocco E, Yeaney G, Williams M (2016) Acute Disseminated Encephalomyelitis (ADEM) Secondary to Neurosyphilis. Clin Med Rev Case Rep 3:143. 10.23937/2378-3656/1410143
Copyright: © 2016 Kahn R, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Acute disseminated encephalomyelitis (ADEM) is a rare autoimmune demyelinating disease of the central nervous system. For our case presentation, a 52-year-old Hispanic male presented with altered mental status and focal neurological defects. MRI showed multiple lesions scattered throughout the brain. VDRL and FTA-ABS was positive in the CSF. Brain biopsy demonstrated foamy macrophages, loss of myelin, and perivascular T cells. The diagnosis was acute disseminated encephalomyelitis secondary to neurosyphilis. This is the only case reported in the literature of neurosyphilis as an inciting event of ADEM, and it should be included in the differential diagnosis of this uncommon condition.
Keywords
Acute disseminated encephalomyelitis, ADEM, Neurosyphilis, Demyelination
Case Report
A 52-year-old Hispanic male mechanic with a past medical history of type II diabetes mellitus presented to our institution with altered mental status. The patient had been in his normal state of health until 3 weeks before admission. At that time, the patient began to experience confusion, disorientation, gait imbalance, and dysphasia. His condition deteriorated daily to where he was no longer able to complete activities of daily living and developed severe focal neurologic deficits with labile mood fluctuations.
Past medical history was positive for diabetes mellitus, controlled on metformin. Family history was positive for multiple sclerosis in his mother. Social history was non-contributory. Upon admission, the patient was afebrile and hemodynamically stable. Physical exam findings showed neurological deficits and mood abnormalities. Routine labs and electrolytes were within normal limits.
Computed tomography without contrast of the brain ordered shortly after arrival showed seven different regions of vasogenic edema throughout the bilateral cerebral hemispheres in the right insula, right parietal, bilateral frontal, and occipital lobes. CT also demonstrated a right basal ganglion lesion measuring 2.1 × 1.9 cm, a right posterior parietal lobe lesion measuring 3.9 × 3.7 cm, a focus in the left front lobe measuring approximately 3.2 × 2.9 cm. Magnetic resonance imaging of the brain with and without contrast showed restricted diffusion in the wall of one lesion with an “advancing front” appearance (Figure 1A and Figure 1B); Large parietal T1 hypo intense lesion with no mass effect (Figure 1C and Figure 1D); Incomplete rim enhancement in the larger lesions and diffuse enhancement in the smaller lesions (Figure 1E and Figure 1F); and an incomplete dark T2 rim present along the medial border of one lesion (Figure 1G and Figure 1H). All lesions were non-ring enhancing. MRI of the spine was attempted; however, the patient remained agitated throughout the study and results were unobtainable.
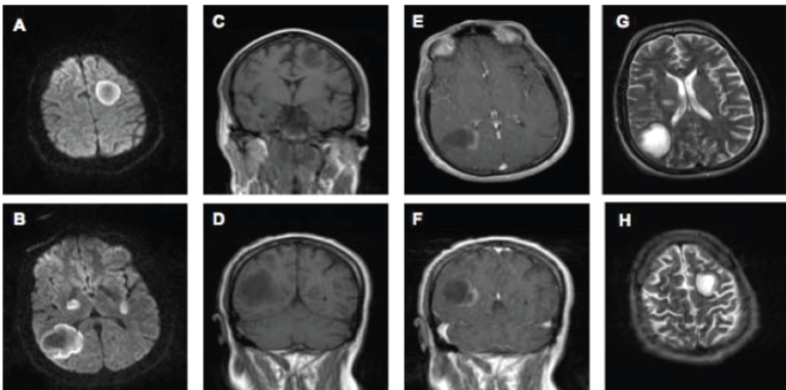
.
Figure 1: Brain MRI with and without contrast.
A,B) Diffusion: Multiple lesions bilaterally demonstrate restricted diffusion in the wall of the lesion with an "advancing front" appearance; C,D) T1 Pre-Contrast: Large parietal T1 hypointense lesion with no mass effect; E,F) T1 Post-Contrast: Incomplete rim enhancement in the larger lesions and diffuse enhancement in the smaller lesions seen; G,H) T2-Dark Rim: Incomplete dark T2 rim is present along the medical border of the larger lesion.
View Figure 1
Given the patient's findings, the differential diagnosis at the time was metastatic masses secondary to malignancy, toxoplasmosis, fungal or bacterial abscess, CNS lymphoma, cryptococcus, or a demyelinating disease. Tests for JC virus, PML, and HIV were all found negative. The lack of extraparenchymal disease and the lack of central diffusion on MRI imaging made metastasis, lymphoma, and abscess less likely. A demyelinating disease was more likely given the incomplete rim of diffusion and enhancement of the lesions as well as the lack of peri-lesional edema and mass effect.
Cerebral spinal fluid from the lumbar puncture showed elevated protein (66 mg/dl) with normal WBC level, and oligoclonal bands with an elevated IGG index (7.91), which was suggestive of a demyelinating disease. A stereotactic brain biopsy showed diffuse foamy macrophages with only rare small CD20-positive B cells and some perivascular small T cells by immunohistochemistry for CD3 (Figure 2A and Figure 2D). Luxol fast blue-H&E showed abundant foamy macrophages and confirmed myelin loss (Figure 2B). Given these findings, the histopathologic differential diagnosis included ADEM, multiple sclerosis, and subacute infarct (destructive myelinolysis); however, relative axonal preservation shown on neurofilament staining favored an active demyelinative process (Figure 2C). The stain for spirochetes in brain biopsy was negative.
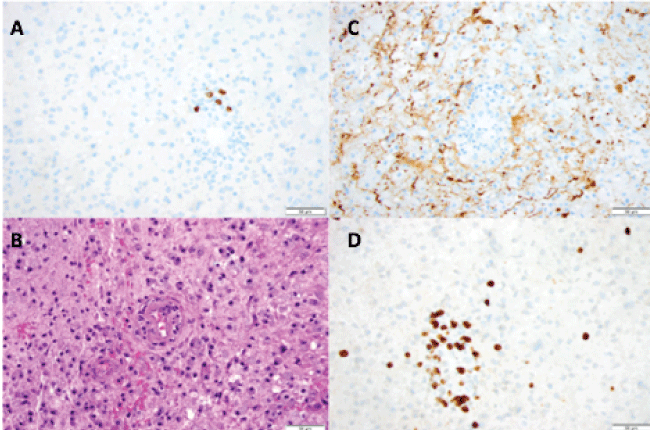
.
Figure 2: Brain biopsy with immunohistochemistry.
A,D) Stereotactic brain biopsy showed diffuse foamy macrophages with only rare small CD20-posive B cells and some perivascular small T cells by immunohistochemistry for CD3; B) Luxol fast-blue H&E shows abundant foamy macrophages and confirms myelin loss; C) Relative axonal preservation shown on neurofilament staining favors active demyelinative process.
View Figure 2
The pathology of perivascular infiltration of lymphocytes, abundant macrophages, and myelin loss is suggestive of a demyelinating disease. Malignancy, toxoplasmosis, fungal or bacterial abscess, CNS lymphoma, cryptococcus, subacute infarct were ruled out by CSF studies, MR image assessment, and stereotactic brain biopsy. The clinical diagnosis of acute disseminated encephomyelitis (ADEM) [1], a rare autoimmune demyelinating disorder usually triggered secondary to an infectious process, was made based on the patient's acute clinical presentation, a lack of history of previously unexplained neurologic symptoms, his neuroimaging, CSF and histopathological findings.
A CSF sample was found reactive for both venereal Disease Research Laboratory test (VDRL) and Fluorescent treponemal antibody absorption test (FTA-ABS), which is diagnostic for neurosyphilis. A family member provided collateral information that the patient was diagnosed with syphilis many years prior and did not seek treatment. The evidence is consistent with a neurosyphilis para-infectious process acting as a triggering factor for the encephalopathy.
The final diagnosis was acute disseminated encephomyelitis (ADEM) secondary to neurosyphilis.
The patient was started on a five-day regimen of high dose methylprednisolone and a 10-day regimen of Penicillin G. On hospital day 3, the patient began to show signs of neurocognitive improvement. The patient's cognitive status-as measured by the Montreal Cognitive Assessment (MOCA), Word-Reading Test (WRAT-4), and the Repeatable Battery for the Assessment of Neuropsychological status (RBANS)-showed objective improvement. His cognition, problem solving skills, and linguistic abilities have significantly improved over time but remained below baseline.
Discussion
Acute Disseminated Encephalomyelitis (ADEM) is a rare disease with an estimated annual incidence rate of 8 per 1,000,000 [2]. It presents as an autoimmune demyelinating disease of the central nervous system triggered by a recent infectious process in most but not all cases [3]. ADEM typically presents with brain edema, perivenous infiltration of lymphocytes, macrophages, or neutrophils, and perivenous swelling with demyelination.
Patients typically present with a rapid deterioration of symptoms including altered mental status, motor deficits, ataxia, nystagmus, seizures, and headaches. It is proposed to occur due to myelin autoantigens (myelin basic protein, proteolipid protein, and myelin oligodendrocyte protein) sharing antigens with those of an inciting pathogen. These infectious antibodies cross react with myelin autoantigens and result in diffuse encephalomyelitis [4,5].
Metastatic disease, toxoplasmosis, fungal or bacterial abscess, CNS lymphoma, and cryptococcus were initially ruled out in this patient. It was then hypothesized that either multiple sclerosis or ADEM were the cause of the patient's clinical presentation. To distinguish patients with ADEM from those with multiple sclerosis, previous investigators have proposed that a patient must present with two of the following three findings: 1. Presence of symptoms (ex: confusion) that are atypical for multiple sclerosis, 2. Gray matter involvement on brain MRI and 3. Absence of oligoclonal bands in the CSF [3].
ADEM usually produces a widespread CNS disturbance, similar to our patient's presentation, while multiple sclerosis is typically monosymptomatic [3]. Likewise, our patient presented with confusion, which is atypical in the presentation of MS. On neuroimaging, lesions secondary to ADEM typically present in greater number than multiple sclerosis, have poor definitions, and are more likely to involve gray matter such as the basal ganglia [3,6]. Such a pattern was evident in our patient's neuroimaging studies. CSF studies in our patient demonstrated the presence of oligoclonal bands and an elevated IgG index; however, these findings can be seen in multiple sclerosis as well as ADEM [3,6]. Likewise, his rapid improvement with steroid therapy is not typical of tumefactive MS, which would be the subtype of MS associated with these lesions. Given the clinical presentation, laboratory as well as pathological findings, and neuroimaging, the final diagnosis is most likely ADEM secondary to a neurosyphilis triggering event.
To our knowledge, this is the first reported case of ADEM occurring secondary to neurosyphilis as an inciting event. Therefore, neurosyphilis should be included in the differential diagnosis of this uncommon condition, as a timely high-dose corticosteroid regimen has been shown to improve clinical outcomes and reduce the recurrence risk [7]. Additionally, initiating corticosteroids in later stages of the disease is believed to be significantly less effective [7].
Acknowledgements
The authors of this paper would like to thank the division of neuropathology at the Cleveland Clinic for providing the histopathological imaging seen in this case report.
References
-
Menge T, Hemmer B, Nessler S, Wiendl H, Neuhaus O, et al. (2005) Acute disseminated encephalomyelitis: an update. Arch Neurol 62: 1673-1680.
-
Leake JA, Albani S, Kao AS, Senac MO, Billman GF, et al. (2004) Acute disseminated encephalomyelitis in childhood: epidemiologic, clinical and laboratory features. Pediatr Infect Dis J 23: 756-764.
-
de Seze J, Debouverie M, Zephir H, Lebrun C, Blanc F, et al. (2007) Acute fulminant demyelinating disease: a descriptive study of 60 patients. Arch Neurol 64: 1426-1432.
-
Poser CM (1969) Disseminad vasculomyelinopathy. A review of the clinical and pathologic reactions of the nervous system in hyperergic diseases. Acta Neurol Scand 37: 3-44.
-
Reik L Jr (1980) Disseminated vasculomyelinopathy: an immune complex disease. Ann Neurol 7: 291-296.
-
Schwarz S, Mohr A, Knauth M, Wildemann B, Storch-Hagenlocher B (2007) Acute disseminated encephalomyelitis: a follow-up study of 40 adult patients. Neurology 56: 1313-1318.
-
Alexander M, Murthy JM (2011) Acute disseminated encephalomyelitis: treatment guidelines. Ann Indian Acad Neuro 14: S60-64.





