Clinical Medical
Reviews and Case Reports
Recovery of an Under-Deployed Renal Artery Stent Responsible for a Complication of Renal Artery Stenosis
Husain Alturkistani1,2, Youri Kaitoukov1, Patrick Gilbert1 and Vincent Oliva1*
1Department of Radiology, Division of Interventional Radiology, Hôpital Notre-Dame, Canada
2Radiology Department, King Saud University, Saudi Arabia
*Corresponding author: Vincent Oliva, Department of Radiology, Division of Interventional Radiology, Hôpital Notre-Dame, Centre hospitalier de l'Université de Montréal (CHUM), 1560, rue Sherbrooke Est, Montréal (Québec), H2L 4M1, Canada, Tel: +1 514 890-8000, E-mail: voliva@sympatico.ca
Clin Med Rev Case Rep, CMRCR-3-123, (Volume 3, Issue 8), Case Report; ISSN: 2378-3656
Received: February 29, 2016 | Accepted: August 04, 2016 | Published: August 08, 2016
Citation: Alturkistani H, Kaitoukov Y, Gilbert P, Oliva V (2016) Recovery of an Under-Deployed Renal Artery Stent Responsible for a Complication of Renal Artery Stenosis. Clin Med Rev Case Rep 3:123. 10.23937/2378-3656/1410123
Copyright: © 2016 Alturkistani H, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
A 74-year-old male patient presented to our emergency room (ER) with recurring episodes of flash pulmonary edema (dyspnea worsened by lying down) and hypertension resistant to medical treatment. He had a history of left nephrectomy for renal cell carcinoma (RCC) and right renal artery angioplasty. We hereby report a case of a complication of renal artery stenosis in a patient with a single kidney secondary to an under-deployed renal artery stent that was retrieved.
Keywords
Stenosis, Stent, Intravascular, Retrieval, Foreign body, Resistant hypertension, Pulmonary edema
Case Report
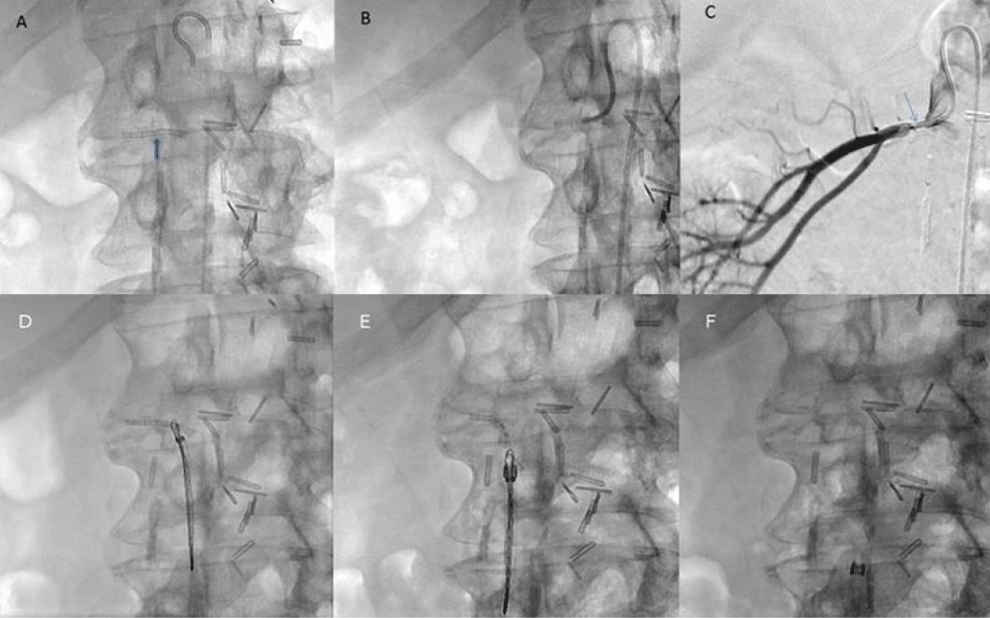
A 74-year-old male patient with a past history of left nephrectomy for renal cell carcinoma and right renal artery angioplasty was referred from another hospital to our ER. Patient suffered numerous bouts of pulmonary edema which came on suddenly and resolved rapidly. Between these bouts, he was asymptomatic, and his only physical abnormality was the persistently elevated blood pressure despite the renal artery angioplasty and medical treatment. There was no other medical problem. On admission, the chest X-ray showed pulmonary edema, while Doppler ultrasound revealed a decreased resistive index in the hilum of the kidney with accelerations and turbulent flow in the middle portion of the right renal artery. Doppler US found severe stenosis in the right renal artery estimated at 80%. The patient was taken to the angiography suite for renal angiography +/- angioplasty. After a right femoral artery puncture, a diagnostic aortic angiography was done with a 4-French (4 Fr) catheter to gain a better understanding of the clinical picture. A foreign body was visualized at the origin of the renal artery. A radiographic one shot image was taken and an under-deployed stent was visualized (Figure 1A). A selective renal arteriography was done with a Chuang catheter at the origin of the right renal artery to confirm the diagnosis: severe stenosis at the origin of the right renal artery secondary to a foreign body (Figure 1B and Figure 1C). The renal artery was partly occluded by a collapsed stent protruding into the aortic lumen.
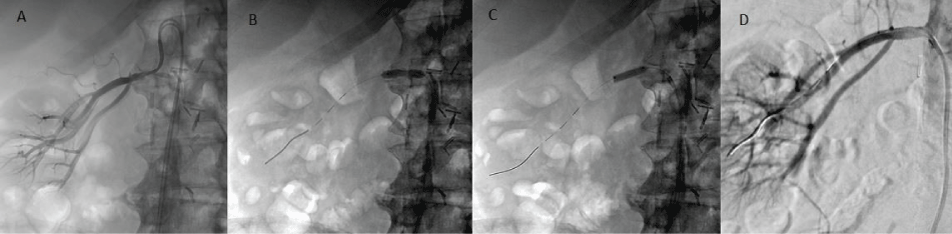
After 3000 IU of heparin was given, a 7Fr RDC guiding catheter was inserted. The proximal free edge of the stent protruding into the aorta was snared (EN Snare, Merit Medical Systems, Inc.) (Figure 1D). Finally, the stent was successfully removed by gentle traction (Figure 1E and Figure 1F). The arteriography post stent removal showed residual renal artery stenosis (Figure 2A). An angioplasty was done with a 5 mm balloon using a Thruway 0.014 inch microguide wire (Figure 2B). A 6 mm × 18 mm monorail pre mounted stent (Express SD, Boston Scientific) was installed (Figure 2C). The final arteriography control showed a patent renal artery without any residual stenosis (Figure 2D). The procedure was complicated by a CFA pseudoaneurysm treated with local US-guided injection of 300 IU of thrombin (RECOTHROM, ZymoGenetics).
.
Figure 1: A) An intra-aortic pigtail catheter (4 Fr). Under-deployed stent (indicated by the blue arrow); B) Catheter at the origin of the right renal artery; C) Contrast injection via Chuang catheter (5Fr) shows severe stenosis (blue arrow) at the origin of the artery while the rest of artery is patent; D,E) Removal of the stent with (EN Snare); F) Image post-removal of under-deployed stent.
View Figure 1
.
Figure 2: A) Arteriography post under-deployed stent removal shows residual renal artery stenosis; B) Angioplasty with a 5 mm balloon was performed using a Thruway microguide; C) Placement of a 6 mm × 18 mm monorail pre mounted stent (Express SD, Boston Scientific); D) Final control arteriography shows a patent renal artery without any residual stenosis.
View Figure 2
Discussion
With the evolving branch of vascular interventional radiology and the increasing range of endovascular therapies and widespread use of guide wires, catheters, stents, embolization materials (such as coils and plugs), IVC filters, etc., loss of instruments (foreign bodies) is a frequently encountered clinical problem [1]. Intravascular foreign bodies pose a risk of distal migration, causing thrombosis, cardiopulmonary and peripheral embolism. In addition to these medical challenges, there are limited options and techniques for their retrieval, which can sometimes be time-consuming, especially after long retention in the blood vessels [2].
Intravascular foreign body retrieval techniques and instruments have evolved tremendously over the last two decades, from an open surgical approach to an endovascular approach [2]. Sandusky described the earliest report in the literature in 1918 of the removal of a migratory intravascular foreign body (FB) from the heart [3], while the earliest percutaneous removal of a foreign body was accomplished by Thomas et al. when they retrieved, through the saphenous vein, a broken wire from the inferior vena cava and right atrium with bronchoscopic forceps in 1964 [4]. In 1968, Ranniger developed the first specialized instrument for the retrieval of intravascular foreign bodies, marking the beginning of the era of endovascular retrieval instrument development [5]. Nowadays, an endovascular percutaneous approach is accepted as the first-line method for removing intravascular FBs [1].
Loop snares are frequently considered the device of choice for FB retrieval [6]. Modern designs allow loops to emerge at a 90° angle to the catheter. Many techniques that use snares exist, such as the proximal or distal grab technique, the coaxial snare technique and the lateral grasp technique [1]. Other devices for FB retrieval include stone retrieval baskets/Dormia baskets used mainly for the biliary system but which could also be used for the endovascular system [6,7]. The small balloon catheter technique can be used for the retrieval of lost stents [8]. FBs have been successfully retrieved using only guide wire when it is used as a snare [9], among other guide wire techniques. Other devices include intravascular retrieval forceps such as Alligator forceps, which should be used judiciously, as they have higher iatrogenic risks [10].
Our case was unique in that the high blood pressure (BP) was not corrected despite the first attempt of renal artery stenosis angioplasty and medical treatment. BP level was 170/100 at time of presentation with filtration glomerular rate at 45 ml/min. The bouts of pulmonary adema which our patient suffered from led to further investigation and referral to our medical institution. The patient's only kidney had to be saved, as he had undergone a prior left nephrectomy. The placement of a renal artery stent was not documented in the patient's history from the referring hospital. There was only a history of right renal angioplasty with no stent mentioned in the chart. After the diagnostic arteriography we found that there is a patent arterial lumen between previous non dilated stent and the renal artery which justified our theory. That's why an attempt to retrieve the old stent was performed and a new stent was placed with a good angiographic result.
Learning Points
1. A clear reporting after every interventional procedure is necessary for patient follow-up.
2. Although cardiogenic pulmonary edemais one of the main differential diagnoses of recurrent pulmonary edema in the elderly, renal artery stenosis or migrating coronary stent should also be considered in a relevant history, in particular if accompanied by hypertension resistant to medical treatment.
References
-
Woodhouse JB, Uberoi R (2013) Techniques for intravascular foreign body retrieval. Cardiovasc Intervent Radiol 36: 888-897.
-
Mousa AY, Gill G, Aburahma AF (2014) New trick for removal of intravascular retained foreign body: a case report and review of literature. Vasc Endovascular Surg 48: 55-57.
-
Sandusky WR (1951) The removal of a migratory intravascular foreign body from the heart; report of a case. Va Med Mon (1918) 78: 64-70.
-
Thomas J, Sinclair-Smith B, Bloomfield D, Davachi A (1964) Non-surgical retrieval of a broken segment of steel spring guide from the right atrium and inferior vena cava. Circulation 30: 106-108.
-
Ranniger K (1968) An instrument for retrieval of intravascular foreign bodies. Radiology Nov 91: 1043-1044.
-
Foster-Smith KW, Garratt KN, Higano ST, Holmes DR Jr (1993) Retrieval techniques for managing flexible intracoronary stent misplacement. Cathet Cardiovasc Diagn 30: 63-68.
-
Guimaraes M, Denton CE, Uflacker R, Schonholz C, Selby B Jr (2012) Percutaneous retrieval of an Amplatzer septal occluder device that had migrated to the aortic arch. Cardiovasc Intervent Radiol 35: 430-433.
-
Cishek MB, Laslett L, Gershony G (1995) Balloon catheter retrieval of dislodged coronary artery stents: a novel technique. Cathet Cardiovasc Diagn 34: 350-352
-
Lee CY (2011) Use of wire as a snare for endovascular retrieval of displaced or stretched coils: rescue from a technical complication. Neuroradiology 53: 31-35.
-
Eeckhout E, Stauffer JC, Goy JJ (1993) Retrieval of a migrated coronary stent by means of an alligator forceps catheter. Cathet Cardiovasc Diagn 30: 166-168.





