Clinical Medical
Reviews and Case Reports
Acute Diarrhea Secondary to Pseudomembranous Colitis in the Adulthood, a Diagnostic Challenge: Case Report
Freddy A. Rodríguez-Hernández1, Giselle Blanco-Lamont1, Cinthia Patricia Vargas-Chávez1, Maria Valeria Jiménez-Báez2*, Cristian Alejandro Aguirre-Alva1, Luis Sandoval-Jurado2 and Luis A. Medina-Andrade3
1Regional General Hospital No. 17, Mexican Social Security Institute, Division of Health Sciences, University of Quintana Roo. Cancún, Quintana Roo, Mexico
2Coordination of Health Research, Mexican Social Security Institute, Delegation Quintana Roo, Division of Health Sciences, University of Quintana Roo, Mexico
3General Surgery Department, General Hospital No. 30, Mexico
*Corresponding author: Maria Valeria Jiménez Báez, Research Health Coordinator, Division of Health Sciences, University of Quintana Roo, Av. Politécnico Nacional s/n, Mexico, Tel: 052-9981-698299, E-mail: Valeria.jimenezb@gmail.com
Clin Med Rev Case Rep, CMRCR-3-114, (Volume 3, Issue 6), Case Report; ISSN: 2378-3656
Received: April 21, 2016 | Accepted: June 28, 2016 | Published: June 30, 2016
Citation: Hernández FAR, Lamont GB, Chávez CPV, Alva CAA, Andrade LAM, et al. (2016) Acute Diarrhea Secondary to Pseudomembranous Colitis in the Adulthood, a Diagnostic Challenge: Case Report. Clin Med Rev Case Rep 3:114. 10.23937/2378-3656/1410114
Copyright: © 2016 Hernández FAR, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Introduction
Clostridium difficile is the main cause of nosocomial diarrhea in the USA with about 450,000 cases annually [1]. It is a gram positive spore-forming bacillus, toxin-producing, related to the use of broad-spectrum antibiotics, prolonged hospital stay, and elderly (> 65 years) [2,3]. Clinical signs include acute diarrhea, severe, dehydration and systemic toxicity secondary to toxins [2,3].
Diagnostic is based on clinical signs, laboratory findings with toxin A and/or B in stool and colonic pseudomembranes identified by colonoscopy, this last with a sensibility and specificity of 97% [4]. If it’s not timely suspected toxic megacolon, septic shock and death, in 5-12%, could be inevitable without correct management [5].
Case Report
An 84 years-old female presented at emergency room with abdominal pain and diarrhea. Pathological background include arterial hypertension treated with losartan 100 mg/24h, cardiac arrhythmia treated with propafenona 75 mg/24h, osteoarthritis treated with non-steroidal anti-inflammatories (COX-2) and chronic diarrhea for the last three months treated with quinolones.
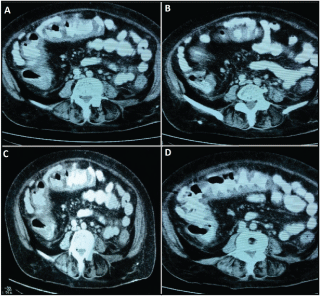
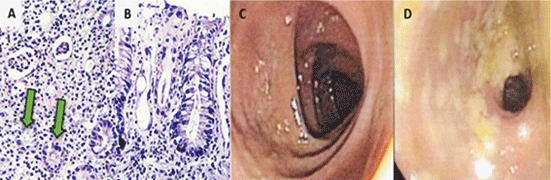
It begins in December 15, 2015 with acute diarrhea, treated initially with ciprofloxacin and metronidazole for 7 days with good outcomes; second episode begins in January 1st 2016 with acute diarrhea treated with Cephotaxime 1 g/8 h for 7 days with diarrhea remission. In January 16, 2016 diarrhea begins with 8 to 10 depositions daily, liquid stool with mucus, blood and abdominal bloating, reason why she came to emergency room, and after 7 days treated with 3rd generation cephalosporin she didn’t present clinical improvement. Laboratories report Hemoglobin 11 g/dl, hematocrit 25%, and leucocytes 18.000 neutrophils 79%. Glucose 100 mg/dL, urea 20 mg/dL, creatinine 0.5 mg/dL, Na: 139 mEq/L, Ca: 7.5 mEq/L, Mg: 2.1 mEq/L, K: 3.2 mEq/L, P: 2.5 mEq/L, Cl: 112 mEq/L. Serial stool test (3 samples) reported blood, mucus, leucocytes, without macroscopic parasites. Computed Tomography scan reported thickness of colonic wall associated with peripheral fat edema secondary to an inflammatory process like colitis (Figure 1). Colonoscopy reported descending and sigmoid colon with mucosal edema, congestion and multiple erosions covered by withe fibrin. Biopsy during the last procedure reported chronic and acute colitis, ischemic colitis, moderate atrophy of colonic mucosa with edema and congestion (Figure 2). With background and the reported studies the diagnosis of Pseudomembranous colitis is done. After 10 days of treatment with vancomycin 250 mg orally every 6 hours diarrhea stops, patient present clinical improvement without sepsis signs after three days of treatment. After these patients was discharged and had not presented recurrence until May 2016.
.
Figure 1: A) y, B) Adcendent and transverse colon thickening; C) y; D) Identifiable acordion sign (concentring colonic mucosa thickening with pericolic fat inflammation).
View Figure 1
.
Figure 2: A) Transverse colon mucosa with diffuse lymphocytic inflammatory infiltrate type, ulceration and infiltration PMN in glandular epithelium; atrophic crypts, sheet own surrounded by hyalinized (ischemic colitis; green arrows), HE 100x. B) Sigmoid colon mucosa with inflammatory infiltrate of lymphocytic type, glandular atrophy and alteration in the pattern mucoprotectant. C-D) Descending and sigmoid colon and mucosal erosions normal vasculature and covered with fibrin.
View Figure 2
Discussion
Clostridium difficile infection incidence is higher in patients over 65 years old, as in the present case (84 years old), with previous use of quinolones as a risk factor, favoring decrease in the normal colonic flora, with a mortality about 5-12% in severe cases [6,7].
The last diarrhea episode present the classic symptomatology of this pathogen, including the presence of 3 or more unformed stools in 24 hours or fewer consecutive hours 1-8, with mucus and blood, and other studies helping to suspect the diagnosis like colonoscopy and stool test [2,4,6,7].
Direct stool cytotoxin with tissue culture and anaerobic culture for toxygenic C. Difficile detects B toxin, and have a sensibility of 80 and > 90% respectively, and specificity of 97% but take more than 48 hours [4,8,9]. Enzyme immunoasay and Glutamate dehydrogenase detect toxin A or B in the first and antigen in the second, with sensibility between 70-80% but a results available in a couple of hours as advantage. The better laboratory test is the quantitative real time polymerase chain reaction (qPCR), detecting the gen for toxin B, with sensibility > 97%, specificity > 97% and results in hours [7]. Unfortunately in our hospital we don’t have this laboratory test and for this reason why proceed to para-clinics including computed tomography, colonoscopy and patient clinical signs, to begin a therapeutic antibiotic probe with vancomycin [4,8,9].
Computed tomography has a sensibility of 70-80% and specificity of 50-80% with four basic criteria for C. Difficile infection diagnosis including: pericolonic wall fat > 4 mm, pericolonic fat thickening, colonic wall modularity and acordion sign (10-20% of severe colitis). Colonoscopy could report hyperemic mucosa, white or yellow plates from 2 to 21 mm and anatomo-pathologic exam reports ulcerated mucosa with purulent exudate, mucous glands pattern and surface content pseudo membranes [8].
It have been reported that metronidazole is less effective than in the past, and is not indicated in repetitive C. Difficile infection, with vancomycin 125-250 mg orally every 6 hours. In our hospital we only have intravenous vancomycin 1 g/10 mL, and we used 1 g diluted in 250 cc of water, giving a quarter of the dose every 6 hours with good results, allowing this use in similar hospitals without the ideal presentation [10,11].
In advanced age patients with acute diarrhea and colonic pseudomembranes the presence of C. difficile infection must be considered as the causative agent and, in case of confirmation by the mentioned laboratory test or clinical high suspicion, begin the antibiotic therapeutic probe.
C. difficile infection stills a diagnostic and some times therapeutic challenge, by lack of the entire laboratory test available in all medical centers and for metronidazole being less effective. The presented case allows us to confirm the use of intravenous presentation of vancomycin being effective as we prepared the drug, a new option in hospitals without the access to oral presentation.
Acknowledgements
Dr. Juan Manuel Álvarez Reyes, Pathology Department, HGR #17 Instituto Mexicano del Seguro Social, Cancún, Quintana Roo.
References
-
Remes-Troche JM (2012) Diarrea asociada con infección por Clostridium difficile, ¿es tiempo de preocuparnos en México? Rev Gastroenterol Mex 77: 58-59.
-
Blanco A, Ruiz O, Otero W, Gómez M (2013) Infección por Clostridium difficile en ancianos. Rev Col Gastroenterol 28: 53-63.
-
Zea J.W, Salazar CL (2012) Enfermedad asociada a Clostridium difficile: prevalencia y diagnóstico por laboratorio. Infectio 16: 211-212.
-
Rodríguez D, Mireleis B, Navarro F (2013) Infecciones producidas por Clostridium difficile. Enferm Infecc Microbiol Clin 31: 254-263.
-
Asensio A, Monge D (2012) Epidemiología de la infección por Clostridium difficile en España. Enferm Infecc Microbiol Clin 30: 333-337.
-
Ramírez-Rosales A, Cantú-Llanos E (2012) Mortalidad intrahospitalaria en pacientes con diarrea asociada a infección por Clostridium difficile. Rev Gastroenterol Mex 77: 60-65.
-
Lance R. Peterson, Ari Robicksen (2009) Does My Patient Have Clostridium difficile Infection? Ann Intern Med 151: 176-179.
-
Berrutti D, Limongi G, Cancela M (2012) Colitis por Clostridium difficile. Arch Med Interna 34: 17-23.
-
Stuart S. Cohen, Gerding D, Johnson S, Kelly C, et al. (2010) Clinical Practice Guidelines for Clostridium difficile Infection in Adults: 2010 Update by the Society for Healthcare Epidemiology of America (SHEA) and the Infectious Diseases Society of America (IDSA). Chicago Journals 31: 431-455.
-
Emily G. McDonald, Tood C. Lee (2015) Clostridium difficile Infection. N Engl J Med 373: 286-288.
-
Susan M. Novak-Weekley, Elizabeth M. Marlowe, John M. Miller, Joven Cumpio, et al. (2010) Clostridium difficile Testing in the Clinical Laboratory by Use of Multiple Testing Algorithms. J Clin Microbiol 48: 889-893.





