Clinical Medical
Reviews and Case Reports
Left Atrial Appendage Occluder with Recurrent Thrombus Formation Visualized by Three-Dimensional Transesophageal Echocardiography
Sebastian Greiner*, Nicolas Geis, Derliz Mereles, Hugo A. Katus, and Stefan Hardt
Department of Cardiology, Angiology and Pneumology, University of Heidelberg, Germany
*Corresponding author: Sebastian Greiner, MD, Department of Cardiology, Angiology and Pneumology, University Hospital Heidelberg, Im Neuenheimer Feld 410, 69120 Heidelberg, Germany, Tel: +49-6221-568852, Fax: +49-6221-567436, E-mail: sebastian.greiner@med.uni-heidelberg.de
Clin Med Rev Case Rep, CMRCR-3-108, (Volume 3, Issue 5), Case Report; ISSN: 2378-3656
Received: April 14, 2016 | Accepted: May 28, 2016 | Published: May 31, 2016
Citation: Greiner S, Geis N, Mereles D, Katus HA, S Hardt (2016) Left Atrial Appendage Occluder with Recurrent Thrombus Formation Visualized by Three-Dimensional Transesophageal Echocardiography. Clin Med Rev Case Rep 3:108. 10.23937/2378-3656/1410108
Copyright: © 2016 Greiner S, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
A 78-year-old Caucasian female with newly diagnosed atrial fibrillation was put on anticoagulation with phenprocoumon. Two months later she developed an intracranial hemorrhage localized in the brainstem. Neurological findings were paresis and paraesthesia of the left body side. The treatment was conservative and symptoms resolved within three months.
.
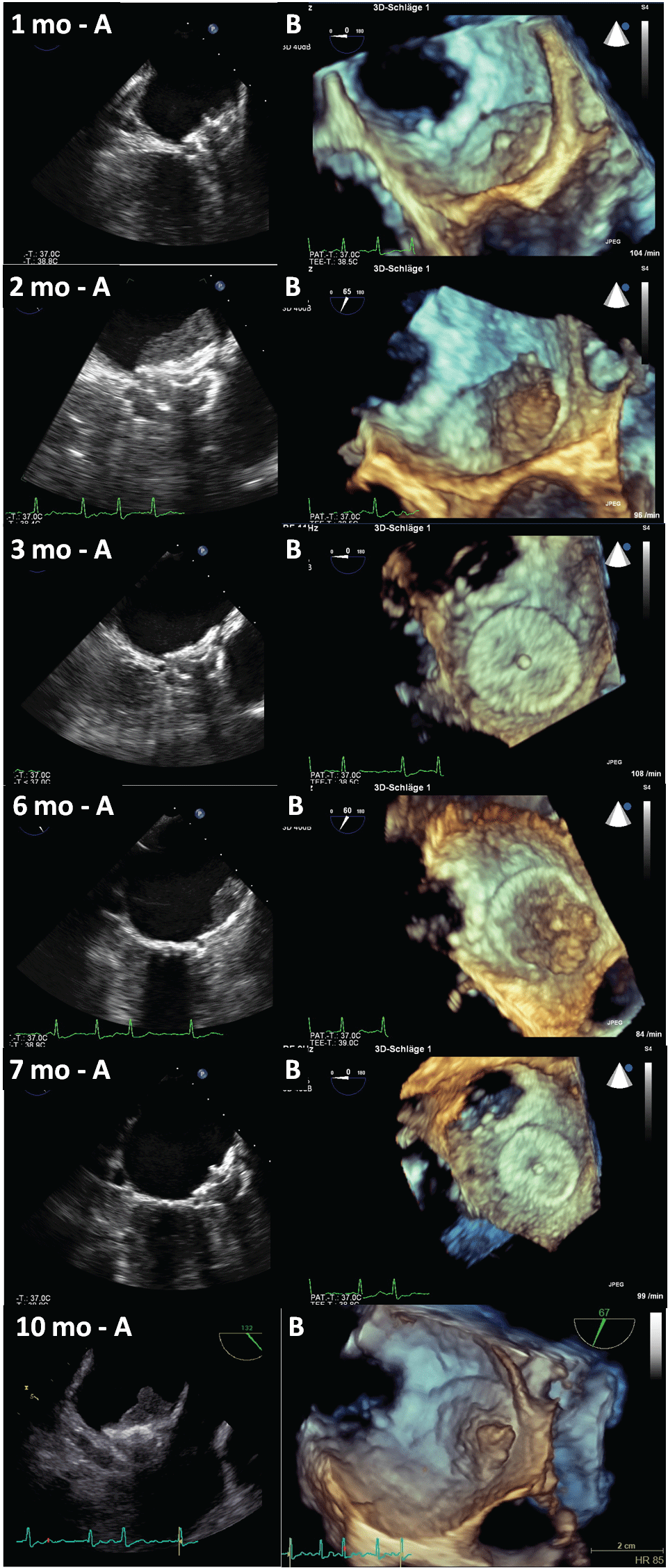
Figure 1: Follow-up examinations of the left atrial appendage occluder (LAA-O) visualized by transesophageal echocardiography with standard view (Panel A) and three-dimensional (3D) echocardiography (Panel B). mo: months
View Figure 1
At the presentation in our outpatient's clinic, the patient described good exercise capacity. No dyspnea, chest pain or syncope was reported. There was no report of thromboembolic events in the patient's or family history. Except for a moderately elevated arterial blood pressure (160/90 mmHg), the physical examination was without pathological findings. Twelve-lead ECG and Holter showed persistent atrial fibrillation. Transthoracic echocardiography revealed moderate bilateral atrial enlargement, all other cardiac dimensions were within normal limits. A normal systolic function was documented and there was no significant diastolic dysfunction of the left ventricle, or significant valvular disorders. Carotid artery stenosis was ruled out by duplex ultrasound. Blood test revealed normal renal function with a glomerular filtration rate (GFR) of 70 ml/min. Other blood parameters including international normalized ratio (INR) and activated partial thromboplastin time (aPTT) were within normal limits. Based on the patient's history und diagnostic results, a CHA2DS2Vasc-Score of 4 and a HAS-BLED-Score of 3 were calculated.
Transesophageal echocardiography (TEE) was carried out under local anesthesia and sedation with midazolam i.v.. No intracardiac thrombi were present, but thrombogenic, low Doppler velocities of blood flow (10 cm/s) were recognized in the left atrial appendage (LAA). Furthermore, size and shape of the LAA were assessed by 3D-TEE. Following interdisciplinary discussion between neurologists and cardiologists, it was advised to perform a percutaneous left atrial appendage transcatheter occlusion (LAA-O) [1,2].
The interventional occlusion of the LAA was conducted eight weeks later with an Amplatzer Cardiac Plug® (ACP) without complications. Following internal SOPs, a TEE examination was carried out four weeks later. TEE demonstrated a correct location of the LAA-O with a complete anatomical closure of the LAA. However, a thin coverage on the upper atrial part of the LAA-O device was recognized. Infection parameters were within normal range and there were no clinical signs of infective endocarditis. Antiplatelet therapy was continued and the next TEE was conducted eight weeks after implantation. Surprisingly, a large thrombus formation was present, so that an effective anticoagulation was necessary. The patient was put on Rivaroxaban® 20 mg per day and antiplatelet therapy was stopped. Follow-up TEE showed complete resolution of the thrombus one month later. The anticoagulation was continued for two months and then switched to ASS. Another follow-up TEE one month later depicted a thrombus formation at the same location which resolved after anticoagulation was re-administered for further 4 weeks. The anticoagulation was switched to ASS afterwards, and a screening for thrombophilia was conducted. With an elevated factor VIII (320% of normal activity) and a recurrent, even lager thrombus formation on the LAA-O in the next follow-up TEE, a permanent effective anticoagulation had to be advised in spite of the intracranial hemorrhage that had occurred before. No new neurological symptoms developed in context of this recurrent thrombus.
LAA-O devices are a promising method to prevent stroke in permanent atrial fibrillation patients with an elevated risk of severe bleeding under anticoagulant therapy [1,2]. As the number of LAA-O procedures increases, there is growing evidence that a number of patients needs further anticoagulation, independent from the implanted device [3,4]. Elevation of factor VIII as found in our patient may be caused by inflammation processes unspecific and represents no defined thrombophilia. However, high factor VIII levels are a common risk factor for venous thrombosis and can be associated with the risk of arterial thrombosis in coronary heart disease and stroke [5]. It has to be realized that a potential thrombogenic surface is placed in direct contact with low blood flow of the enlarged left atrium. So, according to the Virchow's triad and comparable with the use of septal occluders in patients after transient ischemic attack or stroke, a search for thrombophilia was implemented in our internal standard procedures before LAA-O implantation to minimize the risk of recurrent device-associated thrombus as shown by this case report.
Disclosures
All authors state that they have no conflicts of interest to disclose.
References
-
Lane DA, Lip GY (2012) Use of the CHA2DS2-VASc and HAS-BLED scores to aid decision making for thromboprophylaxis in nonvalvular atrial fibrillation. Circulation 126: 860-865.
-
Geis NA, Katus HA, Hardt SE (2013) Percutaneous occlusion of the left atrial appendage. Alternative to oral anticoagulation in patients with atrial fibrillation. Kardiologe 7: 122-129.
-
Fernandez-Rodriguez D, Vannini L, Martin-Yuste V, Brugaletta S, Robles R, et al. (2013) Medical management of connector pin thrombosis with the Amplatzer cardiac plug left atrial closure device. World J Cardiol 5: 391-393.
-
Plicht B, Konorza TF, Kahlert P, Al-Rashid F, Kaelsch H, et al. (2013) Risk factors for thrombus formation on the Amplatzer Cardiac Plug after left atrial appendage occlusion. JACC Cardiovasc Interv 6: 606-613.
-
Kamphuisen PW, Eikenboom JC, Bertina RM (2001) Elevated factor VIII levels and the risk of thrombosis. Arterioscler Thromb Vasc Biol 21: 731-738.





