Clinical Medical
Reviews and Case Reports
Unusual Early Aortic Valve Bioprosthesis Failure due to Fungal Infection
Yolanda Carrascal*, Gregorio Laguna and Nuria Arce
Cardiac Surgery Department, University Hospital, Spain
*Corresponding author: Dr. Y. Carrascal, Cardiac Surgery Department, University Hospital, Avda Ramón y Cajal, 347005 Valladolid, Spain, Tel: 00-34-983-420-000, Fax: 00-34-983-255305, E-mail: ycarrascal@hotmail.com
Clin Med Rev Case Rep, CMRCR-3-085, (Volume 3, Issue 1), Case Report; ISSN: 2378-3656
Received: December 09, 2015 | Accepted: January 21, 2016 | Published: January 23, 2016
Citation: Carrascal Y, Laguna G, Arce N (2016) Unusual Early Aortic Valve Bioprosthesis Failure due to Fungal Infection. Clin Med Rev Case Rep 3:085. 10.23937/2378-3656/1410085
Copyright: © 2016 Carrascal Y, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
We report an early failure of a 19 mm mitroflow A12 aortic pericardial bioprosthetic valve. We excluded all described causes related with early bioprosthesis calcification and degeneration. Neither inflammatory cells nor bacterial colonization were identified in microscopic analysis, but fungal hyphae were observed in the tissue sections from both leaflets, suggesting sub-clinical fungal endocarditis, which might have contributed to early failure.
Keywords
Endocarditis, Heart valve bioprosthesis, Infection, Calcification, Inflammation
Introduction
Anti-calcification treatment providing biological material stability is the determinant factor to avoid structural bioprosthetic valve degeneration. Implantation age under 65-70 years, diabetes mellitus, female sex, dyslipidemia, prosthesis-patient mismatch, metabolic syndrome, smoking habit and calcium metabolism disorders were proposed factors favouring bioprosthetic valve degeneration [1,2]. Proinflammatory reactions involving valve tissue might precipitate or contribute, to early bioprosthesis degeneration. In this case, associated fungal bioprosthesis colonization and induction of proinflammatory toll-like receptor 2 TLR-2-mediated response are suggested as unusual mechanisms precipitating early structural failure.
Case Report
A 70-year-old female, with arterial hypertension, diabetes mellitus type 2, hypertriglyceridemia, factor V deficiency, hypothyroidism in thyroid-replacement therapy and right mastectomy due to breast cancer. She chronically received anastrozole. After echocardiographic diagnosis of severe aortic stenosis, rheumatic mitral valve with posterior leaflet calcification and mild mitral regurgitation, the native valve was replaced with a 19 mm Mitroflow 12A pericardial bioprosthesis (Sorin Group Inc, Mitroflow Division, and Vancouver, Canada). During the immediate postoperative, patient presented with systemic inflammatory response syndrome (SIRS), severe leukocytosis (25570/μl) and increased inflammation markers (highest levels: C-reactive protein: 234.9 mg/L; Lactate: 46 mg/dl, Procalcitonin: 13.92 ngr/ml), solved on 6th postoperative day (POD). She was empirically treated with broad-spectrum intravenous antibiotics between 2nd and 8th POD. All the hemocultures were negative. At 2nd POD, she presented generalized mucocutaneous candidiasis, with favourable response to antifungal therapy (topical clotrimazole and nystatin mouth rinse). Patient was discharged home on 20th POD. Postoperative echocardiographic control showed normofunctional bioprosthesis: mean transvalvular gradient (PTG): 17 mmHg. 3 months after surgery, echocardiographic PTG increased (50 mmHg), with normofunctional leaflets. After aortic valve bioprosthesis implantation, antiagregation with dipyridamole 100 mg /12 hours was initiated and maintained after hospital discharge.
At 15 months, the patient was reoperated because of bioprosthesis dysfunction, with severe stenosis and regurgitation (peak/mean gradient: 126/81 mmHg, respectively), and severe regurgitation of calcified native mitral valve.
During reoperation, we evidenced rigid and calcified bioprosthetic leaflets, with semi-open valve position. The mitral leaflets, annulus and left ventricular outflow tract showed extensive areas of calcification. Aortic bioprosthesis and native mitral valve were replaced with a 19 mm and 25 mm Carbomedics mechanical valve, respectively, in order to avoid a third redo surgery because early and unexpected bioprosthesis degeneration. During postoperative, the patient presented again with SIRS (Procalcitonin: 2.64 ngr/ml; Lactate: 26 mg/dl; C-reactive protein: 375 mg/L; leukocytes: 15020/μl) solved on 6 POD. Postoperative treatment with intravenous meropenem and linezolid was administered between 3rd and 8th PDO. No antifungal treatment was initiated because there was no clinical suspicion of prosthesis valve endocarditis. The hemocultures were negatives. Patient was discharged home on 15th POD. Diagnosis of subclinical prosthetic endocarditis was obtained 3 months after surgery. At this moment, clinical and echocardiographic patient evaluations were normal. Blood cultures were negatives and no antifungal treatment was instituted. In echocardiograpic control at 6 months, left ventricular ejection fraction was 69% with restrictive left ventricular filling. Severe left ventricular hypertrophy. Normal prosthetic mitral valve function. Mean gradient 6 mmHg. Mean aortic transvalvular gradient: 35 mmHg. Systolic pulmonary pressure: 33 Hg.
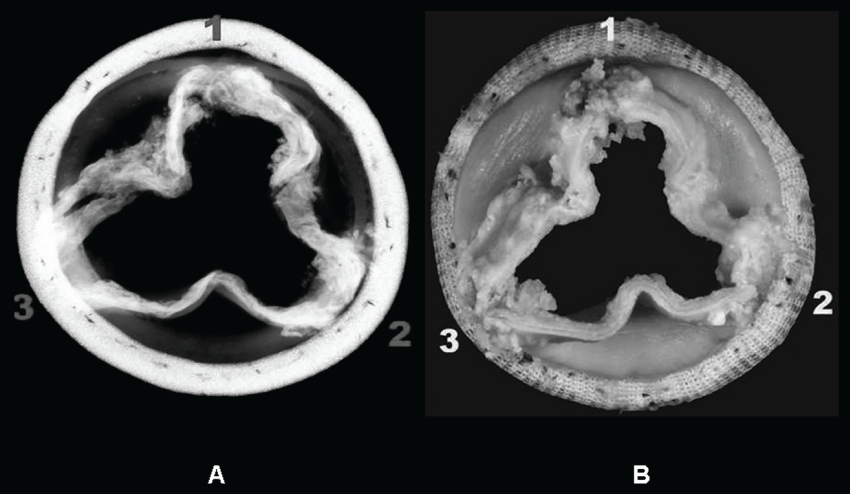
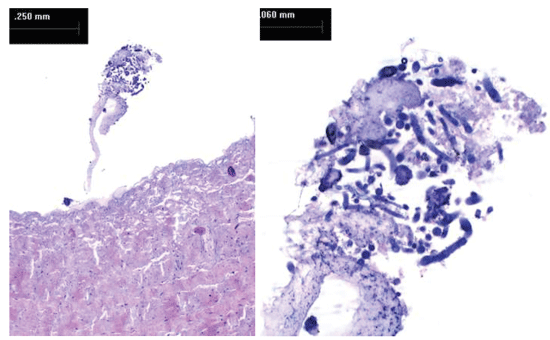
Gross examination of explanted bioprosthesis showed intrinsic and vegetating calcification in all the leaflets, confirmed by X-ray analysis. Pannus and thrombus formation were observed. Two small holes were detected in two different leaflets (< 2 mm). (Figure 1A and Figure 1B) Microscopic analysis of tissue sections confirmed leaflet calcification with Alyzarin Red S staining. Disrupted and homogenized collagen bundles were also observed. We identified pannus and thrombus formation on the mesothelial surface of the leaflet tissue. Calcified nodules were identified within some areas of thrombus formation, with no inflammatory cells or bacterial colonies. We observed fungal hyphae in the tissue sections from both leaflets. (Figure 2A and Figure 2B). Hystological findings of mitral valve described irregular leaflet valve morphology with marked fibrosis, large areas of hyalinization, and multiple foci of dystrophic calcification. No individual cultures of mitral valve was realized.
.
Figure 1: (A) Rx examination: Intrinsic and vegetating calcifications are visible in all the leaflets; (B) Macroscopic view of explanted bioprosthesis.
View Figure 1
.
Figure 2: (A) Sample belly towards the adherent margin: the collagen bundles are locally disrupted and homogenized. Small mesothelial ditches are visible in relation to the fold. Some fungal hyphae inside the thrombus deposition are visible on the mesothelial surface. Some native blood vessels are still visible; (B) (Detail) Fungal hyphae are spread inside the pannus.
View Figure 2
Discussion
Although female sex, diabetes mellitus and dyslipidemia were present in our patient, none of them have been isolated linked to early structural bioprosthesis degeneration. We excluded calcium metabolism disorders: Calcium: 9.9 mg/dl (normal range: 8-11 mg/dl), Phosphorus: 3.1 mg/dl (normal range: 2.7-4.5 mg/dl), osteocalcin: 33 ng/ml (normal range postmenopausic women: 13-48 ng/ml), procollagen-1: 32.77 ng/ml (normal range postmenopausic women: 76.3 ng/ml), beta-crosslaps: 1.06 ng/ml, (normal range postmenopausic women: ng/ml), calcitonin: 4.30 pg/ml (normal range: < 10 pg/ml) and intact-parathormone: 87 pg/ml (normal range: 15-68 pg/ml) (probably raised by chronic administration of anastrozole without calcium supplements).
Echocardiography of early structural bioprosthesis degeneration showed initially [2,3] decreased leaflet mobility, without clinical or hemodynamic translation. Later, PTG increased and clinical dysfunction was patent. In our patient, an increased PTG gradient was patent 3-months after surgery, although leaflet mobility seemed normal. As in this case, the most frequent clinical features in bioprosthesis aortic degeneration are incompetence or mixed lesion [3].
Macro and microscopic findings correspond with others already described [1,3]. Nevertheless, presence of leaflet fungal hyphae suggests a sub-clinical fungal endocarditis that might have contributed to early structural valve deterioration.
Following both cardiac surgeries, the patient presented a SIRS. Long intravenous broad-spectrum antibiotics administration and inflammatory response, could favour mucocutaneous candidiasis and secondary candidemia. Such expression of Candida infection depends on the immune status of the host, further immunity restoration prevented Candida prosthetic valve endocarditis.
Hyphae are more invasive form of Candida tissue infection. Their virulence is secondary to induction of proinflammatory response and TLR-2-mediated protection against fungal infection. Biofilm formation (over host and prosthetic surfaces), secretion of adhesins, acid proteases phospholipases and hydrolases [4] might precipitate an early bioprosthesis degeneration in an aged woman, whose only risk factors were diabetes mellitus and dyslipidemia.
We hypothesize that host tissular hyphae invasion, affected bioprosthesis and host myocardium. Candida myocarditis-pericarditis affects more than 50% of patients with disseminated candidiasis [5]. Usually latent, their most frequent presentation form is as myocardial microabscesses, more frequent after antineoplastic, antibiotic of corticosteroid treatment (such as in presented case).
Probably, after acute infection, microabscesses degenerated in calcification foci, which would explain severe calcification observed in left ventricular outflow tract, aortic and mitral annulus in our patient during re-intervention. In conclusion, subclinical endocarditis could be a risk factor for early bioprosthesis degeneration. Although infrequent, in patients affected by postoperative SIRS and previous history of fungal infection we suggest to investigate the possibility of fungal endocarditis through specific blood cultures.
Acknowledgements
Prof. Marco Galloni MSc. Associate professor Biomaterials Lab Veterinary Morphophysioly Dept, University of Turin.
References
-
Liao KK, Li X, John R, Amatya DM, Joyce LD, et al. (2008) Mechanical stress: an independent determinant of early bioprosthetic calcification in humans. Ann Thorac Surg 86: 491-495.
-
Briand M, Pibarot P, Després JP, Voisine P, Dumesnil JG, et al. (2006) Metabolic syndrome is associated with faster degeneration of bioprosthetic valves. Circulation 114: I512-517.
-
ISTHMUS Investigators (2011) The Italian study on the Mitroflow postoperative results (ISTHMUS): a 20-year, multicentre evaluation of Mitroflow pericardial bioprosthesis. Eur J Cardiothorac Surg 39: 18-26.
-
Nosanchuk JD (2002) Fungal myocarditis. Front Biosci 7: d1423-1438.
-
Felk A, Kretschmar M, Albrecht A, Schaller M, Beinhauer S, et al. (2002) Candida albicans hyphal formation and the expression of the Efg1-regulated proteinases Sap4 to Sap6 are required for the invasion of parenchymal organs. Infect Immun 70: 3689-3700.





