Clinical Medical
Reviews and Case Reports
Long-Segment Slide Tracheo-Bronchoplasty (LSTB) with Contralateral Lung Agenesis: Case Report and Review of the Literature
Daniel J. Weber1, Mark D.Rodefeld1, Bruce H. Matt2 and John W. Brown1*
1Division of Cardiothoracic Surgery, Riley Hospital for Children, Indianapolis, USA
2Department of Otolaryngology-Head and Neck Surgery, Riley Hospital for Children, Indianapolis, USA
*Corresponding author:
John W. Brown MD, Division of Cardiothoracic Surgery, Riley Hospital for Children, 545 Barnhill Dr, Indianapolis, IN 46202, USA, E-mail: jobrown@iupui.edu
Clin Med Rev Case Rep, CMRCR-2-077, (Volume 2, Issue 12), Case Report; ISSN: 2378-3656
Received: September 26, 2015 | Accepted: December 15, 2015 | Published: December 18, 2015
Citation: Weber DJ, Rodefeld MD, Matt BH, Brown JW (2015) Long-Segment Slide Tracheo-Bronchoplasty (LSTB) with Contralateral Lung Agenesis: Case Report and Review of the Literature. Clin Med Rev Case Rep 2:077. 10.23937/2378-3656/1410077
Copyright: © 2015 Weber DJ, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abbreviations
LSTB: Long-segment slide tracheo-bronchoplasty, LSTS: Long-segment tracheal stenosis, STB: Slide tracheo-bronchoplasty
The management of long-segment tracheal stenosis (LSTS) can be quite challenging, particularly in the presence of other pulmonary anomalies. We present a complicated case of LSTS and left bronchial stenosis in a 4-month-old infant with a congenitally absent right lung. This case emphasize that a slide tracheo-bronchoplasty (STB) in the setting of congenital absence of the contralateral lung is complicated but can be performed with reasonable results.
Clinical Summary
A 4-month-old girl with LSTS and congenital absence of the right lung had been a noisy breather but doing quite well until she suddenly developed respiratory failure requiring emergent intubation and ventilatory support. She was transferred to our center where a CT scan demonstrated a LSTS (Figure 1A, Figure 1B and Figure 1C) with and absent right lung. Bronchoscopy revealed complete tracheal rings with a funnel shaped trachea starting at the third tracheal ring and tapering down into the distal trachea and left bronchus. The patient was urgently taken to the operating room for a STB. After mobilization of the trachea down to the left main stem bronchus we noted that the left bronchus was 50% narrowed down to the left upper lobe takeoff and had complete bronchial rings. A STB was performed from the third tracheal ring to the left upper lobe takeoff. Due to systemic pulmonary artery pressures when initially separating from cardiopulmonary bypass as well as some mild hemodynamic instability, the sternum was initially left open and the skin was closed. Five days later the sternum was closed after her severe pulmonary hypertension resolved.
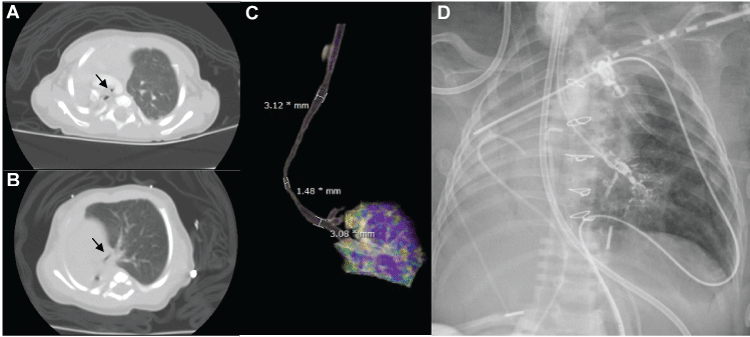
.
Figure 1: Representative computed tomographic images of long-segment tracheal and left mainstem bronchial stenosis with right lung agenesis. (A) Axial panel with severe retroaortic stenosis of distal trachea (arrow); (B) Axial panel with stenosis extending into left main-stem bronchus (arrow); (C) Three-dimensional airway reconstruction with measurements of proximal and distal trachea as well as left bronchus showing the morphology of the stenosis; (D) Fluoroscopic study with contrast instilled via endotracheal tube showing likely stenosis in distal left mainstem airway.
View Figure 1
Her postoperative course was complicated by recurrent stenosis toward the distal aspect of the repair due to transverse aortic arch anterior compression with her heart in the far right chest. When her endotracheal tube was inadvertently pulled back and could not properly replaced she required 3 days of neck ECMO support due to airway compression and was taken back to the operating room for a pericardial patch tracheoplasty for the distal tracheal obstruction (Figure 1D) which healed without issues. She was weaned from ECMO support, had a custom tracheostomy tube placed through the patch site, and was discharged home with ventilator support under the care of her family.
Discussion
The slide tracheoplasty has become the preferred method for repair of long-segment tracheal stenosis in children. Studies have demonstrated low mortality rates in the face of a very challenging problem (Table 1). When compared with previous techniques, it offers a versatile approach with documented mortality rates in the range of 5-20% from recent reports out of two large volume centers [1,2]. These rates appear even better than the results we published in 2009 using the anterior pericardial patch technique. However, it should be noted that we looked at long-term results over more than twenty years where we found 16 of the 26 patients were still alive [3]. In the largest series of slide tracheoplasties recently reported, 47% of the slide tracheoplasties were carried into the proximal bronchus and 9% of patients presented with a single lung. Using multivariate analysis, neither the presence of a single lung nor extending the tracheoplasty into the proximal bronchus was associated with increased risk of death or stent requirement post-operatively [2].
![]()
Table 1: Summary table of recent papers on tracheal stenosis with relevant findings.
View Table 1
Survival with long-segment tracheal stenosis in the setting of lung agenesis is documented to be exceedingly poor with mortality rates around 30% to 50% from small case series over the last fifteen years [4,5]. More recently, successful tracheoplasties have been documented in patients with unilateral lung agenesis or severe hypoplasia [6]. Outcomes from 11 single-lung patients (7 with a congenitally absent lung and 4 with a hypoplastic lung) were found to be similar to 60 patients with two lungs. In particular, the mortality of the single lung group was 18% while it was 24% from the double lung group. Of the 11 with single lungs, 3 underwent slide tracheoplasty (the more recent cases) while 8 of the 60 underwent slide tracheoplasty in the double lung group. Interestingly, slide tracheoplasty was not carried into the bronchus in any of the single lung patients as was necessary in our patient. We have previously encountered two similar cases of an absent contralateral lung but this was our first patient where bronchoplasty was required as well.
Certainly extension of stenosis into the bronchus in the setting on an absent contralateral lung adds to the complexity of the repair and likely increases the morbidity and mortality of the slide tracheoplasty. Consequently, post-operative vigilance is required for complications as well as the development of pulmonary hypertension. Ultimately, good results can be obtained in this challenging group of children. This reinforces recent reports demonstrating that improved outcomes are being obtained with the slide tracheoplasty technique even with coexisting complex anomalies.
References
-
Manning PB, Rutter MJ, Lisec A, Gupta R, Marino BS (2011) One slide fits all: the versatility of slide tracheoplasty with cardiopulmonary bypass for airway reconstruction in children. J Thorac Cardiovasc Surg 141: 155-161.
-
Butler CR, Speggiorin S, Rinjnberg FM, Roebuck DJ, Muthialu N, et al. (2014) Outcomes of slide tracheoplasty in 101 children: a 17-year single-center experience. J Thorac Cardiovasc Surg 147: 1783-1789.
-
Fanous N, Husain SA, Ruzmetov M, Rodefeld MD, Turrentine MW, et al. (2010) Anterior pericardial tracheoplasty for long-segment tracheal stenosis: Long-term outcomes. J Thorac Cardiovasc Surg 139: 18-25.
-
Anton-Pacheco JL, Cano I, Garcia A, Martinez A, Cuadros J, et al. (2003) Patterns of management of congenital tracheal stenosis. J Pediatr Surg 38: 1452-1458.
-
Kocyildirim E, Kanani M, Roebuck D, Wallis C, McLaren C, et al. (2004) Long-segment tracheal stenosis: slide tracheoplasty and a multidisciplinary approach improve outcomes and reduce costs. J Thorac Cardiovasc Surg 128: 876-882.
-
Backer CL, Kelle AM, Mavroudis C, Rigsby CK, Kaushal S, et al. (2009) Tracheal reconstruction in children with unilateral lung agenesis or severe hypoplasia. Ann Thorac Surg 88: 624-630.





