Clinical Medical Reviews and Case Reports
Isolated Ovarian Metastasis in a Woman with Prior History of Superficial Bladder Cancer
Kathleen Mahoney1,3, Mamta Gupta2 and Akash Patnaik1*
1Division of Hematology/Oncology, Beth Israel Deaconess Medical Center, Harvard Medical School, Boston, USA
2Department of Pathology, Beth Israel Deaconess Medical Center, Harvard Medical School, Boston, USA
3Department of Medical Oncology, Dana-Farber Cancer Institute, Harvard Medical School, Boston, USA
*Corresponding author: Akash Patnaik, Division of Hematology/Oncology, Beth Israel Deaconess Medical Center, 330 Brookline Ave, Boston, MA 02215, USA, Tel: 617-735-2048, Fax: 617-735-2062, E-mail: apatnaik@bidmc.harvard.edu
Clin Med Rev Case Rep, CMRCR-2-064, (Volume 2, Issue 10), Case Report; ISSN: 2378-3656
Received: March 16, 2015 | Accepted: October 09, 2015 | Published: October 12, 2015
Citation: Mahoney K, Gupta M, Patnaik A (2015) Isolated Ovarian Metastasis in a Woman with Prior History of Superficial Bladder Cancer. Clin Med Rev Case Rep 2:064. 10.23937/2378-3656/1410064
Copyright: © 2015 Mahoney K, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Primary transitional cell carcinoma of the ovary is a rare form of ovarian cancer, which has many features in common with transitional cell carcinoma of the bladder. Here we describe a case of metastatic transitional cell carcinoma to the ovary in a patient with a remote history of primary superficial urothelial carcinoma. Metastasis from a superficial urothelial carcinoma is exceedingly rare. CK7/CK20 positivity is not typical of transitional cell carcinoma (TCC) of the ovary, thus co-expression of CK7 and CK20 may distinguish metastatic urothelial carcinoma to the ovary from primary ovarian cancer. Strong nuclear WT1 staining, a marker of serous ovarian carcinoma, is frequently present in transitional cell carcinoma of the ovary, thus may also be useful in distinguishing these diagnoses.
Keywords
Transitional cell carcinoma, Bladder, Ovarian, Krukenberg-like
The ovary can be a metastatic site for many primary tumors, the classic scenario being bilateral Krukenberg tumors from primary gastric, but also colon, breast and endometrial cancers [1,2]. We present a case of a metastatic transitional cell carcinoma to the ovary in a patient with a remote history of primary superficial urothelial carcinoma. This case has many unique features, which produced both a diagnostic and a treatment conundrum.
An 82 year old woman presented with abdominal distension and abdominal mass detected on examination. CT scan revealed a 13.3 × 15.5 × 17.4 cm complex cystic mass (Figure 1A), with a solid component in the left pelvis (Figure 1B), but no evidence of ascites, lymphadenopathy, visceral or bony metastasis. She underwent a total abdominal hysterectomy with bilateral salpingo-oophorectomy (TAH-BSO). An intraoperative consultation for pathology was reported as a high-grade carcinoma, most consistent with serous carcinoma. Therefore a complete staging procedure, with pelvic and para-aortic lymph node resection, infra-gastric omentectomy, and peritoneal biopsies, was performed. However, final pathology revealed a high grade invasive transitional cell carcinoma with foci of central necrosis (arrows, Figure 1C), and papillary architecture (Figure 1D) with conventional and micropapillary areas (arrows; Figure E). On immunostains tumor cells were positive for CK7 (Figure 1F), CK20 (Figure 1G) and p63 (Figure 1H). A WT1 immunostain showed focal cytoplasmic staining and only scattered foci of nuclear positivity were seen (Figure 1I).
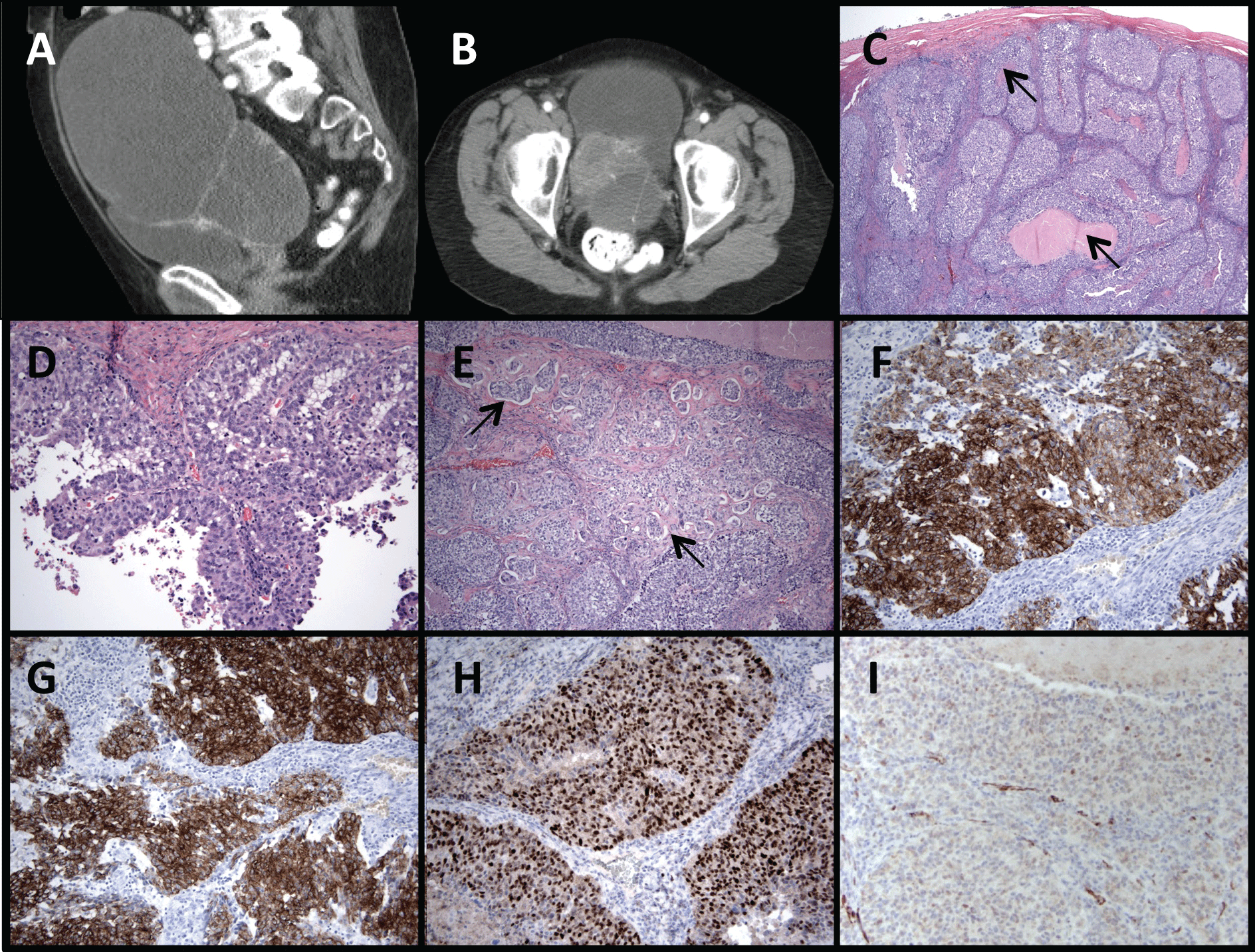
.
Figure 1: Complex pelvic mass 13.3 cm × 15.5 cm × 17.4 cm (A) with solid component in left pelvis (B). High grade transitional cell carcinoma with foci of central necrosis (arrows; C), papillary architecture (D), and with conventional and micro papillary areas (arrows; E). Immuno stains consistent with TCC of the bladder: positive CK 7 (F), positive CK 20 (G), positive p63 (H), and with largely cytoplasmic WT-1 staining with focal nuclear staining (I).
View Figure 1
The patient's past medical history was significant for high-grade papillary urothelial carcinoma with lamina propria invasion diagnosed six years prior to the TAH-BSO. At that time, she was diagnosed in the setting of two episodes of gross hematuria, which prompted CT abdomen and pelvis and found a 2.4 cm mass within the bladder with no associated lymphadenopathy. Cystoscopy revealed a 4 to 5 cm fungating mass on the right lateral wall with multiple satellite lesions measuring up to 2 cm. She underwent transurethral resection of bladder tumor (TURBT), although limited muscularis propria was present in the sample no muscularis propria invasion was identified. She was treated with adjuvant BCG-interferon-α therapy. During the current presentation with high-grade invasive papillary transitional cell carcinoma (TCC) of the ovary, she underwent routine cystoscopy and urine cytology every 3 to 4 months, with surveillance random biopsies, which were negative for recurrent disease in the bladder. A magnetic resonance urogram was performed to rule out upper tract transitional cell carcinoma as a recurrent primary genitourinary lesion, and this also did not reveal any lesions in collecting duct or bladder. In light of the rarity of primary ovarian TCC, a prior history of superficial urothelial carcinoma, and morphological and immunostain similarities between the prior superficial high-grade urothelial carcinoma and the current ovarian cancer, the diagnosis was consistent with a metastatic TCC from her prior superficial urothelial primary lesion.
While there are some studies describing the primary origin of isolated ovarian metastasis, rarely is there significant clinical history reported in such cases [1,2]. Prior to the establishment of immunohistochemistry, it was particularly difficult to interpret whether transitional cell ovarian cancer and urothelial carcinoma with metastasis to the ovary were distinct primaries or metastatic disease respectively [3]. Three case reports with clinical data exist on metastatic urothelial carcinoma from the bladder, with metastasis to the ovary [4-6]. These prior case reports represent a wide spectrum of clinical presentations, different from the case described in this report. The first case described a 47 year old woman with high grade papillary TCC of bladder with lymph node metastasis treated with radical cystectomy and incomplete adjuvant chemotherapy (due to poor performance status), who then presented a year later with her first distant metastatic recurrence to the ovary [6]. This patient was at high risk for metastatic disease, due to lymph node metastasis at initial presentation, and inability to tolerate adjuvant chemotherapy after radical cystectomy. The second case was a 49 year old woman with a right ovarian mass with pelvic and para-aortic lymphadenopathy and microscopic hematuria, who was found to have a pathologic diagnosis of undifferentiated carcinoma of the ovary that resembled TCC. Following hysterectomy and bilateral adnexectomy, she underwent urologic evaluation for microscopic hematuria, and found to have multiple, superficial grade 3 papillary bladder tumors. She was treated with platinum doublet chemotherapy, and had complete resolution of her lymph node metastasis and normalization of CA-125 levels. However her cystoscopy showed no remarkable change in the number and size of the superficial bladder tumors, and she underwent transurethral resection and BCG bladder installation to prevent recurrence [4]. The third reported case was of a 60 year old woman with grade 2 superficial TCC of the bladder resected by TURBT. She subsequently developed additional non-invasive bladders tumors at 3 and 6 months of follow-up, and was treated with TURBT and adjuvant intravesical mitomycin C. She then developed an additional recurrence of a Grade 1 pTa TCC of the bladder after 39 months. She subsequently developed an ovarian metastasis 9 months later, which was found to be grade 3 TCC [5]. Unfortunately 3 months after her total hysterectomy, bilateral oophorectomy and partial omentectomy, she developed pelvic metastases and died within 2 weeks of starting systemic cisplatin-based chemotherapy. Of note, a case of transitional cell carcinoma of the ovary has been reported in a patient with a history of recurrent TCC of the bladder [7]. While the morphology of bladder and ovarian tumors in this case were similar, there was discordance in the immunohistochemical profile between the TCC of the bladder (CK7+, CK20+, CA125-) and the TCC of ovary (CK7+, CK20-, CA125+).
Our case presents a diagnostic conundrum for several reasons. First, late distant recurrence of a single, superficial TCC of the bladder is exceedingly rare. Most superficial tumors that recur do so in the first 5 years, and do so locally, not by distant metastasis [8,9]. Second, oligometastatic TCC to the ovary, with no recurrence at primary site, has not been previously described. Third, it remains conceivable that this represents a primary Stage I ovarian transitional cell carcinoma with no relationship to prior history of superficial TCC. Our case, however, showed marked morphological similarity between the prior lesion in the bladder and current lesion in the ovary, with an immune histochemical profile that supported a primary urothelial over a primary ovarian surface epithelial tumor. While CK7 typically shows diffuse positivity in primary ovarian cancer, CK20 positivity is typically confined to mucinous carcinomas of the ovary [4]. Our case showed a strong, diffuse positivity for both CK7 and CK20, a feature typical for primary urothelial carcinoma and not described in primary TCC of the ovary [4,10]. Strong nuclear WT1 staining is a marker of serous ovarian carcinomas, and is frequently positive in primary ovarian TCC [10]. Our case showed some cytoplasmic positivity for WT-1 with only rare cells with nuclear staining, further supporting a diagnosis of primary urothelial carcinoma metastatic to the ovary..
Primary Transitional Cell Carcinoma (TCC) of the ovary is a rare form of ovarian cancer, and is considered to have better prognosis than serous adenocarcinoma of the ovary [11,12]. Our patient is of Ashkenazi Jewish ancestry with family history of ovarian cancer in a primary relative. Therefore she was tested for BRCA1, given the association between BRCA1 loss and primary ovarian TCC, but was found to be negative [13]. The possibility of Lynch Syndrome, which may be associated with increased risk for urothelial carcinoma, was ruled out by immunohistochemical screening for DNA mismatch repair proteins MLH-1, PMS-2, MSH-2, and MSH-6, all of which showed intact nuclear staining [14]. It is highly unlikely that this patient with a history of primary high grade urothelial carcinoma also developed a separate ovarian primary of a rare phenotype, that was histopathologically and immunohistochemically identical to the prior urothelial primary. It must also be emphasized that despite lack of muscularis propria invasion, the prior urothelial cancer was of high-grade. It is conceivable that a focus of muscularis propria invasion was missed in the urothelial primary due to sampling error, which was responsible for the late recurrence in the ovary.
In summary, we present an interesting case of an elderly woman with isolated ovarian TCC, in the context of remote prior history of superficial high-grade TCC of the bladder. The standard of care for treating metastatic TCC of the bladder is chemotherapy with gemcitabine and cisplatin. However, our patient had only one site of metastatic disease with no recurrence at the primary site. There is no data to support treatment with chemotherapy in this setting, and active surveillance would be a reasonable option after complete surgical resection of the single focus of metastatic of disease. On the other hand, if this represents primary ovarian Stage 1, grade 3, TCC, the patient would potentially benefit from adjuvant platinum-based chemotherapy. We discussed with the patient that a diagnosis of a primary ovarian cancer cannot be excluded, despite the presence of a prior history of high-grade urothelial carcinoma. Given her excellent performance status and goal to maximize the possibility of a cure, we supported her decision to pursue adjuvant chemotherapy with 6 cycles of adjuvant carboplatin. She continues to do well, without evidence of recurrence, over two years after resection of her ovarian mass.
References
-
Abrams HL, Spiro R, Goldstein N (1950) Metastases in carcinoma; analysis of 1000 autopsied cases. Cancer 3: 74-85.
-
Ulbright TM, Roth LM, Stehman FB (1984) Secondary ovarian neoplasia. A clinicopathologic study of 35 cases. Cancer 53: 1164-1174.
-
Young RH, Scully RE (1988) Urothelial and ovarian carcinomas of identical cell types: problems in interpretation. A report of three cases and review of the literature. Int J Gynecol Pathol 7: 197-211.
-
Ishii Y, Itoh N, Takahashi A, Masumori N, Ikeda T, et al. (2005) Bladder cancer discovered by ovarian metastasis: cytokeratin expression is useful when making differential diagnosis. Int J Urol 12: 104-107
-
Kardar AH, Lindstedt EM, Tulbah AM, Bazarbashi SN, al Suhaibani HS (1998) Metastatic transitional cell carcinoma of the ovary from superficial bladder tumour. Scand J Urol Nephrol 32: 73-76.
-
Lee M, Jung YW, Kim SW, Kim SH, Kim YT (2010) Metastasis to the ovaries from transitional cell carcinoma of the bladder and renal pelvis: a report of two cases. J Gynecol Oncol 21: 59-61.
-
Kadiri H, Jahid A, Zouaidia F, Sbitti Y, Ismaili N, et al. (2010) Transitional cell carcinomas in the bladder and ovary: unusual primary association or metastatic lesions? Taiwan J Obstet Gynecol 49: 373-376.
-
Herr HW, Wartinger DD, Fair WR, Oettgen HF (1992) Bacillus Calmette-Guerin therapy for superficial bladder cancer: a 10-year followup. J Urol 147: 1020-1023.
-
Davis JW, Sheth SI, Doviak MJ, Schellhammer PF (2002) Superficial bladder carcinoma treated with bacillus Calmette-Guerin: progression-free and disease specific survival with minimum 10-year followup. J Urol 167: 494-500.
-
Ali RH, Seidman JD, Luk M, Kalloger S, Gilks CB (2012) Transitional cell carcinoma of the ovary is related to high-grade serous carcinoma and is distinct from malignant brenner tumor. Int J Gynecol Pathol 31: 499-506.
-
Silva EG, Robey-Cafferty SS, Smith TL, Gershenson DM (1990) Ovarian carcinomas with transitional cell carcinoma pattern. Am J Clin Pathol 93: 457-465.
-
Eichhorn JH, Young RH (2004) Transitional cell carcinoma of the ovary: a morphologic study of 100 cases with emphasis on differential diagnosis. Am J Surg Pathol 28: 453-463.
-
Soslow RA, Han G, Park KJ, Garg K, Olvera N, et al. (2012) Morphologic patterns associated with BRCA1 and BRCA2 genotype in ovarian carcinoma. Mod Pathol 25: 625-636.
-
van der Post RS, Kiemeney LA, Ligtenberg MJ, Witjes JA, Hulsbergen-van de Kaa CA, et al. (2010) Risk of urothelial bladder cancer in Lynch syndrome is increased, in particular among MSH2 mutation carriers. J Med Genet 47: 464-470.





