Clinical Medical
Reviews and Case Reports
New Onset Nephropathy in a Patient with Psoriatic Skin and Joint Disease
Savvas Psarelis1,2, Sophia Corsava2*, Kyriakos Polycarpou3, Kyriakos Ioannou4, Ioanna Zouvani5 and Elena Nikiphorou6
1Rheumatology Department, Nicosia General Hospital, Cyprus
2St George's University of London Medical School, UK
3Dermatology Department, Nicosia General Hospital, Cyprus
4Nephrology Department, Nicosia General Hospital, Cyprus
5Histopathology Department, Nicosia General Hospital, Cyprus
6Rheumatology Department, Cambridge University Hospitals Foundation Trust, UK
*Corresponding author:
Sophia Corsava, St George's University of London Medical School, UK, E-mail: sophiadr@live.com
Clin Med Rev Case Rep, CMRCR-2-053, (Volume 2, Issue 9), Case Report; ISSN: 2378-3656
Received: June 08, 2015 | Accepted: September 02, 2015 | Published: September 05, 2015
Citation: Psarelis S, Corsava S, Polycarpou K, Ioannou K, Zouvani L, et al. (2015) New Onset Nephropathy in a Patient with Psoriatic Skin and Joint Disease. Clin Med Rev Case Rep 2:053. 10.23937/2378-3656/1410053
Copyright: © 2015 Psarelis S, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
We present the case of a 38-year-old male patientwho presented with new onset nephropathy on a background of psoriatic skin and joint disease. The pathogenesis and link between nephropathy with psoriatic disease is discussed, along with potential diagnostic and management challenges. This case highlights that nephropathy in psoriatic skin and joint disease can and does occur and high vigilance is necessary especially in patients taking potentially nephrotoxic drugs. The complexity of these cases necessitates close management and involvement of appropriate specialists, regular follow-up and careful monitoring.
Introduction
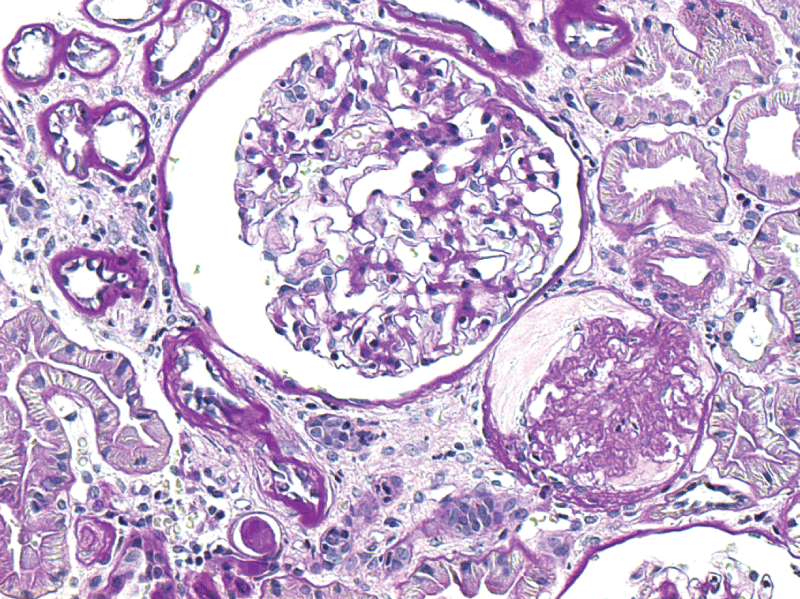
A 38-year-old male patientwith a background history of psoriasis since the age of 21 (1997) was referred to rheumatology due to a widespread flare of joint disease. He had been on cyclosporine 400 mg daily between 2004 and 2012 for his skin, but this was discontinued by the dermatologists due to abnormally elevated urea and creatinine. After stopping cyclosporine, the patient developed joint symptoms and his skin flared shortly (Figures 1,2).
.
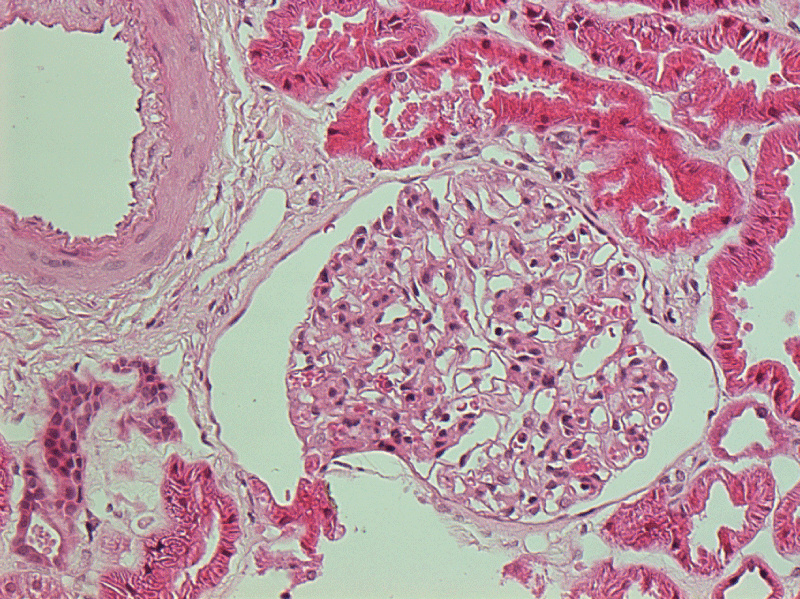
Figures 3 & 4: Electron microscopy demonstrated electron dense depositions within the mesangial and sub endothelial areas.
View Figures 3 & 4
Case Presentation
The patient was referred to rheumatology with a two-month history of a widespread joint flare affecting his shoulders, right elbow and several of his MCP and MTP joints along with evidence of dactylitis. He was commenced on methotrexate 15 mg/week but this had to be soon discontinued due to gastrointestinal side effects. His urinalysis showed a trace of protein. His inflammatory markers were raised with a CRP of 47 (< 5) and ESR of 54 (< 0-14). Creatinine was raised at 2,6 mg/dl [0,51-0,95], urea 90 mg/dl [17-43], C3 and C4 were 125 [90-130] and 35 [10-40] respectively. Serologically he had negative ANA, pANCA/cANCA, dsDNA, anti-cardiolipin and beta 2 glycoprotein antibodies, but lupus anticoagulant was weakly positive. An ultrasound of the kidneys showed slight reduction in the size of the right kidney and was otherwise unremarkable. A 24-hour urine collection revealed 616mg of protein [0-80]. An infective screen which included screening for HIV, hepatitis B and C was negative.
The initial working diagnosis was a flare of skin and joint disease in the context of psoriasis and psoriatic arthritis, with kidney dysfunction thought to be secondary to long-standing use of cyclosporin. The decision was taken to introduce a biologic, etanercept being the first choice following discussion with dermatologists. He was established on 50 mg etanercept subcutaneously once weekly.
Two months into treatment with etanercept, there was marked clinical improvement in both skin and joints. His creatine, urea and proteinuria initially improved but on a subsequent screen 4 months later, there was further deterioration in his creatinine level. At this point, the nephrologists advised for a kidney biopsy. This showed mesangio proliferative glomerulo nephritis with diffuse IgG, IgA, IgM, C3 (C4 negative) and C1q deposition. Electron microscopy demonstrated electron dense depositions within the mesangial and subendothelial areas (Figures 3,4). Congo red staining for amyloid deposition was negative (images available for presentation). Etanercept was discontinued; he was started on oral prednisolone 0.5 mg/kg and was established on mycophenolate 1g BD. The prednisolone was gradually tapered down to 10 mg daily. This unfortunately resulted in a flare of his skin albeit not his joints. Kidney function remained stable with a creatinine of 1.8 and urea of 80. The decision was taken to cautiously re-introduce the etanercept and withhold the mycophenolate, partly due to the patient's request. His skin settled and prednisolone had been eventually completely withdrawn over a period of 8 months.
Discussion
This is a case of long-standing psoriasis/psoriatic arthritis in a 38-year-old male on cyclosporine presenting with a new onset nephropathy at the time of skin and joint flare. The latter could be a consequence of a number of pathologies including secondary amyloidosis, iatrogenic aetiology (e.g. cyclosporine/NSAID related) or IgA nephropathy [1]. Cyclosporine works by inhibiting calcineurin which through a series of complex mechanisms inhibits T-cell activation causing immunosuppresion. Cyclosporin Induced Nephrotoxicity (CIN) is thought to be caused by arteriolopathy: a consequence of altered vascular flow and constriction of the afferent arteriole diameter. An increase in thromboxane and endothelin (vasoconstrictive factors), activates the Renin-Angiotensin-aldosterone System (RAAS) resulting in increased aldosterone and angiotensin II levels, reduced Nitric Oxide and prostacyclin (vasodilator factors) promoting eventually prothrombotic activity in the glomeruli. Decreased levels of COX-2 cyclo oxygenase have also been reported in CIN, as have increased levels of TGFβ1 factor all contributing to treatment-resistant hypertension. However our patient, although on long term cyclosporine did not have any signs or symptoms consistent with CIN. Moreover the kidney biopsy showed C3 and C1q deposits which have not been specifically reported in CNI [2].
Less likely causes would be the co-existence of lupus erythematosus with psoriasis/psoriatic arthritis [3]. The full house immune staining demonstrated on the kidney biopsy would support such a diagnosis, although there were no clinical manifestations of lupus. Other possibilities include post-infective nephropathies like HIV and HCV-related nephropathies which were screened for and shown negative in this case.
Taking into account all the above, the working diagnosis in this case was that of "Psoriatic Nephropathy", a fairly well-described entity [4,5]. Mesangio proliferative glomerulonephritis with IgA, C3 and C1q deposition on the mesangium and basal membranes of glomerular capillaries appears to be the commonest type in psoriasis, although focal proliferative glomerulonephritis and Focal Segmental Glomerulosclerosis (FSC) as well as minimal change disease have also been reported albeit less commonly [4-6].
Recent evidence suggests that an increasing number of patients with skin psoriasis develop renal dysfunction [6-9]. In one case, renal transplant completely alleviated all psoriatic manifestations with the patient remaining psoriasis free ever since [9]. This raises important issues regarding the correlation between psoriasis and kidney disease and merits further investigation. Considering thorough investigations and kidney-biopsy in such patients to identify the exact mechanism of FSC or minimal change disease is thus justifiable and recommended. These findings could be used to develop novel therapeutic regiments for psoriatic patients, optimizing their management.
This case highlights that nephropathy in the context of Psoriatic skin and joint disease can and does occur and high vigilance is necessary. An autoimmune, inflammatory mechanism could link the two although usage of nephrotoxic drugs in psoriasis should be kept in mind and carefully considered. The complexity of these cases necessitates close monitoring and follow-up with involvement of appropriate specialists early on.
References
-
Galla JH (1995) IgA nephropathy. Kidney Int 47: 377-387.
-
Issa N, Kukla A, Ibrahim HN (2013) Calcineurin inhibitor nephrotoxicity: a review and perspective of the evidence. Am J Nephrol 37: 602-612.
-
Zalla MJ, Muller SA (1996) The coexistence of psoriasis with lupus erythematosus and other photosensitive disorders. Acta Derm Venereol Suppl (Stockh) 195: 1-15.
-
VarshavskiÄ VA, Panasiuk NN, Antonov MV, Fal'kovskaia OG (1990) The morphological characteristics of psoriatic nephropathy. Arkh Patol 52: 32-36.
-
Singh NP, Prakash A, Kubba S, Ganguli A, Singh AK, et al. (2005) Psoriatic nephropathy--does an entity exist? Ren Fail 27: 123-127.
-
Pradhan SK, Beriha SS (2014) Focal segmental glomerulosclerosis associated with psoriasis. Saudi J Kidney Dis Transpl 25: 670-671.
-
Dervisoglu E, Akturk AS, Yildiz K, Kiran R, Yilmaz A (2012) The spectrum of renal abnormalities in patients with psoriasis. Int Urol Nephrol 44: 509-514.
-
Nakamura-Wakatsuki T, Kato Y, Sakurai K, Yamamoto T (2013) A case of severe erythrodermic psoriasis associated with IgA nephropathy. Int J Dermatol 52: 1579-1581.
-
Mele C, Salerno MP, Romagnoli J, De Simone C, Castriota M, et al. (2013) Complete clinical remission of psoriasis 6 months after renal transplantation. Transplant Proc 45: 2788-2789.