Clinical Medical
Reviews and Case Reports
A Case of Psoriasis in an HIV Positive Male
Omar F Merchant and Wayne X Shandera*
Baylor College of Medicine, Department of Internal Medicine, Houston, USA
*Corresponding author: Wayne Shandera, Baylor College of Medicine, Department of Internal Medicine, Houston, TX 77030, USA, Fax: 713 798 6400; E-mail: shandera@bcm.edu
Clin Med Rev Case Rep, CMRCR-2-052, (Volume 2, Issue 8), Case Report; ISSN: 2378-3656
Received: April 22, 2015 | Accepted: August 27, 2015 | Published: August 29, 2015
Citation: Merchant OF, Shandera WX (2015) A Case of Psoriasis in an HIV Positive Male. Clin Med Rev Case Rep 2:052. 10.23937/2378-3656/1410052
Copyright: © 2015 Merchant OF, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Patients with Human Immunodeficiency Virus (HIV) infection have an increased risk for several variants of psoriasis as well as being refractory to standard treatment regimens. While psoriasis does not affect HIV survival, quality of life may be significantly impaired and these considerations warrant special attention with management. We report a case of psoriasis in a 43-year-old Hispanic male with HIV infection, adequately managed with antiretroviral therapy. He presented with chronic and persistent guttate, red, scaly plaques on his abdomen, back, buttock as well as the extensor surfaces of his extremities. Lesions responded only mildly to steroids, with adequate response attained by administration of high dose acitretin. HIV-associated psoriasis has a unique pathophysiologic profile and disease course. Optimal therapeutic response is often achieved with special regimens due to the relative contraindication to biologic agents in HIV+ patients.
Keywords
Psoriasis, HIV, AIDS
Case Synopsis
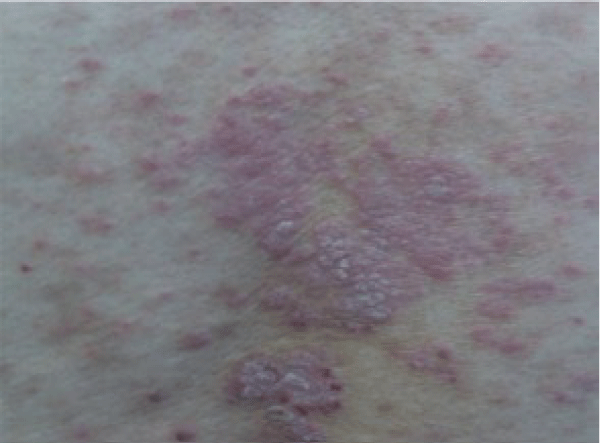
A 43-year-old Hispanic male with known HIV infection diagnosed in 2004 presented with chronic and persistent skin lesions on his abdomen, back, buttock, and extremities (Figure 1). The patient was Peruvian, with a Venezuelan mother, and had lived in Panama, Colombia, Venezuela (including Margarita Island) and Peru. He also served a prison sentence, prior to presentation. Patient lived alone. He reported a history of tuberculous adenitis that was treated with an unknown regimen. His HIV infection was well controlled using various antiretroviral therapy regimens. He had no family history of arthropathy or rheumatic diseases.
On physical examination he was healthy appearing, showed no cachexia, manifested stable vital signs, and had no conjunctival, mucosal or genital lesions, no joint involvement and normal cardiovascular, pulmonary and gastrointestinal exams.
His skin exam was remarkable for guttate, erythematous, scaly plaques on his abdomen, back and buttock as well as the extensor surfaces of his upper and lower extremities. The lesions were not tender and they did not have any drainage.
Pertinent labs were drawn. CBC, basic chemistries, lipids and RPR were within normal limits. His ANA screen was also negative. His most recent CD4 count was 230 cells/cu mm and his viral load was controlled at fewer than 20 copies/cu mm.
Initially, topical steroids were attempted with minimal improvement, and low dose acitretin was also ineffective. High dose acitretin, however, did result in significant response with improvement diffusely in the skin lesions.
Discussion
Psoriasis is prevalent 1-2% of northern Europeans, with 5-42% having concomitant arthritis [1]. The histological findings of psoriasis include proliferation of basal keratinocytes, thickened epidermis, premature desquamation of the stratum corneum and neutrophils in the stratum corneum. Dilated capillaries are also typically evident in the papillary dermis. Psoriasis in HIV-infected patients shows several distinctive characteristics. Irregular acanthosis is commonly noted, with less thinning of the suprabasal layer, fewer microabscesses, necrotic keratinocytes and plasma cells.
Psoriasis has a strong genetic component with 30-50% of patients having PSOR1 locus on chromosome 6p and HLV C26 with early onset and guttate "drop" forms [2]. Infectious agents have been implicated as triggers of psoriasis, specifically group A beta-hemolytic strep and several viruses, which include, HPV5 (found in 80% of patients), HSV, VZV, CMV, parvovirus B19, Human endogenous retrovirus [1]. A group in Houstonian 1987 described worsening of psoriasis and development of arthropathy with administration of systemic immunosuppression [3]. A study in Berlin found that 36 of 700 HIV+ patients had psoriasis, with 80% of them being in late stages of HIV [4]. Their psoriasis course was severe, prolonged and had more frequent exacerbations.
African reviews of HIV patients with skin manifestations described other entities more commonly than psoriasis such as eczema, benign skin tumors, folliculitis, candidosis, seborrheic dermatitis and Kaposi sarcoma [5,6], but also noted that at least in one series psoriasis is one of three main reasons for inpatient admission [7]. A high proportion of psoriatic patients (24%) show the erythroderma form of the disease.
The differential diagnosis for psoriasis in the HIV-infected is extensive but serious consideration needs to be given towards drug reactions, secondary syphilis, lamellar ichthyosis, cutaneous T-cell lymphoma, ringworm (which can resemble annular psoriasis), Norwegian scabies (which may not be pruritic, and shows often scaly plaques), Reiter's syndrome, and fulminant erythroderma [8]. Consultation with dermatology and use of biopsy material read by skilled dermatopathology staff may be essential in establishing a firm diagnosis.
Classically, psoriasis is thought to result from activation of T cells, with treatment targeting the suppression of T cell activity. Paradoxically, in HIV patients, severe immunosuppression is commonly associated with psoriasis, during which time the T cell count is characteristically low. Explanations involving the balance of Th1 and Th2, or CD4 and CD8, and disruption of the pro-inflammatory and anti-inflammatory auto regulation pathways, are offered [9]. Elevated IFN-gamma levels are hypothesized to be a contributing factor. HIV RNA transcripts are found in dermal and epidermal dendritic cells, suggesting a more direct role of HIV on psoriasis progression [1].
A peculiar clinical phenomenon has also been noted in which the psoriasis in patients improves shortly before death. It has been suggested that HIV- nef acts as a superantigen on T cells resulting in increased autoregulation. HLA B-27, associated with psoriasis, is an MHC I molecule, or CD8 cell target, which is also the target of the HIV virus. The disruption of CD8 cells may potentiate patient's underlying risk of developing psoriasis [1]. While there is no consistent model for these findings, there are several empiric explanations found in the literature.
The treatment for HIV-associated psoriasis initially calls for restoring immunity with ART. Standard precautions for triggers including cigarette smoke, alcohol and various medications apply. The first-line use of topical emollients and corticosteroids is useful along with coal tar. Other topical agents such as dithranol and tazarotene may also be effective.
Phototherapy using psoralens (furocoumarins, specifically methoxsalen) with UV light A or UV light B alone may be useful since they inhibit cells proliferation and inflammatory mediators but it is not always effective [10]. Due to the extensive medication regimen HIV+ patients are prescribed, potential interactions must be accounted for (specifically photosensitization with Bactrim). The oral retinoid Acitretin, as used in our patient, is effective and safe for HIV-infected patients due to its lack of HIV-immunosuppressive activity. It has been recommended since the 1990s [11]. It is given in doses between 25 and 50 mg orally daily (with reduction of the higher dose when symptoms abate). Significant side effects include teratogenicity (requiring women to use two forms of birth control, refrain from pregnancy for at least three years and men to refrain from donating sperm for at least three years after use). When combined with UV therapy, it causes rapid, synergistic clearing of lesions [1,12], although it is not universally successful [10]. Synergism may occur between retinoids and the protease inhibitors used for HIV therapy and there may an exacerbation of the classic side effects of retinoids including xerosis, cheilitis, and paronychia.
Other agents that are effective in the management of psoriasis include cyclosporine (1.25 to 2.5 mg/kg/d (the latter for plaque or severe psoriasis) in two divided doses, whose levels may be augmented by the use of protease inhibitors for HIV infection), hydroxyurea (given typically at 1 g/day, with a dose range between 500 mg - 1500 mg, and active also against HIV, especially when combined with didanosine) [13]. A concern with hydroxyurea is dose-related bone marrow suppression, with thrombocytopenia and leucopenia particular concerns. Zidovudine itself has prolonged dose- dependent effects in the management of psoriasis, regardless of CD4 count, possibly due to its interference with DNA synthesis and as a result, keratinocyte proliferation [1,3].
A final group of agents, the biologics are known to be useful in plaque psoriasis but may some efficacy among the HIV-infected. Co-infections of HIV and mycobacterial infections limit their usefulness. The agents that are useful include etanercept, infliximab, efalizumab (which was withdrawn for safety reasons from the US and European markets, with an unexpectedly high rate of progressive multifocal encephalopathy from JC virus among users), adalimumab, alefacept, and ustekinumab. These agents are reviewed recently by Rustin [8].
Conclusion
Psoriasis in the HIV+ patient is highly variable, and requires consideration of a broad differential in the diagnosis. Our patient illustrates the need to maintain a high degree of awareness for the increased risk for psoriatic disease among HIV-infected patients. As with any HIV-related disease, optimizing anti-viral therapy is critical in improving outcomes, but careful consideration needs to be given to the interactions between various medications and the underlying immunosuppressive disease process in HIV-infected patients. Topical acitretin often provides relief in many of these patients without the potentially harmful side-effects of other moreimmunosuppressive regimens but itself is attendant with its own unique set of safety issues.
References
-
Morar N, Willis-Owen SA, Maurer T, Bunker CB (2010) HIV-associated psoriasis: pathogenesis, clinical features, and management. Lancet Infect Dis 10: 470-478.
-
Chandra A, Ray A, Senapati S, Chatterjee R (2015) Genetic and epigenetic basis of psoriasis pathogenesis. Mol Immunol 64: 313-323.
- Duvic M, Rios A, Brewton GW (1987) Remission of AIDS-associated psoriasis with zidovudine. Lancet 2: 627.
-
Wolfer LU, Djemadji-Oudjiel N, Hiletework M, Tebbe B, Husak R, et al. (1998) [HIV-associated psoriasis. Clinical and histological observations in 36 patients]. Hautarzt 49: 197-202.
-
Garbe C, Husak R, Orfanos CE (1994) [HIV-associated dermatoses and their prevalence in 456 HIV-infected patients. Relation to immune status and its importance as a diagnostic marker]. Hautarzt 45: 623-9.
-
Pallangyo KJ (1992) Cutaneous findings associated with HIV disease including AIDS: experience from Sub Saharan Africa. Trop Doct 22 Suppl 1: 35-41.
-
Morar N, Dlova NC, Mosam A, Aboobaker J (2006) Cutaneous manifestations of HIV in Kwa-Zulu Natal, South Africa. Int J Dermatol 45: 1006-1007.
-
Rustin MH (2012) Long-term safety of biologics in the treatment of moderate-to-severe plaque psoriasis: review of current data. Br J Dermatol 167 Suppl 3: 3-11.
-
Parisi R, Symmons DP, Griffiths CE, Ashcroft DM, Identification and Management of Psoriasis and Associated ComorbidiTy (IMPACT) project team (2013) Global epidemiology of psoriasis: a systematic review of incidence and prevalence. J Invest Dermatol 133: 377-385.
-
Jeong YS, Kim MS, Shin JH, Cho JK, Lee HI, et al. (2014) A Case of Severe HIV-Associated Psoriasis Successfully Treated with Acitretin Therapy. Infect Chemother 46: 115-119.
-
Buccheri L, Katchen BR, Karter AJ, Cohen SR (1997) Acitretin therapy is effective for psoriasis associated with human immunodeficiency virus infection. Arch Dermatol 133: 711-715.
-
Lee CS, Li K (2009) A review of acitretin for the treatment of psoriasis. Expert Opin Drug Saf 8: 769-779.
-
Lee ES, Heller MM, Kamangar F, Park K, Liao W, et al. (2011) Hydroxyurea for the Treatment of Psoriasis including in HIV-infected Individuals: A Review. Psoriasis Forum 17: 180-187.