Clinical Medical
Reviews and Case Reports
Mycotic Aneurysm of Native Renal Artery: Case Report and Review of the Literature
Arru Luca1*, De Magistris Luigi1, Arendt Charlie1, Pillet Jeanchristophe1, Azagra Juan Santiago1, Goergen Martine1 and Pouthier Dominique2
1Centre Hospitalier de Luxembourg, Department of General and Vascular Surgery, Luxembourg City, Luxembourg
2Centre Hospitalier de Luxembourg, department of Nephrology, Luxembourg City, Luxembourg
*Corresponding author: Luca Arru, Department of Generaland Vascular Surgery, Centre Hospitalier de Luxembourg, 4 rue E. Barblè, L-1210, Luxembourg city, Luxembourg, Tel: +3524411-6380, Fax: +3524411-6274, E-mail: lucaarrumd@gmail.com/arru.luca@chl.lu
Clin Med Rev Case Rep, CMRCR-2-051, (Volume 2, Issue 8), Case Report; ISSN: 2378-3656
Received: April 13, 2015 | Accepted: August 26, 2015 | Published: August 28, 2015
Citation:Luca A, Luigi DM, Charlie A, Jeanchristophe P, Santiago AJ, et al. (2015) Mycotic Aneurysm of Native Renal Artery: Case Report and Review of the Literature. Clin Med Rev Case Rep 2:051. 10.23937/2378-3656/1410051
Copyright: © 2015 Luca A, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Introduction: Renal native artery is an uncommon localisation of mycotic aneurysm that involves most frequently the artery of renal transplant in immuno-suppressed patients. Septic embolization often occurs during endocarditis and direct infection could complicate endovascular procedures with device placing.
Case report: We herein describe the case of a leaking mycotic aneurysm of the native renal artery (MANRA) in a renal transplanted patient, successfully treated by open aneurysmectomy and prolonged antibiotic therapy. Proximal localisation and history of scleroderma has never been described previously.
Discussion: Conventional treatment of MANRA includes prolonged antibiotic therapy usually associated with endovascular or surgical repair. The 13 cases present in literature are analysed specially regarding the treatment indications based on localisation of the lesions.
Conclusion: The presented case supports the attitude of an early and aggressive treatment of MANRA, in order to prevent rupture and avoid nephrectomy. Surgical or endovascular approach could be indicated according respectively to the proximal and distal localisation.
Keywords
Mycotic aneurysm, Infected aneurysm, Renal aneurysm, Leaking aneurysm, Native renal artery, Kidney transplantation, Scleroderma, Nephrectomy, Endovascular, Endovascular embolization
Introduction
Mycotic aneurysm is defined as a localised and irreversible destruction of arterial wall by bacterial infection. It results in a segmental dilatation of native vessel to at least one and a half times its normal diameter [1]. Renal artery localisation is uncommon. Physiopathology recognises permeation of septic emboli to the vasa vasorum, often during endocarditis or others causes of septicaemia [2]; infection of endovascular devices, such as renal stent, has also been described [3-6].
Lesions are often asymptomatic until rupture and, usually, clinical manifestations may include massive hematuria as observed in half of the reported cases [2-3,7-12]; it may also include flank pain [13], fever and sepsis [4,14].
Streptococcus, Staphylococcus and Pseudomonas are most common pathologic agents [2-4,6,11,15-17]. Conventional treatment includes prolonged antibio-therapy, possibly associated with endovascular or open surgical repair.
The following report describes a case of a leaking mycotic aneurysm: native renal artery localisation in presence of a renal heterotopic transplant has never been observed, neither has the association with a medical history of scleroderma.
Case Report
A 64-year-old man, presenting a history of diffuse type scleroderma with chronic renal failure, under immunosuppressive therapy for heterotopic renal transplantation, was admitted to the emergency room for a persistent lumbar pain and fever. Other relevant antecedents in the patient's medical history are: high blood-pressure, hypothyroidism, atrial fibrillation. The patient's immunosuppressive therapy consisted in Tacrolimus 1.5mg 1x/d and Mycophenolic acid 250mg 2x/d.
At the time of admission, vital signs and physical examination were normal except for pain on palpation of the left flank.
Blood tests revealed hyperleukocytosis (13.700) and C-reactive protein elevation (187 mg/l); serum creatinine was compatible with chronic renal allograft rejection (3.2mg/dL).
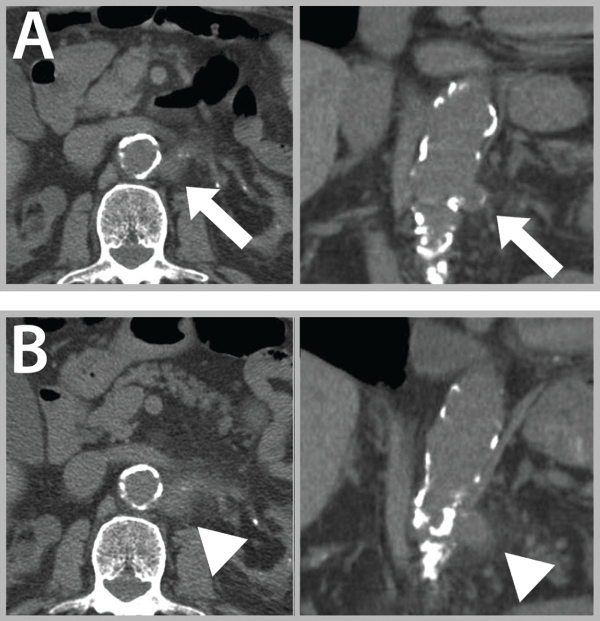
An abdominal-pelvic-CT scan without contrast injection was performed in order to preserve the renal function. It revealed a 2 cm aneurysm of the proximal left native renal artery without sign of rupture (Figure 1A). Wide spectrum antibiotics were introduced on the basis of clinical and radiological suspicion of infected vascular lesion (Cefuroxime 1,5 g 3/day and metronidazole 500 mg 3/day). Three days later, a new CT scan was performed to assess the worsening of symptoms. The second CT scan showed an increase in size and early signs of leakage (Figure 1B).
.
Figure 1: (A) Uncomplicated mycotic aneurysm (arrows) of native renal artery origin, renal bilateral atrophy.
(B) Leaking mycotic aneurysm of native renal artery origin (triangles).
View Figure 1
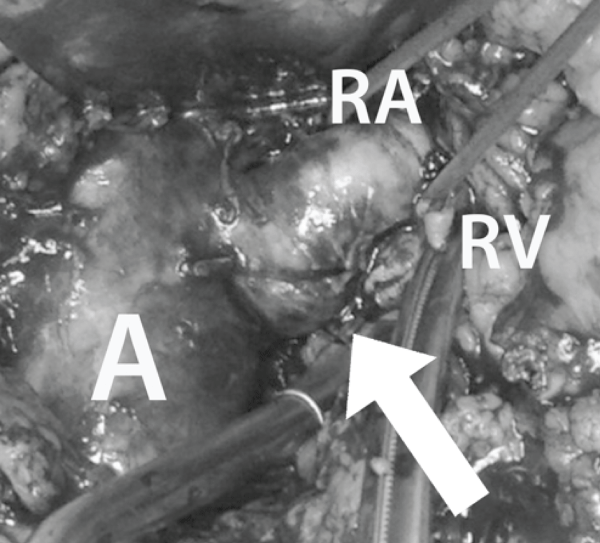
A surgical intervention was formally indicated. Laparotomy confirmed the radiological findings (Figure 2).
.
Figure 2: Leaking mycotic aneurysm (arrows) of native renal artery origin; A: Aorta; RA: Renal Artery; RV: Renal Vein (transsected).
View Figure 2
Following abdominal aorta clamping, above the renal arteries, aneurysmectomy with proximal aortic tangential resection and distal arterial ligation was performed. Arterial revascularisation wasn't necessary because of the renal atrophy. Primary closure of the aortic renal ostium was carried out by separate stitches.
Histological and bacterial examination confirmed the clinical suspicion of infected aneurysm with gram positive cocci germs; the culture of arterial biopsy revealed the growth of Streptococcus Agalactiae, sensitive to Cefturoxime. During the hospital stay, the multiple sets of blood cultures were negative.
Transthoracic echocardiography revealed no vegetation.
The post-operative course was uneventful; Cefturoxime was administered until the 20th post-operative dayand the patient was discharged at post-operative day 25.
No recurrence was found 21 months after surgery.
Discussion
Natural history of mycotic aneurysm of native renal artery (MANRA) is still unclear. The cases in literature often report infected aneurysm of the grafted renal artery in transplanted patients, in which a clear role of steroids and immunosuppressive treatments is documented [7].
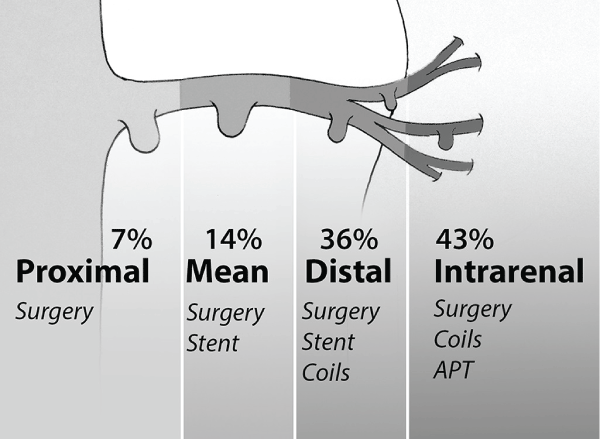
One interesting finding, in the reported case, is that the mycotic aneurysm occurred in the proximal native renal artery instead of transplant renal artery, as is its common occurrence. Furthermore, to our knowledge, the present case is the first report of proximal localisation. Figure 3 shows the distribution of localisation and the treatment based on our review of literature.
.
Figure 3: Distribution of localisation and treatment options; APT: antibiotic prolonged therapy.
View Figure 3
According to the literature, MANRA increases in size quickly and early leakage is frequent, as occurred in the presented case [2]. Acute evolution of MANRA seems to be more frequent and earlier than in atherosclerotic renal artery aneurysm in which, apart from occurrence during pregnancy, rupture is a rare event. The incidence of acute evolution of MANRA is under 5%, but the mortality can rise up to 80% [7-9].
Reported therapeutic options include targeted antibiotic therapy, often followed by endovascular or open surgical repair.
Literature doesn't support any evidence-based treatment: a complete review of the case report since 1977 is shown in table 1.
![]()
Table 1: Complete review of the literature since 1977.
View Table 1
The few cases of MANRA describes often occurred in patients affected by important co-morbidity such as lupus erythematosus, rheumatoid arthritis, diabetes and cardiac risk factors for endocarditis, that is present in the half of the patients[2,3,11-15].
Scleroderma has never been reported in association with MANRA. We cannot state that scleroderma is a risk factor for MANRA considering that the presented case reports the history of a patient under immunosuppressive treatment after renal transplantation.
Analysing the 13 cases reported in literature, localisation of the lesions seems to be decisive factor in the choice of treatment.
Emergency nephrectomy was necessary in 30% of cases reported in literature: in case of involvement of the mean trunk of renal artery or in presence of massive renal infarction [4,14,16,17]. Coils embolization was successful and uncomplicated in 80% of cases [2,3,13-15,16].
An early elective surgical or endovascular treatment may be recommended and the conservative medical management has to be reserved only for the very small intra-renal lesions [10-12].
Endovascular coils embolization, stenting exclusion or booth, appears to be preferred for the management of distal or intra-renal localisations, with a morbidity of 20% [16].
At present, proximal localisation of the MANRA and failure of endovascular procedure still requires open surgery; in the second case, nephrectomy is often mandatory.
Conclusions
The current literature supports the attitude of an early and aggressive treatment in order to prevent rupture and to avoid nephrectomy. Surgical or endovascular approaches may be indicated depending on the proximal and distal localisation, respectively.
References
-
Stengel A, Wolferth CC (1923) Mycotic (Bacterial) aneurysms of intravascular origin. Arch Intern Med (Chic) 31: 527-554.
-
Uno K, Kasahara K, Komatsu Y, Konishi M, Yoshihmoto E, et al. (2012) Rupture of renal mycotic aneurysm that developed during the treatment of streptococcal infective endocarditis and vertebral osteomyelitis. Intern Med 51: 1255-1258.
-
Hall CL, Cumber P, Higgs CMB, Chalmers AH (1987) Life threatening haemorrhage from a mycotic renal pseudoaneurysm treated by segmental artery embolisation. Br Med J 294: 1526.
-
Melamed N, Molad Y (2006) Spontaneous retroperitoneal bleeding from renal microaneurysms and pancreatic pseudocyst in a patient with systemic lupus erythematosus. Scand J Rheumatol 35: 481-484.
-
DeMaioribus CA, Anderson CA , Popham SS , Yeager TD , Cordts PR (1998) Mycotic renal artery degeneration and systemic sepsis caused by infected renal artery stent. J Vasc Surg 28: 547-550.
-
Stout CL, Glickman MH (2010) Renal artery stent infection and pseudoaneurysm management. Ann Vasc Surg. 24: 114.
-
Leonardou P, Gioldasi S, Zavos G, Pappas P (2012) Mycotic pseudoaneurysms complicating renal transplantation: a case series and review of literature. J Med Case Rep 6: 59.
-
Forbes TL, Abraham CZ, Pudopakkam S (2001) Repair of rupted giant renal artery aneurysm with kidney salvage. Eur J Vasc Endovasc Surg 22: 278-9.
-
Tham G, Ekelund L, Herrlin K, Lindstedt EL, Olin T, et al. (1983) Renal artery aneurysms. Natural history and prognosis. Ann Surg 197: 348-352.
-
Ganem EJ, Wallwork DW, Irving W (1977) Intrarenal mycoticaneurysm. Urology 9: 667-669.
-
DuBrow RA, Patel SK (1981) Mycotic aneurysm of the renal artery. Radiology 138: 577-582.
-
Nosher JL, Needell GS, Bialy G, Zatina M (1990) Catheter occlusion of a mycotic renal artery aneurysm with cure of associated renovascular hypertension. Cardiovasc Intervent Radiol 12: 310-312.
-
Rabellino M, García-Nielsen L, Zander T, Baldi S, Llorens R, et al. (2011) Stent-assisted coil embolization of a mycotic renal artery aneurysm by use of a self-expanding neurointerventional stent. Cardiovasc Intervent Radiol 34: 109-112.
-
Chiu KM, Lin TY, Chen JS, Chu SH (2008) Rupture of renal artery aneurysm due to Salmonella infection in a patient with systemic lupuserythematosus. Lupus 17: 135-138.
-
Sliker CW (2005) Renal artery mycotic pseudoaneurysm: Diagnosis with multi-detector computed tomographic angiography. Emerg Radiol. 11: 250-251.
-
Ohebshalom MM, Tash JA, Coll D, Su LM, Schlegel PN (2001) Massive hematuria due to right renal artery mycotic pseudoaneurysm in a patient with subacute bacterial endocarditis. Urology 58: 607.
-
Rabinowitz R, Schillinger JF, Churchill BM (1979) Leaking mycotic aneurysm of renal artery in a child. J Urol 121: 84-85.





