Trauma Cases and Reviews
Liver Injury Associated with Chest Compression in Cardiopulmonary Arrest Patients
Takeshi Nishimura1*, Ayana Okamoto1, Noritomo Fujisaki1, Yoshiyuki Orihara2, Ten Saita2, Kunihiro Shirai1, Isamu Yamada1, Atsunori Nakao1 and Joji Kotani1
1Department of Emergency, Hyogo College of Medicine, Japan
2Department of Cardiology, Hyogo College of Medicine, Japan
*Corresponding author: Takeshi Nishimura, MD, Department of Emergency, Disaster and Critical Care Medicine, Hyogo College of Medicine, 1-1 Mukogawa, Nishinomiya, Hyogo 663-8501, Japan, Tel: +81-798-45-6514, Fax: +81-798-45-6813, E-mail: kogushi1@msn.com
Trauma Cases Rev, TCR-2-024, (Volume 2, Issue 1), Review Article; ISSN: 2469-5777
Received: December 20, 2015 | Accepted: January 09, 2016 | Published: January 12, 2016
Citation: Nishimura T, Okamoto A, Fujisaki N, Orihara Y, Saita T, et al. (2016) Liver Injury Associated with Chest Compression in Cardiopulmonary Arrest Patients. Trauma Cases Rev 2:024. 10.23937/2469-5777/1510024
Copyright: © 2016 Nishimura T, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Introduction: Although chest compression is an effective emergency maneuver for cardiopulmonary arrest (CPA), it carries the risk of complications such as rib fracture, sternal fracture, and organ injury.
Methods: We summarized thirteen case reports of CPA patients presenting with liver injury associated with CPR including chest compression reported between 1993 and 2015 in Japan. Five were male and eight were female; their ages ranged from 31 to 83 years old. Unfractionated heparin was administered to ten patients as a treatment for embolism or indication of veno-arterial extra corporeal membrane oxygenation (VA-ECMO). Twelve cases survived surgery or non-surgical management, but one could not survive with non-surgical management.
Case: Two cases of CPA due to pulmonary thromboembolism were admitted to our department. Spontaneous circulation was restored due to cardiopulmonary resuscitation (CPR), including chest compression. After use of an anticoagulant drug and indication of VA-ECMO, contrast-enhanced computed tomography (CE-CT) revealed infra-diaphragmatic liver injury in both cases. One patient could survive with damage control surgery (DCS), while the other patient could not survive with non-surgical management.
Conclusion: Patients with hemorrhagic diathesis are at critical risk for complications from chest compression. Aggressive treatments, including DCS, might be effective, even for patients treated with anticoagulant drugs.
Keywords
Chest compression, Liver injury, Pulmonary arterial embolization
Introduction
On one hand, chest compression is a useful, life-saving emergency technique for patients suffering from cardiopulmonary arrest (CPA). On the other hand, complications associated with chest compression, such as rib fracture, sternal fracture, and thoracic and abdominal organ injuries, have been widely reported.
We reviewed and summarized fifteen cases associated with liver injury following chest compression for CPA reported in Japan. In some cases, cardiopulmonary resuscitation (CPR) was administered by medical assistants such as doctors or medical technicians, but in other cases, CPR was given by bystanders. Five patients were male and eight were female, with ages ranging from 31 to 83 years old. Unfractionated heparin was administered to ten patients as a treatment for embolism or indication of veno-arterial extra corporeal membrane oxygenation (VA-ECMO), and other cases suffered from amniotic fluid embolisms or disseminated intravascular coagulation (DIC). All cases had problems with refractory coagulopathy. While twelve cases survived surgery or non-surgical management, one could not survive with non-surgical management.
Two patients were admitted to our emergency department due to pulmonary thromboembolism following CPA. The patients received CPR with chest compression and adrenaline. VA-ECMO was administered and spontaneous circulation was restored. After resuscitation, contrast-enhanced computed tomography (CE-CT) revealed massive hemorrhages in the abdominal cavities and liver injuries induced by chest compression. One case could survive with damage control surgery (DCS), but the other case could not survive with non-surgical management.
When we encounter patients with unstable vital signs after return of spontaneous circulation, complications from chest compression such as organ injury may be hidden. Anti-coagulant drugs could turn a minor fissure into a severe hemorrhage. As emergency clinicians, we should be aware of the complications from chest compression and the risk of anti-coagulant therapy; hence, we examined and evaluated these cases in Japan.
Japanese Literature Review
Method and baseline characteristics
On September 2015, we authors searched for all reported cases of liver laceration associated with chest compression on several databases including English and Japanese literature. First author performed screening with databases. We examined cases reported from 1993 to 2015 in Japan, which were extracted from the Ichushi and Pubmed library by searching for the keywords "chest compression", "complication" and "liver injury" (Table 1). We excluded traumatic CPA patients. Thirteen cases were found and examined in detail. We examined data related to cardiac arrest such as patient age, sex, initial electro cardiogram (ECG) rhythm, CPR information, usage of anticoagulant drugs, liver injury site, treatment, and prognosis. Time from cardiac arrest to return of spontaneous circulation was unknown. Five cases were male and eight were female, with ages ranging from 31 to 83 years old. Median age was unknown because one case was in their fifties.
![]()
Table 1: This table shows the review of cases with liver injury caused by chest compression including our cases.
View Table 1
Diagnosis
Almost all cases were diagnosed as liver injuries associated with chest compression or intra-abdominal hemorrhage by ultrasound or CE-CT. Five cases were defined as pulmonary thromboembolism leading to CPA; therefore, unfractionated heparin was administered as a treatment after resuscitation and diagnosis. Dosage of unfractionated heparin was unknown. Our two cases were also diagnosed as pulmonary thromboembolism after resuscitation and indication of ECMO. In both cases 5,000 units of unfractionated heparin were administered rapidly following diagnosis.
Five cases had suffered from acute coronary syndrome (ACS), while the other cases involved amniotic embolism, cardiac sarcoidosis, and hyperkalemia during esophageal surgery, respectively.
Initial ECG and CPR performer
All cases were assessed as CPA and CPR was administered immediately. Five cases (four of the ACS cases and one cardiac sarcoidosis case) revealed ventricular fibrillation (VF) in ECG. Two cases with pulmonary thromboembolism and hyperkalemia showed pulseless electrical activity (PEA). Initial ECG was unclear in the other cases because the data was not included in the publications.
CPR, including chest compression, was performed by bystanders in four cases. Medical technicians and doctors performed CPR in the other seven cases. In the one case admitted with poor prognosis, CPR was administered by a medical technician. Because of liver injury associated with chest compression, Meron et al. reported no differences between CPR performed by medical professionals and CPR performed by lay bystanders secondary to visceral hemorrhage. In our two cases, medical technicians and doctors experienced in CPR performed chest compression. Although some previous reports have demonstrated that inappropriate chest compression site had the potential for complications, results of this review cannot indicate no association.
Hemostasis and anti-coagulant drugs
Unfractionated heparin was used to treat embolization or maintain ECMO in ten cases. Dosage was unknown in each case due to no statement in manuscript. In both of our cases, 5,000 units of unfractionated heparin was given immediately following diagnosis and activated clotting time was adjusted from 150 to 200 seconds to keep ECMO running smoothly. The other two cases were characterized as obstetric DIC and hemorrhagic DIC, respectively. Transfusion, including fresh frozen plasma, was the key treatment.
Location of liver injury
CT or intra-operative findings showed subcapsular hematoma of the liver in seven cases. Liver fissure appeared in six cases at the left lobe, both lobes and the falciform ligament. Some cases were complicated, with multiple liver injuries.
According to anatomical features, the left lobe or falciform ligament of the liver are easy to injure because these sections are located immediately below the xiphoid process. Therefore, compression of the rib cage leads to liver damage around the ligament and left lobe. Gallbladder injury was rarely reported at the injury site, whereas hematoma or bleeding arising from the nearby falciform ligament or lateral site of liver were cited more frequently. A damaged posterior segment of the liver due to chest compression as in our cases had never been reported previously in the literature. Although some reasons such as inadequate positioning for chest compression, right heart strain resulting from pulmonary hypertension, and damage from the diaphragm were hypothesized, no obvious cause was established.
Treatment and Results
In a previous study in 1993, liver injury was treated by hepatic resection. Since 2010, as the concept of trauma surgery has become more widespread, DCS, including peri-hepatic gauze packing, was performed in four cases. Our surgical team also administered DCS in case 2. Although inadequate gauze packing contributed to re-admission to the operating room, tertiary surgery allowed closure of the patient's abdomen after a 40 hour interval from onset.
Our one case could not be successfully treated with non-surgical management. The fact that all cases with surgical procedures could survive with DCS indicates that early indication of surgical procedure might have the potential to dramatically change the prognosis of a patient with liver injury treated with anti-coagulant agents or DIC associated with chest compression.
Case Presentation
We present 2 cases of liver injury secondary to chest compression which we recently experienced in addition to the Japanese case series.
Case 1
A 78-year-old woman was admitted to our emergency department complaining of losing consciousness. She was not feeling well since the beginning of the day. She was losing consciousness when standing up, so her family requested an emergency room visit. Her past medical history included vertebral compression fracture, hypertension, rheumatoid arthritis, and diabetes mellitus. On arrival, her vital signs were as follows: Glasgow Coma Scale score (GCS) was E1V1M1, and her pupils were not dilated with rapid light reflex; heart beat was 130 beats per minute; blood pressure was 95/60 mmHg; respiratory rate was 16 breaths per minute; arterial blood saturation was 80%; and pulmonary artery oxygen 56 mmHg with 10 liters of oxygen. Echocardiography demonstrated dilation of the right ventricle and exclusion of the left ventricle. These findings supported the diagnosis of pulmonary thromboembolism, so the patient was transferred to the CT suite. Suddenly, her vital signs became unstable during radiography scanning and she had PEA. During chest compressions and infusion of adrenaline, VA-ECMO was administered under radiography. After a 10 minute interval of PEA, her spontaneous circulation was restored. Angiography revealed an embolism at the pulmonary artery, which was diagnosed as pulmonary thromboembolism. The patient received an infusion of 5,000 units of unfractionated heparin following hypothermia therapy in the intensive care unit (ICU).
Twenty-four hours after admission, because of worsening anemia and instability of circulation, contrast-enhanced CT (CE-CT) revealed intra-abdominal fluid collection, infra-diaphragmatic liver injury, and extravasation at the injury site. Without an episode of trauma and abdominal fluid collection at the emergency room, it was hypothesized that chest compression induced the lethal liver injury during CPR. Although conservative therapy was continued including transfusion, she died on the third day after hospitalization.
Case 2
An 81-year-old woman had been hospitalized due to surgery for scoliosis. A few days after the operation, when she was practicing walking in the rehabilitation room, she suddenly fainted. Our rapid response team soon arrived at the rehabilitation room and recognized her unstable vital signs and lack of coma. She was transferred to the emergency room with the following vital signs: GCS: E3V1M6; heart rate: 79 beats per minute; blood pressure: 73/46 mmHg; arterial saturation under 12 liter oxygen: 99%. Electrocardiogram revealed a complete right bundle branch block of the pulmonary artery, as well as something floating at the right ventricle and dilation of the right ventricle. D-dimer was remarkably elevated (D-dimer: 45.0 mcg/ml).
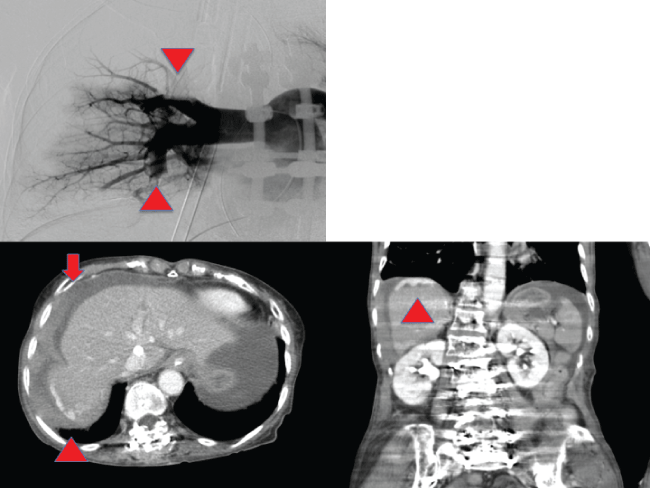
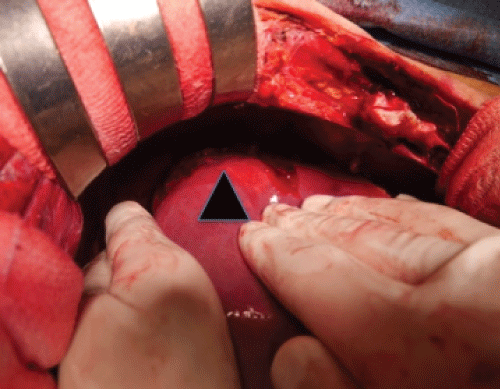
Unexpectedly, she fell into PEA and CPR was started. Chest compressions contributed to the start of VA-ECMO under radiography. Arterial angiography revealed thromboembolism of the right upper and lower pulmonary artery branches. The patient received an immediate infusion of 5,000 units of unfractionated heparin. We diagnosed her with pulmonary thromboembolism due to deep vein thrombosis resulting from spinal surgery (Figure 1). Although her vital signs stabilized, VA-ECMO could not work smoothly due to hypovolemic state. Ultrasonography revealed an echo-free space in the abdominal cavity. CE-CT revealed an intra-abdominal fluid collection and liver injury at the posterior area (Figure 1). No bone fractures near the liver were found. Transfusion could not improve the patient's unstable state, and angiography could not detect the injured vessels; therefore, we performed laparotomy for hemostasis. Massive bloody ascites were found and the infra-diaphragmatic area of liver was found to be damaged (Figure 2). We decided to perform DCS to control massive hemorrhage. Peri-hepatic gauze packing and transient abdominal closure were performed. In the ICU, the patient was treated with a massive transfusion. Despite transfusion and warming, hemorrhage was not controllable, and therefore the patient was re-admitted to the operating room for confirmative peri-hepatic gauze packing. With stabilization of her vital signs, abdominal wall closure was performed after a 40 hour interval from laparotomy. After tracheotomy at 22 hospitalized days, continuous rehabilitation could contribute to improvement of her of activities of daily living.
.
Figure 1: (Left upper) Arrowheads show obstruction of the upper and lower branches of the pulmonary artery in right pulmonary angiography; (Left lower) Arrow demonstrates fluid collection in the abdominal cavity in CE-CT. Arrowhead shows extravasation at the posterior segment in CE-CT at axial figure; (Right lower) Arrowhead shows extravasation at the posterior segment in CE-CT at coronal figure.
View Figure 1
.
Figure 2: Intra operative findings revealed liver injury at the posterior segment (black arrowhead). Continuous bleeding flowed from the injury site.
View Figure 2
Discussion
The American Heart Association recommends that CPR be used to potentially save the lives of patients who suffer from CPA [1]. Even non-medical staff can perform CPR without any device. However, while CPR skills can be learned by ordinary people, complications related to chest compression such as rib and sternal fractures have been reported, occurring at a rate of approximately 21-67% [2]. Liver injury happens rarely (at a rate of about 2%), but when these injuries do occur, they can be lethal and life-threatening [3,4]. Particularly, CPR with anti-coagulant drugs as a treatment might change a minor injury to a life-threatening situation.
In our two cases, paramedics and doctors with CPR expertise performed chest compression. CE-CT revealed only liver injury without any other visceral injuries. Clinical signs, including instability of circulation, progression of anemia and dropping hematocrit, and swelling of the abdominal wall, indicate hemorrhage in the abdominal cavity. Ultrasonography or CE-CT might detect an injury associated with chest compression. According to anatomical features, hematoma tends to arise around the falciform ligament or the lateral segment of the liver.
In case 1, stabilization of the circulatory system was gained with VA-ECMO. Hypothermia treatment was implemented because of cardiac arrest about ten minutes after ICU admission. On the next day, abdominal fluid collection accounted for the exacerbating anemia. Focused assessment of sonography revealed no fluid collection in the abdominal cavity immediately following resuscitation; therefore, we diagnosed the patient with visceral injury resulting from chest compression. We assumed that anti-coagulant drugs and hypothermia might promote a visceral minor injury to a lethal condition. Despite rapid and massive transfusion, the patient's blood pressure deteriorated gradually. Her family did not want more invasive treatment, so the patient died on post-admission day three.
In case 2, VA-ECMO did not work smoothly due to hypovolemia. CT revealed fluid collection in the abdominal cavity; hence, we performed laparotomy to stop the bleeding immediately. The first surgery could not confirmatively work; therefore, we re-attempted peri-hepatic gauze packing. The abdomen was closed after 60 hours from onset. Since angiography could not detect an injured vessel, we decided to perform surgery for hemorrhage due to venous injury. Early recognition of hemoperitoneum and early decision to perform laparotomy, based on our experience with case 1, rescued the patient in case 2.
Few cases of patients able to survive after liver injury due to chest compression have been reported [5]. We summarized cases of liver injury associated with chest compressions in Japan (Table 1). Yamasaki et al. described successful cases associated with pulmonary embolization and liver injury in Japan [6]. Matsukawa et al. reported a pregnant woman with refractory DIC resulting from an amniotic fluid embolism [7]. A case of cardiac sarcoidosis leading to liver injury with chest compression was also described [8].
Although twelve of thirteen cases survived, one case could not survive secondary to refractory hemorrhage. While four of twelve successful cases had angiography, a bleeding point was not found in three cases. Four cases were treated with transfusion and non-surgical management. Seven cases had laparotomy due to unstable vital signs. Of seven cases, four cases were treated with DCS, two cases were treated with electrocoagulation, and one was treated with left lobe resection. Hence, all cases suffered refractory coagulopathy, and a strategy including DCS in the early phase was recommended.
Sakamoto et al. compared interventional radiology and laparotomy treatments in traumatic cases with liver injury [9]. However, cases involving anti-coagulant drugs, ECMO, obstetric DIC, and similar situations are more complicated. Although a systematic review is difficult to establish, early recognition of a patient's condition and deciding on a strategy including laparotomy and DCS in the early phase have the potential to improve a patient's prognosis, even when they are treated with anti-coagulant agents and coagulopathy.
Conclusion
We examined and evaluated cases of liver injury associated with chest compression for CPA. Our experiences and review reveal that CPR has the unintended potential to result in liver injury for patients with these severe conditions. In cases of exacerbating anemia with unstable vital signs after resuscitation following chest compression, early recognition of visceral injury and appropriate choice of treatment might be able to dramatically improve a lethal condition.
Conflict of Interests
No conflicts of interest were declared.
References
-
Mancini ME (2011) Working together, nurses can make a difference in resuscitation outcomes: an update on the American Heart Association's 2010 guidelines for cardiopulmonary resuscitation and emergency cardiovascular care. Jpn J Nurs Sci 8: 7-10.
-
Hashimoto Y, Moriya F, Furumiya J (2007) Forensic aspects of complications resulting from cardiopulmonary resuscitation. Leg Med (Tokyo) 9: 94-99.
-
Krischer JP, Fine EG, Davis JH, Nagel EL (1987) Complications of cardiac resuscitation. Chest 92: 287-291.
-
Meron G, Kurkciyan I, Sterz F, Susani M, Domanovits H, et al. (2007) Cardiopulmonary resuscitation-associated major liver injury. Resuscitation 75: 445-453.
-
Monsuez JJ, Charniot JC, Veilhan LA, Mougue F, Bellin MF, et al. (2007) Subcapsular liver haematoma after cardiopulmonary resuscitation by untrained personnel. Resuscitation 73: 314-317.
-
Yamasaki M, Misumi H, Abe K, Kuwauchi S, Ito J, et al. (2014) Massive Pulmonary Embolism with liver injury associated with chest compressions during Cardiac Resuscitation. Ann Thorac Surg 98: 310-311.
-
Hiroyoshi M, Shigehiro S, Norihisa T, Yasuhiro F, Satoshi O, et al. (2010) A case of liver injury caused by cardiopulmonary resuscitation of a woman immediately post-partum. Journal Japan Surgical Association 71: 2402-2405.
-
Kuwata H, Muyazaki Y, Adachi T (2011) Intra-abdominal bleeding from liver injury after cardiopulmonary resuscitation. J Jpn Soc Intensive Care Med 18: 415-416.
-
Sakamoto Y, Mashiko K (2008) Treatment Strategy for Hepatic Injury. Journal of Abdominal Emergency Medicine 28: 803-807.





