Obstetrics and Gynaecology Cases - Reviews
Retained Placenta Accreta Presenting as Secondary Postpartum Hemorrhage
Yilun Koethe1, Gabrielle A Rizzuto2 and Maureen P Kohi1*
1Department of Radiology and Biomedical Imaging, University of California, USA
2Department of Pathology and Laboratory Medicine, University of California, USA
*Corresponding author: Maureen P. Kohi, Department of Radiology and Biomedical Imaging, University of California, 505 Parnassus Ave., M-361, San Francisco, CA 94143, USA, Tel: 415-290-5119, Fax: 415-476-0616, E-mail: maureen.kohi@ucsf.edu
Obstet Gynecol Cases Rev, OGCR-2-035, (Volume 2, Issue 3), Case Report; ISSN: 2377-9004
Received: April 14, 2015 | Accepted: April 30, 2015 | Published: May 02, 2015
Citation: Koethe Y, Rizzuto GA, Kohi MP (2015) Retained Placenta Accreta Presenting as Secondary Postpartum Hemorrhage. Obstet Gynecol Cases Rev 2:035. 10.23937/2377-9004/1410035
Copyright: © 2015 Koethe Y, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Background: Abnormal placentation and retention of products of conception (RPOC) are common causes of postpartum hemorrhage.
Case: A 37-year-old gravida 4 para 1 woman with history of Asherman�s syndrome from prior dilatation and curettage presented in spontaneous labor. Following normal vaginal delivery, portions of the placenta were retained and had to be manually extracted. Two weeks following delivery, she returned to the hospital with persistent vaginal bleeding. Ultrasound and magnetic resonance imaging demonstrated an endometrial mass with vascularity. During her hospital evaluation, she continued to bleed and ultimately underwent a total abdominal hysterectomy.
Results: Gross and histologic evaluation of the uterus demonstrated retained placenta accreta.
Conclusions: Our case highlights that while rare, retained invasive placenta can present as a cause of secondary PPH, particularly in patients with Asherman�s syndrome.
Keywords
Retained products of conception, Invasive placenta, Postpartum hemorrhage, Vaginal bleeding, Hysterectomy, Asherman�s syndrome
Introduction
Abnormal placentation and retention of products of conception (RPOC) are two common etiologies of postpartum hemorrhage (PPH) [1]. Invasive placenta is the invasion of chorionic villi across the confinement of decidua basalis, and the adherence of the placenta on to the uterine myometrium. Its incidence has increased to 3 per 1000 deliveries over the past decade, and is strongly associated with placenta previa and prior uterine surgeries [2]. Trauma to the endometrium from prior uterine interventions such as dilatation and curettage (D&C), cesarean delivery, or myomectomy may result in Asherman�s syndrome, a condition which produces partial or complete obliteration of the uterine cavity and/or cervical canal and may result in menstrual abnormalities, dysmenorrhea, infertility, recurrent pregnancy loss, and postpartum hemorrhage [3].
RPOC is the incomplete expulsion of placenta or trophoblastic tissue after birth, with an incidence of 3%-5% after routine vaginal delivery [4]. While both invasive placenta and RPOC commonly cause postpartum bleeding [1], RPOC is typically managed by manual extraction or D&C [5,6], and invasive placenta is treated through conservative management or hysterectomy [7].
Ultrasound (US) is the leading imaging modality for diagnosing both invasive placenta antenatally and suspected RPOC postnatally. Magnetic resonance imaging (MRI) can be an useful adjunct to US to outline the extent of placental invasion or make the diagnosis when US is inclusive [8,9]. Failure to detect invasive placenta can lead to life-threatening bleeding, with an estimated maternal mortality of 6%-7% [2]. Similarly, failure to diagnose RPOC can cause persistent primary and secondary PPH [9].
Herein, we present a case of retained invasive placenta that was undetected antenatally and in the immediate postpartum course that ultimately resulted in hysterectomy.
Case Report
A 37-year-old gravida 4 para 1 woman with a history of factor XI deficiency presented at 39 weeks gestation in spontaneous labor. Her obstetrical history was significant for two prior spontaneous abortions and a normal spontaneous vaginal delivery which was complicated by retained placenta (without an invasive component) for which she underwent a D&C. A year after the birth of her child, she had difficulty conceiving and was diagnosed with Asherman�s syndrome. However, without any intervention for her Ahserman�s syndrome, she was able to conceive spontaneously approximately 2 years after the birth of her first child. Her antepartum US demonstrated a normal anterior placenta without any evidence of previa or invasion (Figure 1). Her labor progressed normally and she delivered a female infant weighing 2940 grams with excellent Apgar scores of 9 and 10, at 1 and 5 minutes, respectively. The estimated blood loss for the delivery was 400mL.
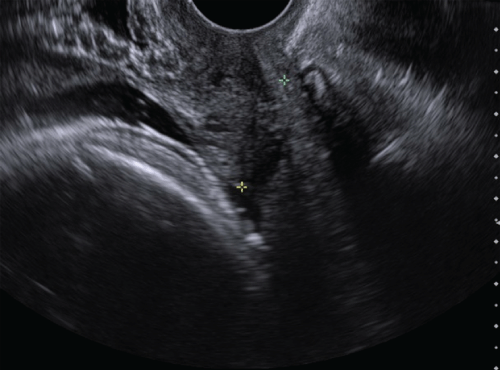
.
Figure 1: Greyscale ultrasound demonstrates a normal anterior placenta without evidence of previa and closed cervix.
View Figure 1
Following the delivery, portions of the placenta were retained and were extracted manually and with banjo curettage under US guidance. The estimated blood loss for the D&C was 200mL. Her immediate postpartum course was uncomplicated, and she was discharged on postpartum day 2.
Two weeks later, she returned to the hospital with persistent vaginal bleeding. On exam, her uterus was 12 weeks in size, and her quantitative human chorionic gonadotropin level was 14 IU/L. Her nadir hemoglobin and hematocrit were 8.6g/dL and 25.7% with a prolonged activated partial thromboplastin time of 66.6 seconds, consistent with her factor XI deficiency. During her evaluation, she became hemodynamically unstable with hypotension and tachycardia. She received two units of packed red blood cells and 10 units of fresh frozen plasma. Greyscale US illustrated a 3.5cm heterogeneous and vascular endometrial mass (Figure 2), with predominantly peripheral enhancement on color Doppler (Figure 3).
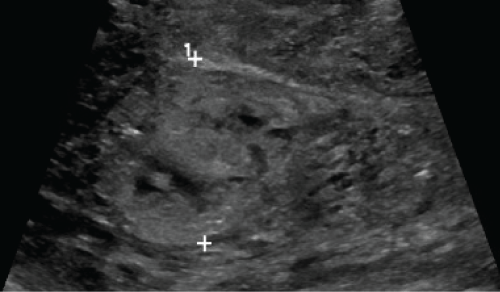
.
Figure 2: Greyscale ultrasound demonstrates a 3.5 cm heterogeneous mass in the endometrial cavity.
View Figure 2
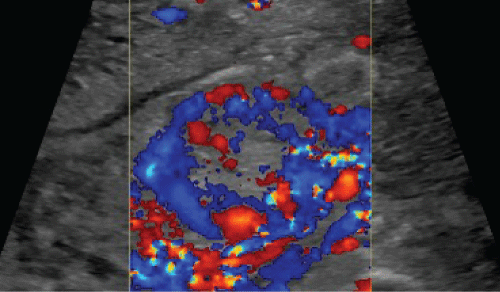
.
Figure 3: Color Doppler ultrasound demonstrates marked predominant peripheral vascularity of the endometrial mass.
View Figure 3
MRI pelvis demonstrated a 2.9cm x 3.6cm x 3.0cm heterogeneous soft tissue mass within the endometrial cavity, extending into the myometrium in the right posterolateral aspect of the fundus and the body (Figure 4). The leading diagnosis was retained placenta. In light of her ongoing vaginal bleeding and hemodynamic instability, she was taken to the operating room and underwent a total abdominal hysterectomy. Her postoperative course was uncomplicated and she was discharged on postoperative day 4.
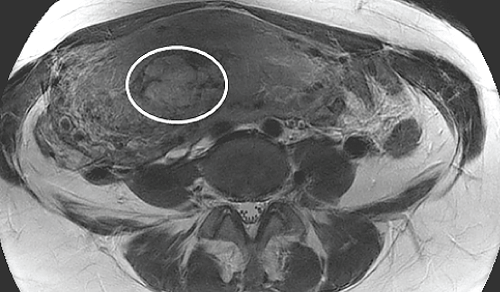
.
Figure 4: Axial T2-eighted MR image of the pelvis demonstrates a 2.9cm x 3.6cm x 3.0cm, heterogeneous soft tissue mass within the endometrial cavity that extends into the right posterolateral aspect of the myometrial fundus and body (white circle).
View Figure 4
Results
Gross pathology of the uterus demonstrated a 7cm x 5cm x 1.5cm soft tissue mass in the endometrial cavity which had invaded the myometrium to a depth of 1cm (Figure 5a). Microscopically, degenerating and necrotic chorionic villi were adherent to the myometrium, without intervening endometrium, consistent with placenta accreta (Figure 5b).
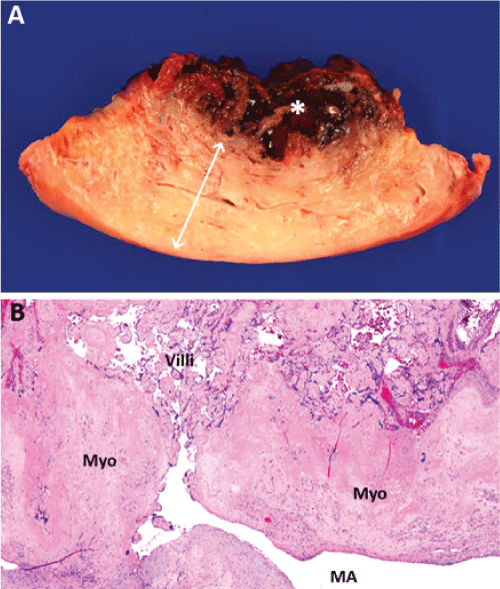
.
Figure 5: Gross and microscopic evidence of placenta accreta on pathology. (a) Gross photograph of representative full thickness section through uterine wall showing myometrium (rough boundary delineated by white arrow) and soft tissue mass (white asterisk, *). (b) Hematoxylin & eosin stain showing degenerating placental villi adherent to myometrium (Myo) without intervening endometrium. Note that placenta villous parenchyma appears directly perfused by a large maternal myometrial artery (MA). Scale bars: (a) 1 cm, (b) 500 microns.
View Figure 5
Discussion
Abnormally invasive placenta is a potentially life-threatening complication of pregnancy, for which high index of suspicion should be held, even in cases with normal antepartum imaging. The mechanism by which invasive placenta occur is unclear, but poor decidualization from prior mechanical trauma or primary deficiency are thought to play a role [10]. By that logic, prior uterine procedures such as D&C and their associated complications including Asherman�s syndrome, have been implicated to cause invasive placenta. This patient�s history of Asherman�s syndrome should raise the clinician�s suspicion for developing placental invasion.
Obstetric US is the gold standard for the antenatal diagnosis of invasive placenta, and has a sensitivity ranging between 78%-93%. Sonographic findings that suggest placental invasion include vascular lacunae within the placenta, loss of hypoechoic retroplacental zone, abnormal color Doppler surrounding the myometrium, and reduced myometrial thickness [8]. However, thus far, there is no validated criterion that weighs and combines individual imaging parameters to predict the likelihood of placenta invasion [11]. Furthermore, these studies have considerable bias in their protocol, as a single clinician reviewed the images and typically only high-risk patients were enrolled. In fact, when six experienced clinicians, all blinded to clinical status, reviewed the US of invasive placenta in a more recent study, the sensitivity decreased to 53.3%, with considerable inter-observer variations [12]. Therefore, a potential pitfall of clinical practice, as underscored by this case, is to rely on a negative US for ruling out invasive placenta without considering all risks and mechanisms of decidua disruption.
RPOC occurs more frequently than invasive placenta, but is uncommon to present as a remnant after its initial extraction and as delayed bleeding. Secondary PPH is defined as excessive blood loss 24 hours to 6 weeks postpartum, and occurs among 0.5%-2% of women in developed countries [13]. It is difficult to distinguish RPOC from other causes of secondary PPH such as gestational trophoblastic disease or endometritis by clinical history alone. US can differentiate these conditions, and is the leading diagnostic imaging modality for secondary PPH. Grey scale US suggests the presence of RPOC when a thickened endometrial echo complex or an endometrial or intrauterine mass is seen, with sensitivity of 80% and 79% respectively. Color Doppler enhances the diagnostic accuracy of US by differentiating RPOC from nonvascular masses or clots, and cautions obstetricians in cases of high vascularity [9]. MRI and hysteresocopy may also serve as adjuncts to US in determining the cause of the PPH [14].
In this case, remnant RPOC was a less-likely candidate for this patient�s bleeding given her inherited coagulopathy that predisposes her to PPH, her lack of fevers and pain, and her immediate postpartum extraction of RPOC. This further advocates for US imaging in all cases of suspected RPOC.
Invasive placenta is typically diagnosed antenatally or during delivery, and is less commonly associated with secondary PPH and RPOC. When RPOC is diagnosed after it has been initially extirpated during immediate postpartum period, focal placental invasion with localized adherence to the uterus should be considered, as demonstrable in this case. US and MRI can detect invasive placenta in RPOC by visualizing the presence and the extent of vascular soft tissue invasion into the myometrium. On MRI, RPOC presents as an enhancing soft-tissue mass within the endometrial canal. Features of local invasion include abnormal uterine bulging and extension into the myometrium. However, heterogeneous signal enhancement may vary depending on the degree of hemorrhage and tissue necrosis [8,9]. The treatment options for RPOC with invasive placenta include conservative management in patient who prefer uterine preservation or hysterectomy. D&C or hysteroscopic removal of invasive placenta can lead to massive hemorrhage, requiring emergent hysterectomy and massive transfusion [7]. Therefore, should a patient with a history of uterine procedure have remnant RPOC, clinicians should proceed with caution, and refer the patient to a multidisciplinary care center that can manage these highly morbid complications [12].
Conclusions
This case report highlights an uncommon case of retained invasive placenta presenting as secondary PPH. It demonstrates the value of diagnostic imaging for suspected cases of RPOC. In particular, US and MRI can distinguish placental invasion from routine RPOC, drastically alter the treatment, and avoid unexpected maternal mortality and morbidity from undetected invasive placenta.
References
-
Katz MD, Sugay SB, Walker DK, Palmer SL, Marx MV (2012) Beyond hemostasis: spectrum of gynecologic and obstetric indications for transcatheter embolization. Radiographics 32: 1713-1731.
-
Publications Committee, Society for Maternal-Fetal Medicine, Belfort MA (2010) Placenta accreta. Am J Obstet Gynecol 203: 430-439.
-
March CM (2011) Asherman's syndrome. Semin Reprod Med 29: 83-94.
-
Epperly TD, Fogarty JP, Hodges SG (1989) Efficacy of routine postpartum uterine exploration and manual sponge curettage. J Fam Pract 28: 172-176.
-
Rosenstein MG, Vargas JE, Drey EA (2014) Ultrasound-guided instrumental removal of the retained placenta after vaginal delivery. Am J Obstet Gynecol 211: 180.
-
Ben-Ami I, Melcer Y, Smorgick N, Schneider D, Pansky M, et al. (2014) A comparison of reproductive outcomes following hysteroscopic management versus dilatation and curettage of retained products of conception. Int J Gynaecol Obstet 127: 86-89.
-
Sentilhes L Goffinet F, Kayem G (2013) Management of placenta accreta. Acta Obstet Gynecol Scand 92: 1125-1134.
-
Baughman WC, Corteville JE, Shah RR (2008) Placenta accreta: spectrum of US and MR imaging findings. Radiographics 28: 1905-1916.
-
Sellmyer MA Desser TS, Maturen KE, Jeffrey RB Jr, Kamaya A (2013) Physiologic, histologic, and imaging features of retained products of conception. Radiographics 33: 781-796.
-
Khong TY (2008) The pathology of placenta accreta, a worldwide epidemic. J Clin Pathol 61: 1243-1246.
-
Rac MW, Dashe JS, Wells CE, Moschos E, McIntire DD, et al. (2015) Ultrasound predictors of placental invasion: the Placenta Accreta Index. Am J Obstet Gynecol 212: 343.
-
Silver EM, Fox KA, Barton JR, Abuhamad AZ, Simhan H, et al. (2014) Center of excellence for placenta accreta. Am J Obstet Gynecol S0002-9378(14)02248-0.
-
Hoveyda F, MacKenzie IZ (2001) Secondary postpartum haemorrhage: incidence, morbidity and current management. BJOG 108: 927-930.
-
Torrenga B, Huirne JA, Bolte AC, van Waesberghe JH, de Vries JI (2013) Postpartum monitoring of retained placenta. Two cases of abnormally adherent placenta. Acta Obstet Gynecol Scand 92: 472-475.





