Obstetrics and Gynaecology Cases - Reviews
An Unusual Cause of Transient Fetal Ascites: Fetal Ovarian Cyst Rupture
Demirpençe S*
Sifa University, Turkey
*Corresponding author: Demirpençe S, Sifa University Ankara Street 35100, Bornova, Izmir, Turkey, Tel: 905055000000, E-mail: savasdemirpence@yahoo.com
Obstet Gynecol Cases Rev, OGCR-1-016, (Volume 1, Issue 2), Case Report; ISSN: 2377-9004
Received: November 07, 2014 | Accepted: December 03, 2014 | Published: December 07, 2014
Citation: Demirpençe S (2014) An Unusual Cause of Transient Fetal Ascites: Fetal Ovarian Cyst Rupture. Gynecol Cases Rev 1:016. 10.23937/2377-9004/1410016
Copyright: © 2014 Demirpençe S. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Background: Ovarian cysts can be diagnosed prenatally and in case of persistence are found in newborns. Most resolve spontaneously, although in a small proportion of cases they can persist and be symptomatic. Fetal ascites may be due to immune or non immune hydrops fetalis or to a series of locoregional problems (bowel perforation, ovarian cyst rupture, uroperitoneum, etc�) However, it can be an isolated finding rarely.
Case: In this case we reported an isolated fetal ascites case that was caused by fetal ovarian cyst rupture.
Conclusion: Fetal hydrops may be expression of a systemic or abdominal locoregional pathological process. We mentioned that fetal ovarian cyst rupture could be a cause of isolated fetal ascites with report of a case.
Keywords
Fetal ascites, Fetal ovarian Cyst, Fetal ovarian cysts rupture
Introduction
Fetal ascites can be recognized as a symptom of fetal hydrops and can develop in response to numerous etiologic factors. Recently, due to frequent usage of Rh immunglobulin, non-immune hydrops is more frequent then immune hydrops fetalis in contrast to past. Fetal ascites can also occur isolated, independent from other serousal cavities and subcutaneous tissue due to various congenital abnormalities.Etiology of primary isolated fetal ascites is unclear. Most of them progresses to hydrops fetalis and some of them resolves spontaneously. In female newborns, ovarian cysts are frequently detected abdominal masses. Usually they are not symptomatic and resolve spontaneously. According to the common use of antenatal ultrasonography, we are now diagnosing ovarian cysts more common both in utero and in the female newborn. However, they are rarely symptomatic and they have no clinical significance. In this presentation is wereported an unexpected etiologic factor of fetal ascites; fetal ovarian cyst rupture.
Case Report
A 34-year-old woman, gravity 2, parity 0, abortion 1 is referred to our clinic at 32 weeks of gestation because of fetal intraabdominal cystic formation. She had an unremarkable prenatal course until this week. In history, she had no clinical significant coincident disease. We learned that she attended all prenatal visits regularly and she had no pathological finding in all previous visits. In the 32 weeks ultrasonography fetal biometric measurements were compatible with gestational age, amniotic fluid volume was in normal limits. Maternal blood pressure was 110/70mmHg and fetal movements were well. In ultrasonography, we detected a 15mm spheric cystic formation on the left lower side of fetal abdomen (Figure 1). The cystic formation was not related with renal structures and bladder. In Doppler sonography, we did not determine intracystic blood supply. Bilateral renal structures were evaluated normal, fetal intestines were observed normal. Because the fetus is female, cystic formation was anechoic and fetus had no accompanying genitourinary or gastrointestinal abnormalities we primarily suggested on an ovarian cyst. Due to existing fetal ovarian cyst, patient was evaluated about maternal diabetes and hypothyroidism again and resulted both in normal limits. Patient was called for a new visit two weeks later. In the 34 th week, ultrasonography fetal growth was compatible with 34 weeks gestation and amniotic fluid was in normal limits again. Only abdominal circumference was measured as 99% for this week because of fetal intraabdominal fluid. In fetal abdomen, a widespread fluid accumulation was observed (Figure 2). Ovarian cyst on the left fetal ovary was regressed and was measured 14 mm with an ellipsoid shape (Figure 3). Intestines were floating in this fluid. Fetal thorax was normal and hydrothorax was not determined. We did not observe edema in subcutaneous tissue and we evaluated as primary fetal ascites even so we performed an assessment about hydrops fetalis. Hemogram, HbA1c, VDRL, TORCH screening, Parvovirus B-19 screening, indirect coombs test and anticardiolipin IgM and IgG studies were performed. Middle cerebral artery peak systolic velcity was 55cm/sec(1,12 MOM) and was not signifying fetal anemia. We recommended fetal karyotyping to patient because it could be an onset of fetal hydrops. However, patient did not accept karyotyping. All screenings were evaluated normal. Fetal cardiac examination was normal. Patient was called for a visit two weeks later again. At the next examination in the 36th weeks of pregnancy, fetal ultrasonography was completely normal. We have not observed any fetal ovarian cyst and fluid in fetal abdomen was completely regressed. Patient had a 3400 gr healthy baby on the 40th weeks of her pregnancy. No abnormal finding determined in postnatal abdominal ultrasonography.
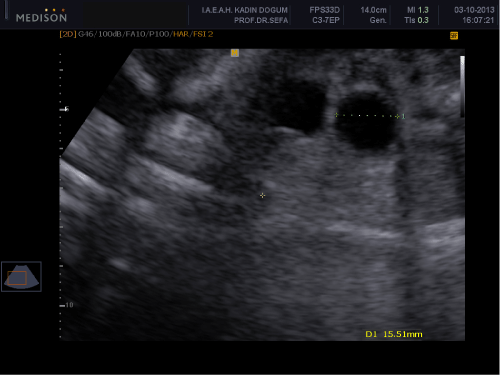
Figure 1: 15mm spheric cystic formation on the left fetal ovary.
View Figure 1
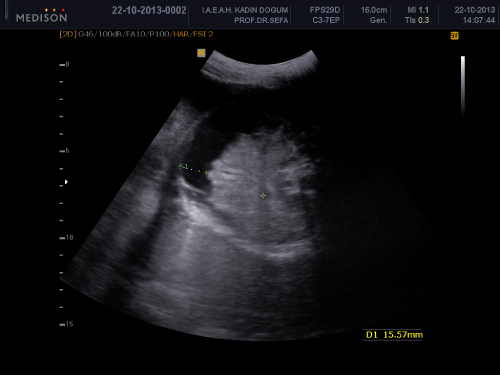
Figure 2: idesread uid accumulation in etal adomen.
View Figure 2
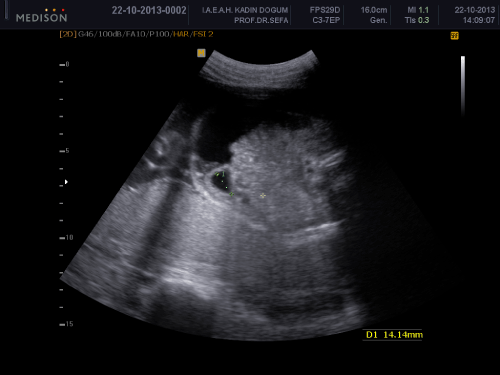
Figure 3: 14mm regressed ovarian cyst on the left fetal ovary.
View Figure 3
Discussion
Hydrops Fetalis is classically defined as pathologic fluid accumulation in soft tissues and in at least two fetal serousal cavities including skin and placental edema, pericardial and pleural effusion and ascites with or without evidence of isoimmunization [1]. It is classified as immune and non-immune hydrops fetalis. Isolated fetal hydrothorax or ascites is a condition characterized by accumulation of pleural effusion or acidic fluid,without generalized skin edema, due to various etiologies [2]. Fetal ascites is generally a part of the symptoms in cases with non-immune fetal hydrops, which refers to fluid collection in at least two body cavities or fluid collection in one cavity plus diffuse subcutaneous edema. The etiology of non-immune fetal hydrops includes numerous causes such as chromosomal anomalies, intrauterine infections, cardiac failure, and structural anomalies of various organs. Fetal ascites sometimes occur independently without fluid accumulation in any other serousal cavities or subcutaneous tissue. It has defined as primary isolated ascites. Primary isolated ascites can be a sign of fetal hydrops at early stage and may progress to hydrops consecutively. We suggested karyotyping to the patient because ascites could be an onset for hydrops. It may be isolated and usually as a result of abnormalities of abdominal structures like posterior urethral valve or cloacal persistence, bowel obstruction if they rupture or rupture of any abdominal mass. In a large series study focusing on non-immune fetal ascites, it was reported that,the fetal ascites accompanied by fetal hydrops had more unfavorable prognosis in comparison with isolated fetal ascites without an association of hydrops [3]. Isolated hydrops can progress and cause hydrops and polyhydramnios by compression to inferior vena cava and other abdominal organs. In our case ascites resolved spontaneously in two weeks and did not cause any complication.
Hormonal stimulation is generally considered responsible for the etiology of fetal ovarian cysts (fetal gonadotropins, maternal estrogen and placental human chorionic gonadotropin). The incidence of ovarian cysts has been estimated at more than30 % (this estimate is based on an investigation of stillborn or infants who died within 28 days ofbirth) [4]. They are often isolated and associated with pregnancies complicated by maternal diabetes, toxemia or Rh isoimmunization, probably because of the increased placental production of hcG in these conditions [5]. Especially mesentericcyst,urachuscyst,ectopichydronephrotickidney,intestinalduplicationanomalies,cysticteratomaandintestinal obstructionshould be kept in mind in differantial diagnosis [6]. After a complete ultrasound examination, we did not detect any other anomaly in other structures. Because fetus was female and cyst was pelvic lacolized, we condensed on ovaries. Formation of the cystic mass was anechoic and was not vascularized, these were approving our diagnosis. Ovarian cysts are usually diagnosed in a routine obstetrical ultrasonography with detection of a fetal cystic pelvic mass. Once the prenatal diagnosis of a probable ovarian cyst has been made, it is most important toperform serial ultrasound examinations to detect any structural change (size, appearance) in the mass or complications (hidramnios, ascites, torsion). A simple ovarian cyst has smooth borders and does not have internal echogen structures [7]. Most of them are unilateral. Anechoic simple cysts can become complex masses, featuring echogenicity, ?uid levels, and internal echoes. The presence of these ultrasonograhic elements is usually considered a symptom of torsion. Prenatal torsion, the most frequent complication of prenatal ovarian cysts, has anabout 40% incidence. The torsion occurs more frequently in voluminous cysts, but it also occurs in cysts sized 2cm [8]. Some cysts remain anechoic inside the uterus. In our case due to size and ultrasonographic apperance we decided to follow patient. A relevant number of them, about 50%, resolve spontaneously after birth [9].
Like adults when an ovarian cystic formation ruptures, haemorrhage can result from that ovary. When rupture is actualized, ruptured cystic formation changes in size and shape, abdominal bleeding causes ultrasonographic ascites image.
In our case two weeks later we detected the cystic formation changed in size and shape. With this information we related free fluid with ruptured ovarian cyst. In accordance with this, two weeks later fetal ascites regressed spontaneously.
In conclusion, fetal ovarian cyst rupture should be evaluated as an etiological factor in isolated fetal ascites cases. Evaluation of fetal ovaries is difficult and generally ovaries can not be differentiated from other pelvic structures. However, when an abdominal detected cystic formation is presented, fetal ovaries should be kept in mind.
References
-
Nose S, Usui N, Soh H, Kamiyama M, Tani G, et al. (2011) The prognostic factors and the outcome of primary isolated fetal ascites. Pediatr Surg Int 27: 799-804.
-
Moroy P, Tuncay P, �elen S, Sucak A, Mungan T, et al. (2004) Isolated Fetal Ascites: Evaluation of Six Cases. Gynecol Obstet Reprod Med 10: 164-166.
-
Favre R, Dreux S, Dommergues M, Dumez Y, Luton D, et al. (2004) Nonimmune fetal ascites: a series of 79 cases. Am J Obstet Gynecol 190: 407-412.
-
Kwak DW, Sohn YS, Kim SK, Kim IK, Park YW, et al. (2006) Clinical experiences of fetal ovarian cyst: diagnosis and consequence. J Korean Med Sci 21: 690-694.
-
Hasiakos D, Papakonstantinou K, Bacanu AM, Argeitis J, Botsis D, et al. (2008) Clinical experience of five fetal ovarian cysts: diagnosis and follow-up. Arch Gynecol Obstet 277: 575-578.
-
�zel A, Duymus R, Bayram A, �akmak�i E, Sever N, et al. (2011) Fetal over kist torsiyonu tanisinda perinatal ultrason ve manyetik rezonans goruntulemenin rolu. S.E.E.A.H. Tip B�lteni 45: 56 -59.
-
Giorlandino C, Bilancioni E, Bagolan P, Muzii L, Rivosecchi M, et al. (1994) Antenatal ultrasonographic diagnosis and management of fetal ovarian cysts. Int J Gynaecol Obstet 44: 27-31.
-
Grapin C, Montagne JP, Sirinelli D, Silbermann B, Gruner M, et al. (1987) Diagnosis of ovarian cysts in the perinatal period and therapeutic implications (20 cases). Ann Radiol (Paris) 30: 497-502.
-
Noia G, Riccardi M, Visconti D, Pellegrino M, Quattrocchi T, et al. (2012) Invasive fetal therapies: approach and results in treating fetal ovarian cysts. J Matern Fetal Neonatal Med 25: 299-303.





