Obstetrics and Gynaecology Cases - Reviews
Paraganglioma and Pregnancy: Management of Cesarean Delivery and Subsequent Laparoscopic Adrenalectomy
Jennifer T. Mallek1*, JMonique Ho2, Christiana Shaw3, Mark Rice1 and Tammy Euliano1
1Department of Anesthesiology, University of Florida College of Medicine, Florida, USA
2Department of Obstetrics and Gynecology, University of Florida College of Medicine, Florida, USA
3Department of Surgery, University of Florida College of Medicine, Florida, USA
*Corresponding author: Jennifer Mallek, MD, Department of Anesthesiology, University of Florida College of Medicine, 1600 SW Archer Road, PO Box 100254, Gainesville, FL 32610, USA, Tel: 352-328-9888, E-mail: jmallek@anest.ufl.edu
Obstet Gynecol Cases Rev, OGCR-1-013, (Volume 1, Issue 2), Case Report; ISSN: 2377-9004
Received: October 21, 2014 | Accepted: November 27, 2014 | Published: November 30, 2014
Citation: Mallek JT, Ho M, Shaw C, Rice M, Euliano T (2014) Paraganglioma and Pregnancy: Management of Cesarean Delivery and Subsequent Laparoscopic Adrenalectomy. Gynecol Cases Rev 1:013. 10.23937/2377-9004/1410013
Copyright: © 2014 Mallek JT, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Background: Pheochromocytomas and paragangliomas are rare catecholamine-secreting tumors (CSTs) that may lead to severe hypertensive episodes and are associated with high maternal and fetal mortality when undiagnosed or mismanaged.
Case: A 28-year-old full-term, normotensive woman with presumed pheochromocytoma underwent a cesarean under epidural anesthesia, which was also used for postoperative pain control, followed by an uncomplicated adrenalectomy under general anesthesia 5 days later. The pathology report revealed a paraganglioma. The patient and her newborn were discharged home in good health 2 days following surgery.
Conclusion: We describe the diagnostic workup and perioperative management of a patient with a known CST, as well as the management of her neonate. In addition, we describe potential anesthetic complications in this situation and how these issues were addressed.
Introduction
Pheochromocytomas and paragangliomas are rare catecholamine-producing neuroendocrine neoplasms that may lead to severe hypertensive episodes. The incidence in the general population is 0.05% to 0.12% and is lower in pregnancy (0.002%�0.007%) [1].
These tumors (also known as catecholamine-secreting tumors [CSTs]) are known as �the great mimic� as their presentation is highly variable. It is estimated that 30% to 40% of patients are asymptomatic at presentation [1]. CSTs are increasingly being discovered incidentally via imaging studies, CT and MRI scans, that are performed for other reasons, and up to 70% of pheochromocytomas are diagnosed this way [2]. The classic symptoms of episodic headache, sweating, and tachycardia, in association with hypertension, can easily be confused for the more common causes of hypertension in pregnancy and are treated differently. It is important to maintain a high level of suspicion as pheochromocytomas are one of the most threatening medical conditions to mother and fetus.
We describe the diagnostic workup as well as the perioperative anesthetic management of a term pregnant woman with a recent diagnosis of a CST.
Case Report
A 28-year-old otherwise healthy female presented to an outside institution at 30 weeks gestation with right-sided back/shoulder pain radiating to her flank and abdomen. Ultrasound at that time revealed a right adrenal mass. On questioning, the patient reported two episodes of palpitations, sweating, and anxiety lasting seconds that occurred earlier in her pregnancy. She was not documented to be hypertensive at any time. A 24h urine revealed elevated normetanephrines at 945�g/dL (normal maximum 650�g/dL) and normal metanephrines. Thoracic MRI revealed a 3.8-� 3.5-� 4cm solid, smoothly marginated mass of the right adrenal gland (Figure 1). At this point, a presumed diagnosis of pheochromocytoma was made. The patient was discharged home and returned to our institution at 36 weeks gestation for delivery and tumor management. Her past medical history was significant only for a cesarean delivery for arrest of descent 7 years previously.
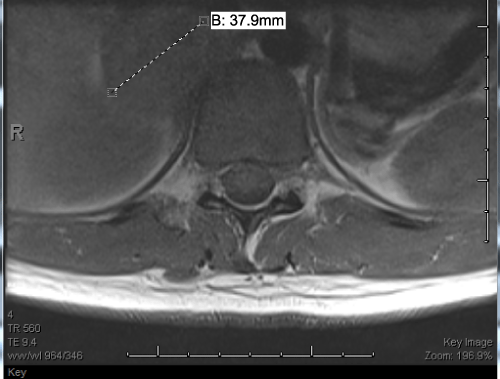
Figure 1: T1 Weighted Thoracic MRI obtained at 32 weeks gestation.
View Figure 1
Upon admission, the patient was started on phenoxybenzamine pretreatment for the week prior to her planned cesarean. She quickly achieved therapeutic levels as evidenced by orthostatic hypotension in the setting of euvolemia. Frequent fetal monitoring, via intermittent fetal heart monitor and daily non-stress tests, was reassuring. A joint obstetric, surgical, anesthesia, and pediatric conference developed a management plan that included a repeat cesarean performed at 36 weeks 6 days gestation, followed, after a week of uterine involution, by laparoscopic adrenalectomy to avoid both an overly large or second abdominal incision at cesarean and theoretical risk of carbon dioxide embolus by performing a laparoscopic procedure in the setting of dilated pelvic vasculature.
On the day of the cesarean delivery, the patient was started on magnesium sulfate 2g/h to aid with arterial hypertension control, in addition to 1.5 times her maintenance intravenous (IV) fluid rate. Her morning dose of phenoxybenzamine was held. She was premedicated with midazolam at her request to aid with anxiolysis and standard monitors were placed followed by an awake left radial arterial catheter. A lumbar epidural catheter was placed in the sitting position with negative aspiration; no test dose was administered out of concern that the epinephrine in the test dose solution could aggravate catecholamine release from the tumor. With the patient in the supine position with left uterine displacement, the epidural was slowly bolused with 0.5% ropivacaine followed by 3% 2-chloroprocaine to achieve a T4 level. Mild hypotension was treated predominately with crystalloid boluses. Phenylephrine is a direct-acting alpha agonist that is the preferred agent for treatment of hypotension in these tumors, thus phenylephrine, 100�g at a time, was titrated in to help maintain blood pressure until sufficient crystalloid was administered (approximately 3L). The magnesium infusion was discontinued shortly after bolusing the epidural. An uneventful low transverse cesarean section and bilateral tubal interruption were performed by routine manner. One difference is that the uterus was not exteriorized and the uterine incision was closed in 1 layer. The surgeon took special care not to apply pressure over the tumor.
A live-born female infant was delivered weighing 3121g and was immediately handed to the neonatal intensivist team. Although, meconium-stained fluid was noted, the neonate had an initial vigorous cry. The fluid was suctioned from the nose and mouth and the infant stopped crying and had little to no respiratory effort. Bag mask ventilation was started with a heart rate in the 50s and the infant was subsequently intubated. Heart rate and color improved with positive pressure ventilation; however, no respiratory effort was noted. A peripheral IV was started and a 30mL bolus of crystalloid was administered. Apgar scores were 2 and 9 at 1 and 5 min, respectively. The neonate was then transferred to the neonatal intensive care unit (NICU). On arrival there were borderline low mean arterial pressures of 38mmHg that responded to an addition a l30mL bolus of crystalloid. There was no additional hypotension. Extubation occurred within 2h and the mother was able to hold and breastfeed the infant less than 2h later. The neonate remained in the NICU for 2 days for observation due to concern about hypotension and/or respiratory depression.
Following the operation, postoperative pain was controlled with a continuous epidural infusion of 0.125% ropivacaine with 2�g/mL fentanyl at 8mL/h with a demand bolus option of 4mL every 30 minutes. This method was selected to avoid hypertension from histamine release caused by other analgesics and poorly controlled pain. Pain was well-controlled without hypertensive episodes or adjuvant opioid use and the catheter was removed on postoperative day 3. The majority of surgical pain from a cesarean usually subsides by the third day. The infusion medication was trialed off in the morning and once the patient had full recovery of sensation and demonstrated adequate pain control, the catheter was removed. If patient reported significant pain at this time the epidural infusion would have been restarted. Removal of the epidural catheter also allowed for patient ambulation and Foley catheter removal. The patient desired to breastfeed therefore phenoxybenzamine was held for two days postoperatively to establish milk supply and to avoid the potential for neonatal hypotension, as suggested by Breastfeeding Medicine consultation. She was then restarted on phenoxybenzamine 10mg BID. At this point, she started to discard pumped milk to maintain her supply.
On postoperative day 5, she underwent an uneventful laparoscopic adrenalectomy. A Harmonic scalpel was used to dissect the mass at the level of the superior portion of the kidney and retroperitoneum. The inferior vena cava (IVC) was identified and dissection proceeded on medial side of the mass alongside the IVC. Mobilization of the mass then proceeded from medial to lateral fashion and from an inferior to superior fashion. The mass was noted to be grossly within the adrenal gland and at the inferior border. Two clips were placed distally and one proximally adjacent to the mass. There were no significant hypertensive episodes, with maximum systolic pressure less than 140 mmHg. Just prior to clamping the adrenal vein, a norepinephrine infusion was started at 0.01�g/kg/min and titrated up to a maximum rate of 0.07�g/kg/min. This was titrated off just prior to extubation. Postoperatively, the patient was observed overnight in the ICU and was breastfeeding once again on postoperative day 1. She was discharged home postoperative day 2 with her newborn.
Pathologic examination revealed the tumor to be adjacent to and histologically distinct from the adrenal gland, although histologically indistinguishable from pheochromocytoma.It was, therefore, a paraganglioma by definition. Preoperative elevation of normetanephrines and not metanephrines, are consistent with this finding as paragangliomas typically secrete norepinephrine but not epinephrine.
CommentHypertension, gestational or chronic, occurs in 3.6% to 9.1% of pregnancies [3]. The classic symptoms of pheochromocytoma, episodic headache, sweating, and tachycardia in association with hypertension, can be confused with more common causes. Hypertensive crisis in pregnancy is often treated with labetalol because of its wide availability, combined alpha and beta blockade, and its low impact on uteroplacental blood flow at lower doses. It has documented transplacental transfer, but transient and minor neonatal side effects. Labetalol does not have adequate alpha-blocking properties in the setting of a CST [4]. Blockade of vasodilatory beta-2 receptors without prior blockade of alpha receptors may incite a hypertensive crisis. Surgery, pain,and other factors (Table 1) can also incite a hypertensive crisis [1].
![]()
Table 1: Factors Exacerbating Hypertension in Patients with Release from CSTs
View Table 1
Pheochromocytomas secrete varying amounts of epinephrine, norepinephrine, and to a lesser extent dopamine. They can secrete a single catecholamine or any combination of the three mentioned above. Pheochromocytomas associated with MEN2 syndromes always secrete epinephrine. Paragangliomas, however, are unlikely to secrete epinephrine. It is postulated that the enzyme phenylethanolamine N-methyltransferase, which converts epinephrine to norepinephrine requires cortisol as cofactor. A CST outside of the adrenal gland does not have sufficient cortisol levels needed for significant conversion [5].
Antihypertensive treatment of pheochromocytoma in pregnancy is not well defined, as the literature largely consists of case reports [6]. Medical management of pheochromocytoma and paraganglioma are the same and should usually be initiated with alpha-adrenergic blockade. Phenoxybenzamine, a long-acting irreversible alpha blocker, is often used in the pregnant and non-pregnant patient. It is generally considered safe for the fetus; however, it does cross the placenta, leading to transient hypotension [4]. Whether phenoxybenzamine is excreted in breast milk remains unknown, although its low molecular weight makes transfer likely. This neonate experienced mild hypotension that responded promptly to fluids, and respiratory depression that may have been related to phenoxybenzamine, midazolam, or other causes.
Once a patient is adequately alpha-blocked, as indicated by orthostatic hypotension following adequate volume replacement, beta-blockade can be added to control tachycardia and tachyarrhythmias [4]. Transient orthostasis is acceptably safe for the fetus to achieve maternal benefit, if done in a monitored setting where prompt treatment is available. Caution is required in patients with cardiomyopathy who may develop pulmonary edema secondary to withdrawal of beta stimulation. Catecholamine-induced cardiomyopathy can lead to development of intractable hypotension, bradycardia, and asystolic arrest following beta-blockade [7].
Vaginal delivery was once thought contraindicated in parturients with pheochromocytoma, as labor can stimulate massive catecholamine release. However, some authors report successful vaginal delivery when the clinical condition is well-controlled [1]. Considering our patient�s low likelihood of successful trial of labor because of her obstetric history, a cesarean delivery was deemed safest for the patient and her fetus; the patient concurred.
Once the patient is medically optimized, surgical excision of the tumor follows. If diagnosed before 24 weeks gestation, laparoscopic tumor removal and continuation of the pregnancy is recommended. When diagnosis occurs after 24 weeks, as in this case, uterine volume complicates laparoscopic removal [1,4]. Patients have been successfully managed with combined and separate procedures for cesarean and tumor removal, although published cases to date have waited 2 to 6 weeks before adrenalectomy if it was not done at the time of cesarean [8]. Laparoscopic extirpation of pheochromocytomas in non-pregnant patients is preferred based on established reduction of postoperative complications, hospital stay, and recovery. If this was to be performed combined, it would require a large open incision with significantly longer recovery time or a technically more challenging laparoscopic adrenalectomy with a theoretically increased risk for carbon dioxide embolus secondary to dilated pelvic veins. Following adequate uterine involution and Pfannenstiel healing after delivery, a laparoscopic adrenalectomy was able to be performed shortly after the Cesarean. Because of these factors, our patient�s desire to bond with her newborn, and surgeon familiarity with the laparoscopic approach, we opted to proceed with the staged approach.
Anesthetically, our first task was determining the optimal intraoperative management for the cesarean. General anesthesia for scheduled cases on otherwise healthy parturients is associated with an increase in complications, mostly relating to aspiration and hypoxia, relative to regional anesthesia. In addition, the latter allows for minimal systemic drug levels to mother and fetus, as well as maternal awareness during delivery. Given these factors, in addition to our patient�s favorable hemodynamic profile, we proceeded safely with a regional technique.
The second obstacle we faced was postcesarean pain management in a patient with a CST. Because most long-acting opioids such as morphine, codeine, and oxycodone can trigger catecholamine release from the tumor via histamine, we avoided our standard neuraxial morphine and instead continued her epidural infusion. Fentanyl and tramadol are not known to significantly stimulate histamine release, but tramadol has serotonin-norepinephrine reuptake inhibitor properties that could potentially exacerbate hypertension [4]. Hydromorphone releases histamine to a much smaller extent than morphine and can likely is used safely.
We have described the diagnostic workup and successful perioperative management of a patient with a known CST. With appropriate medical management, these patients can safely deliver via cesarean under epidural anesthesia. In addition, we have demonstrated an epidural to be extremely beneficial in providing adequate postcesarean pain management prior to removal of her tumor while allowing establishment of milk supply. A short 5-day interval from cesarean to the preferred approach of laparoscopic adrenalectomy, to allow Pfannenstiel closure and uterine involution and minimize maternal time on alpha blockade at hypertensive risk, appears feasible. The uniqueness of this situation emphasized the importance of teams with varying expertise working together at a specialized center equipped to handle complex cases.
References
-
Plu I, Sec I, Barr�s D, Lecomte D (2013) Pregnancy, cesarean, and pheochromocytoma: a case report and literature review. J Forensic Sci 58: 1075-1079.
-
Oshmyansky AR, Mahammedi A, Dackiw A, Ball DW, Schulick RD, et al. (2013) Serendipity in the diagnosis of pheochromocytoma. J Comput Assist Tomogr 37: 820-823.
-
Roberts CL, Ford JB, Algert CS, Antonsen S, Chalmers J, et al. (2011) Population-based trends in pregnancy hypertension and pre-eclampsia: an international comparative study. BMJ Open 1: e000101.
-
Mazza A, Armigliato M, Marzola MC, Schiavon L, Montemurro D, et al. (2014) Anti-hypertensive treatment in pheochromocytoma and paraganglioma: current management and therapeutic features. Endocrine 45: 469-478.
-
Chen H, Sippel RS, O'Dorisio MS, Vinik AI, Lloyd RV, et al. (2010) The North American Neuroendocrine Tumor Society consensus guideline for the diagnosis and management of neuroendocrine tumors: pheochromocytoma, paraganglioma, and medullary thyroid cancer. Pancreas 39: 775-783.
-
Miller C, Bernet V, Elkas JC, Dainty L, Gherman RB (2005) Conservative management of extra-adrenal pheochromocytoma during pregnancy. Obstet Gynecol 105: 1185-1188.
-
Kinney MA, Narr BJ, Warner MA (2002) Perioperative management of pheochromocytoma. J Cardiothorac Vasc Anesth 16: 359-369.
-
Berends FJ, Harst EV, Giraudo G, Terkivatan T, Kazemier G, et al. (2002) Safe retroperitoneal endoscopic resection of pheochromocytomas. World J Surg 26: 527-531.





