Obstetrics and Gynaecology Cases - Reviews
Hydrops Fetalis Associated with Congenital Cytoplasmic Body Myopathy
Yuki Kodama*, Hiroshi Sameshima, Masatoki Kaneko, Hideki Kawaguchi, Tomoaki Ikeda and Tsuyomu Ikenoue
Department of Obstetrics and Gynecology and Perinatal Center, University of Miyazaki, Japan
*Corresponding author: Yuki Kodama, MD, Faculty of Medicine, Department of Obstetrics and Gynecology, University of Miyazaki, 5200 Kihara, Kiyotake, Miyazaki 889-1692, Japan, Tel: +81-985-85-0988, Fax: +81-985-85-6149, E-mail: yuki_kodama@med.miyazaki-u.ac.jp
Obstet Gynecol Cases Rev, OGCR-3-099, (Volume 3, Issue 6), Case Report; ISSN: 2377-9004
Received: August 13, 2016 | Accepted: November 24, 2016 | Published: November 26, 2016
Citation: Kodama Y, Sameshima H, Kaneko M, Kawaguchi H, Ikeda T, et al. (2016) Hydrops Fetalis Associated with Congenital Cytoplasmic Body Myopathy. Obstet Gynecol Cases Rev 3:099. 10.23937/2377-9004/1410099
Copyright: © 2016 Kodama Y, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
We report a case of congenital cytoplasmic body myopathy which manifested as fetal hydrops in utero and was subsequently confirmed by muscle biopsy after birth. It is speculated that fetal hydrops is one of the clinical features associated with congenital cytoplasmic body myopathy, which is, to our knowledge, reported here for the first time.
Keywords
Nonimmune hydrops fetalis, Cytoplasmic body myopathy, Congenital myopathy, Floppy infant, Respiratory failure
Introduction
Cytoplasmic body myopathy (CBM) is a rare congenital myopathy characterized by the presence of numerous cytoplasmic bodies in the skeletal muscle fibers. Following the first report of CBM by Nakashima, et al. in 1970 [1], the literature now contains more than 30 cases. Clinical features vary from severe congenital type to mildly progressive adult-onset type. We experienced a case of very severe congenital type CBM, which manifested as generalized hydrops fetalis at 33 weeks of gestation.
Case Report
A 27-year-old, gravida 3, para 1 Japanese woman was transferred to our hospital because of fetal pleural effusion and polyhydroamnios at 33 weeks of gestation. She had a family history of amyotrophic lateral sclerosis in her maternal uncle. Her first child, a 3-year-old boy, was healthy. The pregnancy had been uneventful until 33 weeks, when the patient noticed the fetus was less active than before. On admission, the fetus had bilateral pleural effusion, ascites, and subcutaneous edema. The amniotic fluid index was more than 30 cm. Fetal ejection fraction of the left ventricle was 60 to 80%, and the heart rate was 140-150 beat per minute without arrhythmia. There were no signs of anomaly. Screening tests results were blood type A, Rh positive, indirect Coombs test negative, 75 g OGTT normal, TORCH screening negative, and Kleihauer-Betke staining negative. At 36 weeks and 0/7, the biophysical profile score was 2 of 10. A non-reactive result and prolonged deceleration was seen on fetal heart rate monitoring and the baby was delivered by cesarean section.
The baby was male, generally edematous, and weighed 3.022 grams, with an Apgar score of 3 at 1 min and 5 at 5 min. He showed respiratory difficulty with bilateral pleural effusion, ascites, micrognathia, and foot-drop. He required respiratory support with bilateral chest drainage. The pleural effusion was composed of more than 90% lymphocytes. After milk feeding was started, chylothorax was diagnosed by identification of chylon in the pleural effusion. He was floppy and showed no spontaneous body movements except for fingers and eyes in spite of stabilization of blood gases and vital signs. Deep tendon reflexes were completely absent.
Various examinations were performed to ascertain the etiology of the "floppy infant" condition. Creatinine kinase (54 U/L) was normal (normal range, 10-200 U/L), aldolase (39.6 U/L) was mildly elevated (normal range, < 32 U/L), and serum lactate and pyruvate were normal at 7.9 mg/dl (normal range, < 27 mg/dl) and 0.64 mg/dl (range, 0.3-0.9 mg/dl), respectively. Deoxyribonucleic acid (DNA) analyses excluded myotonic dystrophy and Werdnig-Hoffmann disease. Chromosomal abnormalities seen in Prader-Willi syndrome were absent. Cranial computed tomography showed several calcifications in the periventricular white matter and right basal nuclei. Blood and urine tests for cytomegalovirus and toxoplasmosis were all negative. Cerebrospinal fluid analysis also showed normal cell counts and protein concentrations. Electroencephalography, auditory brainstem responses, and funduscopy were all normal. Nerve conduction velocity was within the normal range. Injection of edrophonium hydrochloride did not reverse the hypotonus, ruling out myasthenia gravis.
At 4-months of age, biopsy from the quadriceps femoris muscle revealed numerous cytoplasmic bodies in muscle fibers of various sizes. Electron microscopy showed the cytoplasmic bodies had a homogeneous dense central core surrounded by thin radiating filaments (Figure 1). On the basis of these findings, the patient was diagnosed as having congenital CBM.
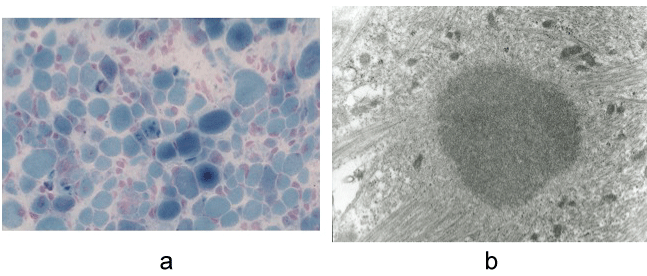
.
Figure 1 Biopsy from left quadriceps femoris muscle. a) Optical microscopic observations modified Gomori trichrome stain. Cytoplasmic bodies are apparent in a variety of muscle fibers. X 260; b) Electron micrograph shows a cytoplasmic body with a dense central core and radiating peripheral filaments. X 10,000.
View Figure 1
As the patient required life-long respiratory support, he was transferred to a chronic care hospital, where he died of cardiac arrest following convulsions and metabolic acidosis at 2.5 years. Autopsy was not performed.
Comments
A review of the English literature searched by the keywords of "cytoplasmic body myopathy" and "infant" reveals that this is the first report of congenital severe CBM associated with hydrops fetalis. Nonimmune fetal hydrops is the end feature of a variety of pathophysiologic mechanisms. It is sometimes accompanied by congenital myopathy such as congenital myotonic dystrophy or nemaline myopathy. The etiologic relationship between congenital myopathy and fetal hydrops has not been clearly established.
There are several pathophysiological processes proposed to account for the pathway of fetal hydrops. The pathogenesis leading to hydrops is unclear in this case. It was not caused by cardiac failure because the cardiac function was normal. Hypoplasia of the thorax may be the contributing factor because of interference with amniotic fluid transfer through the lung. The lymphatic abnormality might have resulted in increased interstitial fluid and caused fetal hydrops with chylothorax.
Cytoplasmic bodies were initially reported by Engel in 1962 in patients with advanced denervation and myotonic dystrophy [2]. Since then, cytoplasmic bodies have been identified in a variety of neuromuscular diseases including metabolic disorders, muscular dystrophies, and neurogenic atrophies. Cytoplasmic bodies are the morphologic hallmark of sporadic as well as familial congenital myopathies including CBM. When a muscle biopsy specimen shows a high percentage of muscle fibers containing cytoplasmic bodies and no other definitive diagnosis can be made, CBM is the most likely diagnosis.
Mizuno, et al. reported a severe case of congenital CBM [3]. They classified 18 cases of CBM into four clinical patterns according to onset and severity of clinical prognosis: 1) severe congenital, 2) benign congenital, 3) severe juvenile and 4) severe adult. According to this classification, our case was classified as a severe congenital form of CBM.
We have summarized the cases of congenital CBM excluding Werdnig-Hoffmann disease thus far reported in table 1. Six of the total 8 cases had cardiorespiratory failure, while in the 3 cases [4,5], the clinical course was benign. Typically, congenital or early infantile cases have dysphagia, a high arched palate, micrognathia, facial weakness, and hypotonus of all skeletal muscles [6-8].
![]()
Table 1: Clinical and laboratory data in reported cases of congenital cytoplasmic body myopathy.
View Table 1
Our case was CBM first manifested as hydrops fetalis at 33 weeks of gestation. There are no known causes other than congenital myopathy that lead to nonimmune hydrops fetalis, and thus we speculate that fetal hydrops is one of the clinical features associated with CBM.
Acknowledgement
We would like to thank Dr. Ikuya Nonaka for his valuable comments on muscle biopsy, and Drs. Keiichiro Kohno and Hiroshi Aikou for their help in taking care of this patient.
References
-
Nakashima N, Tamura Z, Okamoto S, Goto H (1970) Inclusion bodies in human neuromuscular disorders. Arch Neurol 22: 270-278.
-
Engel WK (1962) The essentiality of histo and cytochemical studies of skeletal muscle in the investigation of neuromuscular disease. Neurology 12: 778-794.
-
Mizuno Y, Nakamura Y, Komiya K (1989) The spectrum of cytoplasmic body myopathy: Report of a congenital severe case. Brain and Development 11: 20-25.
-
Gobel HH, Schloon H, Lenard HG (1981) Congenital myopathy with cytoplasmic bodies. Neuropediatr 12: 166-180.
-
Wolburg H, Shcolte W, Langohr HD, Peiffer KH, Heckl RW, et al. (1982) Slowly progressive congenital myopathy with cytoplasmic bodies. Report of two cases and a review of the literature. Ciln Neuropathol 1: 55-66.
-
Bertini E, Ricci E, Boldrini R, Servidei S, Fusilli S, et al. (1990) Involvement of respiratory muscles in Cytoplasmic body myopathy - a pathological study. Brain Dev 12: 786-806.
-
Sekijima Y, Ikeda S, Katai S, Matsuda M, Hashimoto T, et al. (1995) Cytoplasmic body myopathy with hypertrophic cardiomyopathy. Intern Med 34: 166-170.
-
Reed L, Young J, Gobel HH, Schochet Jr SS (1997) Congenital Cytoplasmic body myopathy: Case report. J Child Neurol 12: 149-152.





