Obstetrics and Gynaecology Cases - Reviews
Challenges Diagnosing Appendiceal Neoplasms during Pregnancy
Vidya Bharati1, Angeles Alvarez- Secord1, Thomas M Price1* and John Migaly2
1Department of Obstetrics and Gynecology, Duke University, Durham, USA
2General Surgery, Duke University, Durham, USA
*Corresponding author: Thomas M Price, MD, Departments of Obstetrics and Gynecology, Duke University, 5704 Fayetteville Road, Durham, NC 27713, USA, E-mail: price067@mc.duke.edu
Obstet Gynecol Cases Rev, OGCR-3-098, (Volume 3, Issue 6), Case Report; ISSN: 2377-9004
Received: August 15, 2016 | Accepted: September 28, 2016 | Published: November 01, 2016
Citation: Bharati V, Alvarez-Secord A, Price TM, Migaly J (2016) Challenges Diagnosing Appendiceal Neoplasms during Pregnancy. Obstet Gynecol Cases Rev 3:098. 10.23937/2377-9004/1410098
Copyright: © 2016 Bharati V, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Background: Appendiceal mucocele is a rare, often asymptomatic, tumor with malignant potential. For evaluation during pregnancy, the natural upward progression of the appendix must be considered.
Case: A 31 y.o. G1P0 at 18 weeks estimated gestational age was found to have an echogenic 7.8 cm by 3.1 cm mass adjacent to the uterine fundus during an anatomy scan. Follow-up radiology performed abdominal ultrasound was normal. Patient delivered vaginally without incident. Three years later she was diagnosed with a right complex pelvic mass identified during a saline infusion sonogram for fertility evaluation. Carcinoembryonic antigen was elevated. Laparoscopic surgery revealed appendiceal mass and normal gynecological findings. Pathology demonstrated low grade mucinous neoplasm of the appendix.
Conclusion: This case emphasizes the asymptomatic tendency and malignant potential of appendiceal mucoceles. Additionally, the diagnosis may have been delayed due to selection of an abdominal ultrasound for confirmation.
Introduction
Appendiceal mucocele was recognized by Rokitansky in 1842 and formally defined by Feren in 1876 [1]. This clinical description is associated with pathological phenomena varying from mucin producing epithelium with low-grade cytologic abnormalities pushing into the adjacent appendiceal wall without destructive characteristics to frank adenocarcinoma with invasion. It occurs in 0.3-0.4% of appendectomies, more commonly in females (4:1) and more frequently in patients over 50 [2]. Approximately 50% of cases are asymptomatic. While others present with the following symptoms: pain and mass in the right lower quadrant, gastrointestinal bleeding, nausea, vomiting, weight loss and non-specific change in bowel function. Uncommon presentations of appendiceal mucocele, mainly described in case reports include torsion, intestinal intussusception, intestinal obstruction, hematuria, and obstructive uropathy. Complications from appendiceal mucocele include malignant transformation, perforation with development of mucinous ascites and peritoneal implants (pseudomyxoma peritonei (PMP), torsion and bowel obstruction.
Cases during pregnancy have been incidentally found during ultrasonography or Cesarean delivery or after presentation with acute abdominal pain mimicking appendicitis [2]. Pregnancy offers additional challenges due to the upward movement of the appendix during gestation. This case of delayed diagnosis of appendiceal mucocele illustrates some pragmatic issues which should be considered during evaluation and reviews characteristics of this rare condition.
Case
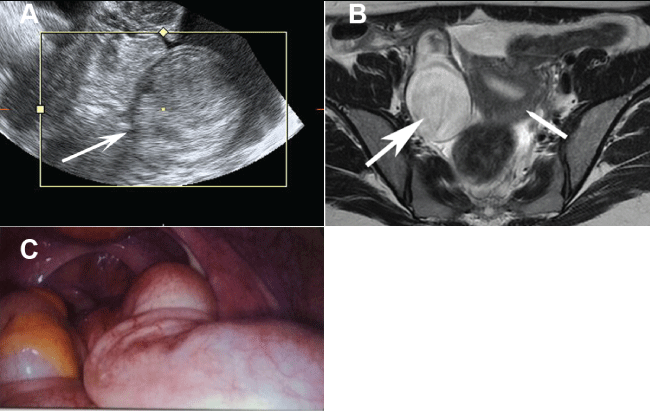
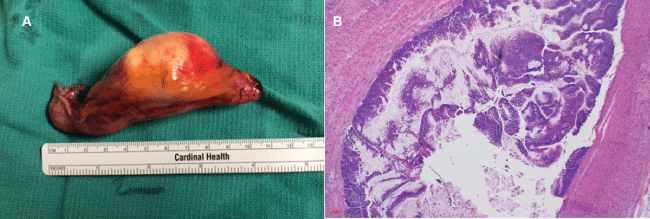
A 34 y.o. G1P1 presented 3 years previously with her first pregnancy conceived on clomiphene citrate. An obstetrician performed anatomy scan at 18 weeks revealed a 7.8 cm by 3.1 cm complex mass in the right upper quadrant adjacent to the uterine fundus. Four days later, a follow-up radiology performed abdominal ultrasound was normal. She had a normal spontaneous vaginal delivery at 36 weeks estimated gestational age. After failed attempts to conceive a second pregnancy with clomiphene citrate induction, she underwent a saline infusion sonogram for consideration of in vitro fertilization. A 5.75 cm by 3.66 cm complex mass was seen in the right pelvis separate from the uterus and ovary, characterized by echogenic layers (Figure 1). MRI revealed a 6.2 cm by 3.8 cm by 5.4 cm complex cystic mass in the right pelvis between the uterus and right ovary. Tumor markers showed cancer antigen 125 of 23.5 U/mL (< 35), cancer antigen 19-9 of 19 U/mL (< 40), carcinoembryonic antigen of 2.7 ng/mL (< 2.5) and human epididymis protein 4 of 33 pmol/L (0-150). Patient underwent a laparoscopic appendectomy with partial cecectomy with pathology showing low-grade appendiceal mucinous neoplasm (LAMN) (Figure 2). Her postoperative course was uncomplicated and no adjuvant therapy was warranted. Although there is no way to prove that the mass seen at age 31 years and then at 34 years are the same, this is highly suggested by similar size, appearance and location.
.
Figure 1 Images of appendiceal mucocele. (A) Transvaginal ultrasound shows pelvic mass with echogenic layering referred to as the onion skin sign; (B) Pelvic MRI showing increased T2 signal of the appendiceal mucocele (large arrow) adjacent to the uterus (small arrow); (C) Laparoscopic view of appendiceal mucocele.
View Figure 1
.
Figure 2 Pathology of appendiceal mucocele. (A) Gross specimen showing dilated appendix; (B) Histopathology of low grade mucinous neoplasm of the appendix characterized by a villiform mucinous epithelial proliferation replacing the mucosa and lack of muscularis destruction. (Photomicrograph courtesy of Dr. Cynthia D. Guy, Department of Pathology, Duke University).
View Figure 2
Discussion
This case illustrates very practical issues for investigation of a possible asymptomatic appendiceal mass in pregnancy. The enlarging uterus results in upward displacement of the appendix and rotation of the cecum such that the base of the cecum is anteriorly and outwardly displaced [3]. In pregnancy the location of the appendix may vary considerably but median locations include at the level of L5 in the 1st trimester, L4 in the 2nd trimester and close to L3 in the 3rd trimester. In our case the mass was incidentally found during an anatomy scan, as it is common practice to evaluate the adnexa. The upward movement of the appendix resulted in easy identification to the right of the uterine fundus. In contrast the mass was not seen during an abdominal ultrasound in radiology. Although the American Institute of Ultrasound in Medicine (www.aium.org) recommends bowel evaluation, including the appendix during abdominal scans, this is not commonly done unless specifically requested. More commonly, evaluation is performed of the liver, gallbladder and biliary tract, pancreas, spleen and aorta. Although it is not possible to prove that the mass seen during pregnancy and that seen 3 years later are the same; it is highly suggested given the same location, similar size and appearance.
Appendiceal mucoceles often have a classic appearance on ultrasound of multiple echogenic layers referred to as "onion skin-like" circles. An outer diameter of greater than 15 mm is highly predictive of an appendiceal mucocele with a sensitivity of 83% and specificity of 92% [4]. Computed tomography (CT) shows a well-encapsulated low intensity mass in which wall calcification may be seen, whereas MRI shows a well-circumscribed mass with low signal intensity on T1-weighted images and high signal intensity on T2-weighted images.
The pathologic classification of appendiceal mucocele is not uniform and recommendations for consensus were recently published by the Peritoneal Surface Oncology Group International [5]. Low-grade appendiceal mucinous neoplasm (LAMN) is characterized by undulating or flattened epithelium of the mucosa with minimal cytologic atypia growing into the overlying layers of submucosal connective tissue (stroma), muscle layers or through the serosa. This "pushing" invasion lacks the destructive characteristics of carcinoma. High-grade appendiceal mucinous neoplasm (HAMN) has the same characteristics except with increased atypia. Mucinous adenocarcinoma differs by infiltrative invasion characterized by tumor budding of single cells or groups of cells into the stroma or beyond. A desmoplastic response in the stroma consists of fibrosis with a proteoglycan-rich extracellular matrix. Mucinous adenocarcinoma may be divided into well, moderately or poorly differentiated with or without significant presence of signet ring cells [5]. LAMN and HAMN are found 4-5 times more often than adenocarcinoma [6]. Although not totally proven, there is the concern that LAMN may progress to cystadenocarcinoma with time, as seen with colon adenomas progressing to carcinomas [7].
LAMN restricted to the appendix by pathology with tumor free margins after surgery is benign with no risk of subsequent recurrence [8]. Approximately 20% of patients with LAMN with evidence of extra-appendiceal cells or mucin will develop pseudomyxoma peritonei (PMP) characterized by the accumulation of cells or mucin in the abdominal cavity [9]. In a study of 18 patients with LAMN and negative pelvic cytology, 28% with histological evidence of extra-appendiceal acellular mucin developed PMP while 40% with extra-appendiceal cells developed PMP [10]. PMP is classified as acellular mucin if cells are not present, low-grade mucinous carcinoma peritonei or disseminated peritoneal adenomucinosis (DPAM) characterized by cells with low-grade histologic features, high-grade mucinous carcinoma peritonei or peritoneal mucinous carcinomatosis (PMCA) characterized by cells with high-grade histologic features and high-grade mucinous carcinoma peritonei with signet ring cells or peritoneal mucinous carcinomatosis with signet ring cells [5]. Typical 5-year survival rates for DPAM are PCMA are 75% and 14% respectively [7].
The described patient was found to have a borderline elevated carcinoembryonic antigen (CEA) [11]. Most cases of LAMN even with localized extra-appendiceal mucin from perforation, have normal markers including CEA, cancer antigen 19-9 (CA-19-9) and cancer antigen 125 (CA-125) [10]. In contrast, approximately 75% of PMP cases have an elevation in one of these three markers [12] with CEA having the highest baseline sensitivity at 72.6% in a study of 62 patients with PMP [11]. Elevated baseline tumor markers correlate with lower disease free survival time after cytoreductive surgery with heated intraperitoneal chemotherapy [12]. The surgical management of appendiceal mucocele depends upon the need to obtain negative margins. Cases without evidence of extension to the appendiceal base or outside the appendix may be approached with appendectomy. Instances with possible involvement of the appendiceal base require appendectomy with complete removal of the base possibly requiring partial cecectomy preserving the ileo-cecal valve. A laparoscopic verses open approach depends upon the surgeon's capabilities and the amount of disease. Complete removal of disease without gross spill is the goal. Abdomino-pelvic washings for cytology should be considered in all cases but is needed with any evidence of mucin within the pelvis. More aggressive surgery with right hemicolectomy is used with spread of disease to the colon, for lymph node involvement and with signet ring adenocarcinoma. In cases of intraperitoneal spread without gross disease in the colon, there is no evidence that right hemicolectomy improves survival compared to appendectomy [13].
Evidence of disease outside of the appendix or of PMP requires cytoreductive surgery with heated intraperitoneal chemotherapy (CRS-HIPEC). The goal of CRS is to remove all macroscopic disease and the omentum.
Appropriate follow-up of LAMN continues to be an area of debate. Most agree that lesions restricted to the appendix without microscopic perforation or extra-appendiceal mucin or cells may be conservatively managed with observation. A definitive plan for lesions with microscopic perforation or extra-appendiceal mucin or cells, but negative pelvic cytology, continues to be debated. Options include long-term surveillance for up to 5 years with semi-annual CT or MRI imaging and tumor markers verses cytoreductive surgery with heated intraperitoneal chemotherapy. The disadvantage of the former is the time consuming testing and the approximate 10% recurrences after 5 years; whereas the latter exposes 60-80% of patients to invasive surgery without need [14,15].
In conclusion, this case of delayed diagnosis of an appendiceal mucocele during pregnancy brings out several teaching points. The upward deviation of the appendix during pregnancy rendered the appendiceal mass visible during an obstetrical anatomy sonography. The mass was not visualized on a confirmation abdominal ultrasound as the technique commonly targets specific organs not including the bowel or appendix unless specifically requested. Delayed diagnosis for several years may have increased the risk of progression to PMP resulting in a more serious clinical situation.
Acknowledgements
Photomicrograph courtesy of Dr. Cynthia D. Guy, Department of Pathology, Duke University.
References
-
Idris LO, Olaofe OO, Adejumobi OM, Kolawole AO, Jimoh AK (2015) Giant mucocele of the appendix in pregnancy: A case report and review of literature. Int J Surg Case Rep 9: 95-97.
-
Haase E, Yoo D, Sugarbaker PH (2009) Management of appendiceal pseudomyxoma peritonei diagnosed during pregnancy. World J Surg Oncol 7: 48.
-
Lee KS, Rofsky NM, Pedrosa I (2008) Localization of the appendix at MR imaging during pregnancy: utility of the cecal tilt angle. Radiology 249: 134-141.
-
Lien WC, Huang SP, Chi CL, Liu KL, Lin MT, et al. (2006) Appendiceal outer diameter as an indicator for differentiating appendiceal mucocele from appendicitis. Am J Emerg Med 24: 801-805.
-
Carr N, Cecil T, Mohamed F, Sobin L, Sugarbaker P, et al. (2016) A Consensus for Classification and Pathologic Reporting of Pseudomyxoma Peritonei and Associated Appendiceal Neoplasia: The Results of the Peritoneal Surface Oncology Group International (PSOGI) Modified Delphi Process. Am J Surg Pathol 40: 14-26.
-
Kim SH, Lim HK, Lee WJ, Lim JH, Byun JY (1998) Mucocele of the appendix: ultrasonographic and CT findings. Abdom Imaging 23: 292-296.
-
Misdraji J (2015) Mucinous epithelial neoplasms of the appendix and pseudomyxoma peritonei. Mod Pathol 28: S67-79.
-
Misdraji J, Yantiss RK, Graeme-Cook FM, Balis UJ, Young RH (2003) Appendiceal Mucinous Neoplasms: A Clinicopathologic Analysis of 107 Cases. Am J Surg Pathol 27: 1089-1103.
-
Smeenk RM, van Velthuysen MLF, Verwaal VJ, Zoetmulder FAN (2008) Appendiceal neoplasms and pseudomyxoma peritonei: A population based study. Eur J Surg Oncol 34: 196-201.
-
Foster JM, Sleightholm RL, Wahlmeier S, Loggie B, Sharma P, et al. (2016) Early identification of DPAM in at-risk low-grade appendiceal mucinous neoplasm patients: a new approach to surveillance for peritoneal metastasis. World J Surg Oncol 14: 243.
-
Baratti D, Kusamura S, Martinetti A, Seregni E, Laterza B, et al. (2007) Prognostic Value of Circulating Tumor Markers in Patients with Pseudomyxoma Peritonei Treated with Cytoreductive Surgery and Hyperthermic Intraperitoneal Chemotherapy. Ann Surg Oncol 14: 2300-2308.
-
Taflampas P, Dayal S, Chandrakumaran K, Mohamed F, Cecil TD, et al. (2014) Pre-operative tumour marker status predicts recurrence and survival after complete cytoreduction and hyperthermic intraperitoneal chemotherapy for appendiceal Pseudomyxoma Peritonei: Analysis of 519 patients. Eur J Surg Oncol 40: 515-520.
-
Barrios P, Losa F, Gonzalez-Moreno S, Rojo A, Gomez-Portilla A, et al. (2016) Recommendations in the management of epithelial appendiceal neoplasms and peritoneal dissemination from mucinous tumours (pseudomyxoma peritonei). Clin Transl Oncol 18: 437-448.
-
Fish R, Selvasekar C, Crichton P, Wilson M, Fulford P, et al. (2014) Risk-reducing laparoscopic cytoreductive surgery and hyperthermic intraperitoneal chemotherapy for low-grade appendiceal mucinous neoplasm: early outcomes and technique. Surg Endos 28: 341-345.
-
McDonald JR, O'Dwyer ST, Rout S, Chakrabarty B, Sikand K, et al. (2012) Classification of and cytoreductive surgery for low-grade appendiceal mucinous neoplasms, Br J Surg 99: 987-992.





