Obstetrics and Gynaecology Cases - Reviews
Dextroposition of the Heart Reflected Mosaic Fetal Trisomy 18
Mohamed A Satti1,2*, Frederico G Rocha1,2, Erin Beaver3 and Jeffrey M Dicke3
1Maternal Fetal Medicine, Department of Obstetrics and Gynecology, Hurley Medical Center, USA
2Michigan State University, College of Human Medicine, USA
3Maternal Fetal Medicine, Department of Obstetrics and Gynecology, Washington University, USA
*Corresponding author: Mohamed Satti, MD, Maternal Fetal Medicine, Department of Obstetrics and Gynecology, Hurley Medical Center/Michigan State University 2158, Fox Hill Dr, Apt 10 Grand Blanc, Michigan 48439, USA, Tel: 408-207-3278, E-mail: satti97@gmail.com
Obstet Gynecol Cases Rev, OGCR-3-086, (Volume 3, Issue 3), Case Report; ISSN: 2377-9004
Received: February 05, 2016 | Accepted: April 25, 2016 | Published: May 02, 2016
Citation: Satti MA, Rocha FG, Beaver E, Dicke JM (2016) Dextroposition of the Heart Reflected Mosaic Fetal Trisomy 18. Obstet Gynecol Cases Rev 3:086. 10.23937/2377-9004/1410086
Copyright: © 2016 Satti MA, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Introduction: Trisomy 18 in every cell is associated with multiple congenital anomalies.
Case: Anatomy scan of a 22 year old prima gravida at 19w4d showed a viable IUP with unilateral choroid plexus cyst and dextroposition of the heart without any other observed structural malformation. Amniocentesis revealed a normal male pattern for chromosomes 13, 18, 21, X and Y by fluorescence in situ hybridization (FISH). However, chromosomal microarray analysis (CMA) for circulating fetal DNA in maternal blood was consistent with mosaic Trisomy 18. The karyotype of cultured dividing cells confirmed 47, XY, +18/46, XY mosaicism.
Conclusion: Dextroposition of the heart (dextrocardia) identified by ultrasound prompted fetal DNA screening plus subsequent amniocyte analysis that together confirmed mosaic fetal aneuploidy.
Introduction
Trisomy 18 or Edwards's syndrome is a rare chromosomal anomaly due to the presence of an extra chromosome 18 defining full or mosaic trisomy 18 [1]. The first reported infants were described in 1960 by Edwards et al. and Smith et al. [2,3] resulting in 1 in 5500 live births [4,5] with a 3:1 female to male ratio. The abnormality results from meiotic nondisjunction that is enhanced in frequency by advanced maternal age. The ~10% of trisomy 18 fetuses that survive to birth have substantially reduced survival with a median survival range of 3 to 14.5 days [1,6,7].
Trisomy 18 is typically associated with multiple congenital anomalies. Currently in the North America and Europe most cases of trisomy 18 are prenatally diagnosed, based on screening by maternal age, maternal serum marker screening, or detection of sonographic abnormalities during the second and third trimester [8,9]. The prenatal sonographic pattern of trisomy 18 is characterized by growth restriction, polyhydramnios, "strawberry-shaped" cranium (brachycephaly and narrow frontal cranium), choroid plexus cysts, overlapping of hands fingers (second and fifth on third and fourth respectively), congenital heart defects, omphalocele, and single umbilical artery [9-11]. Many studies show that 80%-100% of patients with trisomy 18 have congenital structural heart defects. In these fetuses, the common heart defects detected by prenatal ultrasonography are ventricular septal defect, patent ductus arteriosus, atrial septal defect and complex congenital heart defects [12-15]. The most common brain lesions observed by prenatal ultrasound are cerebellar hypoplasia, mega cisterna magna and choroid plexus cysts [12].
Cardiac malposition is a rare but important finding when detected on fetal ultrasound. Dextroposition of the heart is a congenital anomaly in which the heart is positioned in the right chest but normally oriented, with the cardiac apex pointing to the left. Dextroposition of the heart is almost always associated with intrathoracic malformations. The most common malformations are: left congenital diaphragmatic hernia (75%), cystic adenomatoid malformations located in the left chest (19%), and other pulmonary malformations (6%) [16].
Case
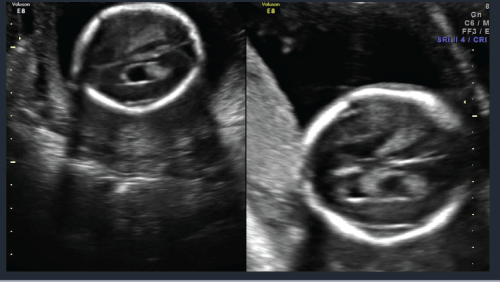
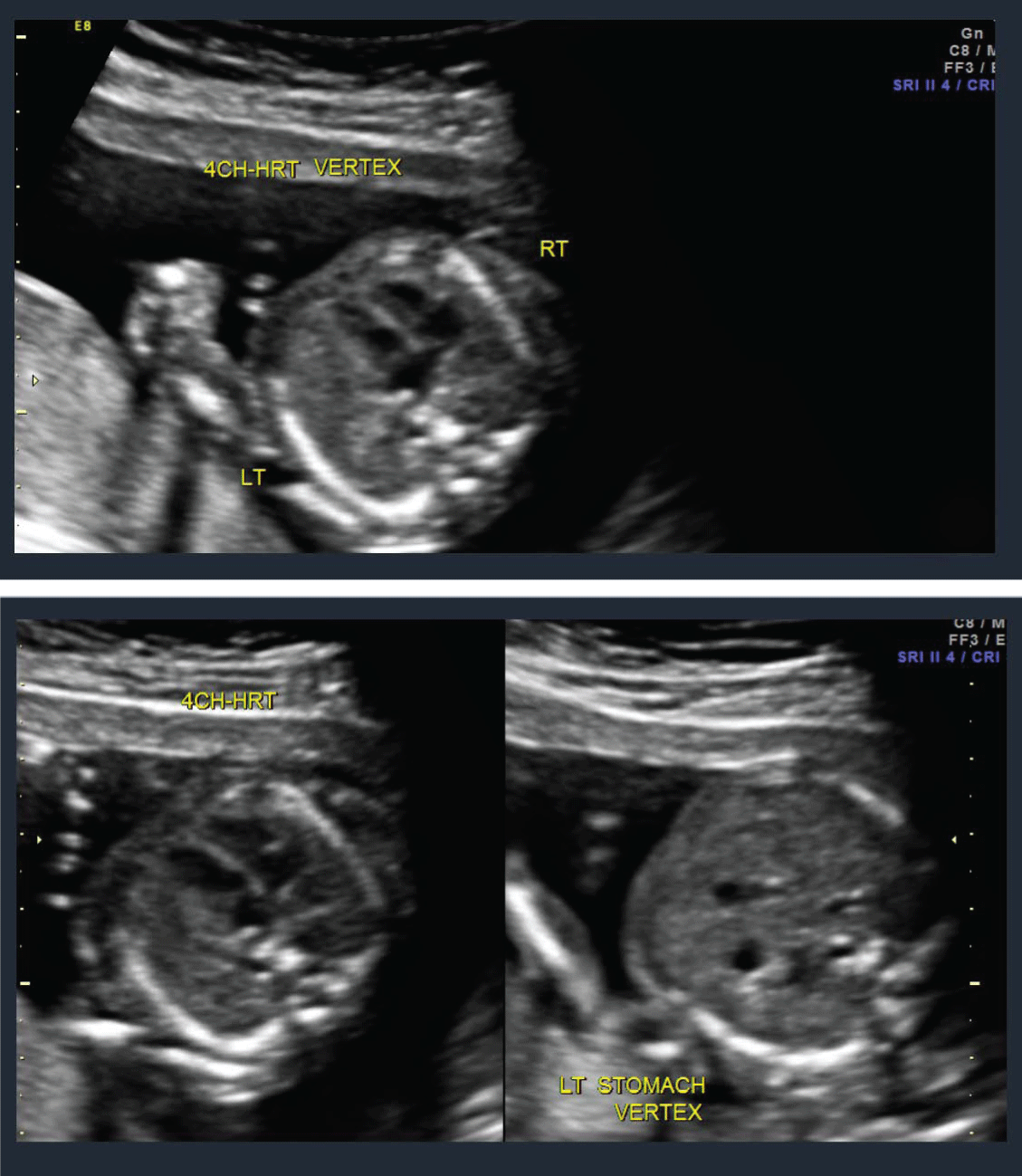
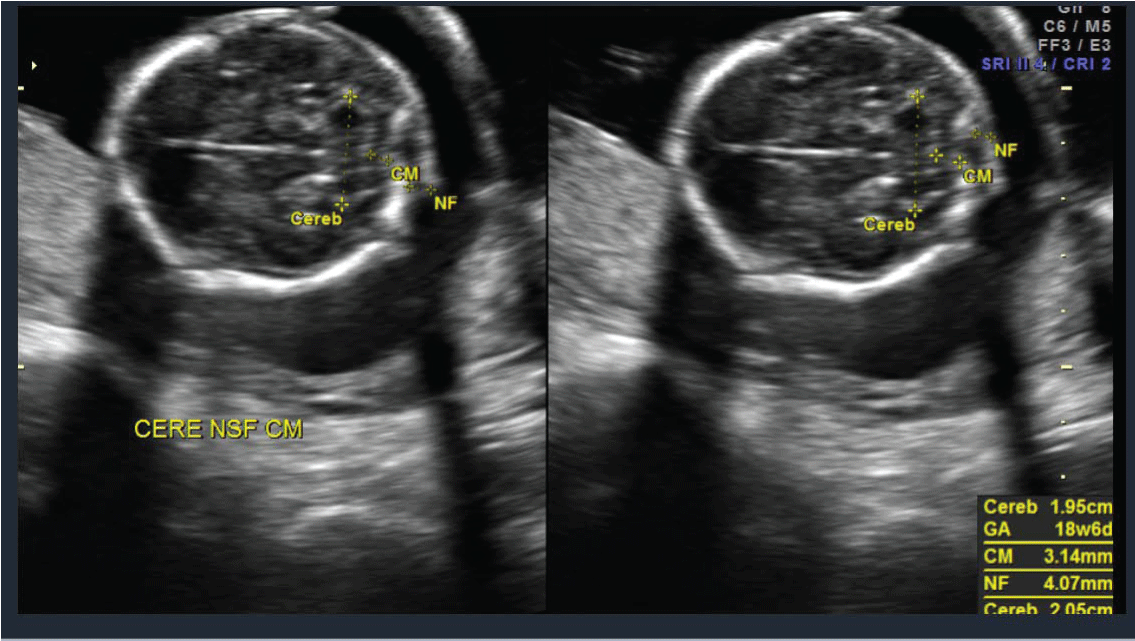
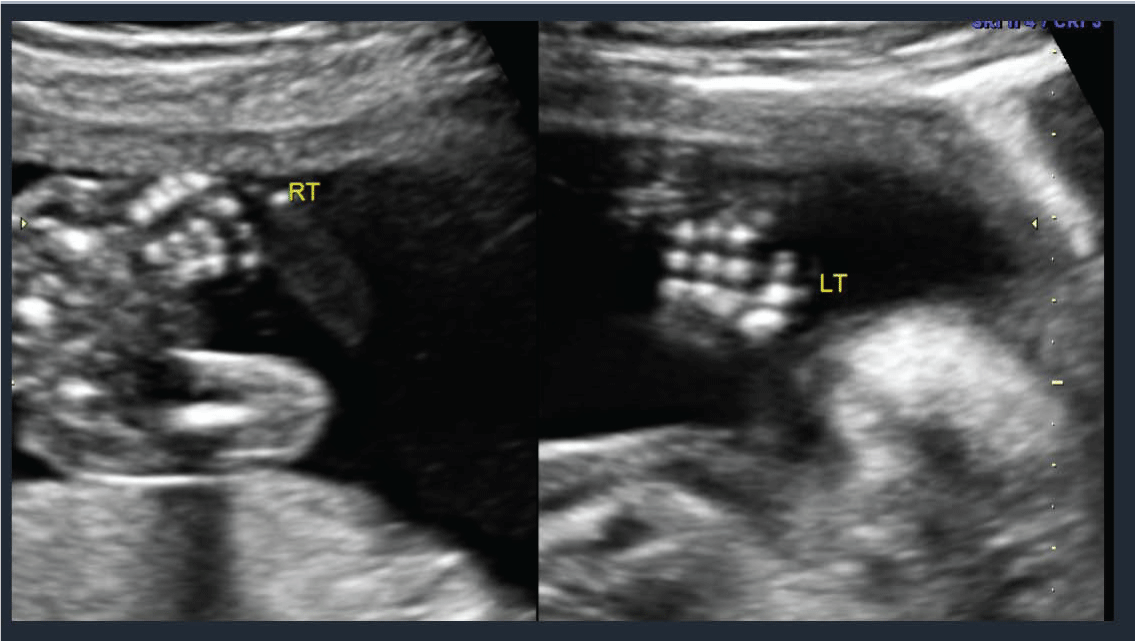
A 22 year old prima gravida patient received a viability ultrasound in the Obstetrics and Gynecology Department at 13 weeks 4 days to test for any obvious phenotypic abnormalities. Simultaneously she declined any aneuploidy screening. Subsequently she was completed regular prenatal visits in the antenatal care clinic. She had her next ultrasound at 19 weeks 4 days at which time she also had a mid-trimester anatomy scan. The viable intrauterine pregnancy was noted to be accompanied by a unilateral choroid plexus cyst (Figure 1) along with dextroposition of the heart (Figure 2). The fetal heart was in the right hemithorax without discrete mass lesions in the left hemithorax while the stomach was appropriately positioned in the left upper quadrant and the diaphragm appeared intact. No complex cardiac defects or other structural malformations were identified (Figure 3 and Figure 4). Estimated fetal weight was appropriate for gestational age (Figure 5). These findings were discussed and all prenatal testing options were reviewed with the patient and her spouse. At 20 weeks 4 days a repeated ultrasound showed isolated dextroposition of the heart with the choroid plexus cyst no longer being evident. At that time the patient requested and completed amniocentesis. Amniotic fluid alpha fetoprotein levels were also normal. Initial cytogenetic analysis for common chromosome aneuploidy revealed a normal male pattern for chromosomes 13, 18, 21, X and Y by fluorescence in situ hybridization (FISH). However, the chromosomal microarray analysis (CMA) suggested mosaic Trisomy 18. An abbreviated karyotype subsequently confirmed three copies of chromosome 18 in three out of four metaphase cells confirming 47,XY,+18/46,XY mosaicism. The patient elected to terminate the pregnancy based upon these results, Dilation and evacuation (D&E) was completed at 22 weeks and 4 days. The autopsy revealed: Normal heart in shape and size. Left pulmonary artery, aortic trunk with valve and atrial septum are disrupted. The right pulmonary artery is intact and normal. The left atrial appendages are normal in shape and consistent with normal isomerism. The mitral, pulmonic and tricuspid valves are normal. There is no ventricular septal defect. Heart anomalies are not identified. However, the examination was suboptimal due to fetal disruption secondary to the (D&E) procedure.
Conclusion
Mosaicism reported in ~2% of all CVS biopsies is confined to the placenta in ~4 of 5 cases and modifies fetal cells in the remaining ~1 in 5 cases [17]. Mosaicism can involve all chromosome abnormalities from whole chromosome aneuploidy to aneuploidy gene on chromosome regions. Chromosome karyotypes most reliably illustrate visible chromosome abnormalities in the clearly banded proportion of mosaic cells in the sampled dividing cellular tissue. In contrast, fluorescence in situ hybridization may not detect a very low proportion of mosaicism in poor quality interphase nuclei. The proportion of mosaicism in each result depends upon sample quality and available time to isolate mitotic cells.
The trisomy 18 amniocyte karyotypes confirm the fetus has true trisomy 18 mosaicism. A wide diversity of sonographic abnormalities has been reported for Trisomy 18 during positive mid-trimester anatomic surveys. However, no known case had been reported for heart dextroposition reflecting mosaic trisomy 18. Our future identification of heart dextroposition will include consideration of testing for fetal gene aneuploidy.
Conflicts of Interest
None of the authors have any conflicts of interest.
Precise
This article demonstrates true trisomy 18 fetal mosaicism with an abnormal cardiac phenotype.
References
-
Embleton ND, Wyllie JP, Wright MJ, Burn J, Hunter S (1996) Natural history of trisomy 18. Arch Dis Child Fetal Neonatal Ed 75: 38-41.
-
Edwards JH, Harnden DG, Cameron AH, Crosse VM, Wolff OH (1960) A new trisomic syndrome. Lancet 7128: 787-790.
-
Smith DW, Patau K, Therman E, Inhorn SL (1960) A new autosomal trisomy syndrome: multiple congenital anomalies caused by an extra chromosome. J Pediatr 57: 338-345.
-
Kosho T, Nakamura T, Kawame H, Baba A, Tamura M, et al. (2006) Neonatal management of trisomy 18: clinical details of 24 patients receiving intensive treatment. Am J Med Genet A 140: 937-944.
-
Jones KL (2006) Smith's recognizable patterns of human malformation, 6th Ed Elsevier Saunders, Philadelphia.
-
Rasmussen SA, Wong LY, Yang Q, May KM, Friedman (2003) Population-based analyses of mortality in trisomy 13 and trisomy 18. JM Pediatrics 111: 777-784.
-
Root S, Carey JC (1994) Survival in trisomy 18. Am J Med Genet 49: 170-174.
-
Irving C, Richmond S, Wren C, Longster C, Embleton ND (2011) Changes in fetal prevalence and outcome for trisomies 13 and 18: a population-based study over 23 years. J Matern Fetal Neonatal Med 24: 137-141.
-
Yamanaka M, Setoyama T, Igarashi Y, Kurosawa K, Itani Y, et al. (2006) Pregnancy outcome of fetuses with trisomy 18 identified by prenatal sonography and chromosomal analysis in a perinatal center. Am J Med Genet 140: 1177-1182.
-
Sepulveda W, Wong AE, Dezerega V (2010) First-trimester sonographic findings in trisomy 18: a review of 53 cases. Prenat Diagn 30: 256-259.
-
Cho RC, Chu P, Smith-Bindman R (2009) Second trimester prenatal ultrasound for the detection of pregnancies at increased risk of Trisomy 18 based on serum screening. Prenat Diagn 29:129-139.
-
Lin HY, Lin SP, Chen YJ, Hung HY, Kao HA, et al. (2006) Clinical characteristics and survival of trisomy 18 in a medical center in Taipei, 1988-2004. Am J Med Genet A 140: 945-951.
-
Carey JC (2010) Trisomy 18 and trisomy 13 syndromes. In: Cassidy SB, Allanson JE, Management of genetic syndromes. (3rd edn), John Wiley & Sons, New York 807-823.
-
Van Praagh S, Trumen T, Firpo A, Bano-Rogriga A, Freid R, et al. (1989) Cardiac malformations in trisomy-18: a study of 41 postmortem cases. J Am Coll Cardiol 13: 1586-1597.
-
Musewe NN, Alexander DJ, Teshima I, Smalhorn JF, Freedom RM (1990) Echocardiographic evaluation of the spectral cardiac anomalies associated with trisomy 18 and 13. J Am Coll Cardiol 15: 673-677.
-
Falkensammer CB, Ayres NA, Altman CA, Ge S, Bezold Li (2008) Fetal cardiac malposition: incidence and outcome of associated cardia and extracardiac malformation. Am J perinatal 25: 277-281.
-
Lebo RV, Novak RW, Wolfe K, Michelson M, Robinson H, et al. (2015) Discordant circulating fetal DNA and subsequent cytogenetics reveal false negative, placental mosaic, and fetal mosaic cfDNA genotypes. Journal of Translational Medicine 13: 260.