Obstetrics and Gynaecology Cases - Reviews
Amniotic Fluid Embolism: Combined Treatment with Surgery and Extracorporeal Membrane Oxygenation Support - A Case Report
Arnon Wiznitzer1*, Rinat Gabbay-Benziv1, Eran Altman1, Dan Aravot2 and Philippe Biderman3
1Helen Schneider Hospital for Women, Rabin Medical Center, Petach-Tikva, Israel
2Cardiac Surgery department, Rabin Medical Center, Petach-Tikva, Israel
3Cardiothoracic surgery intensive care unit, Rabin Medical Center, Petach-Tikva, Israel
*Corresponding author: Arnon Wiznitzer, MD, Department of Obstetrics and Gynecology, Helen Schneider Hospital for Women, Rabin Medical Center, Petah Tiqwa 49100, Israel, Tel: +972-3-9377680, E-mail: ArnonW@clalit.org.il
Obstet Gynecol Cases Rev, OGCR-3-082, (Volume 3, Issue 2), Case Report; ISSN: 2377-9004
Received: December 20, 2015 | Accepted: March 24, 2016 | Published: March 28, 2016
Citation: Wiznitzer A, Gabbay-Benziv R, Altman E, Aravot D, Biderman P (2016) Amniotic Fluid Embolism: Combined Treatment with Surgery and Extracorporeal Membrane Oxygenation Support - A Case Report. Obstet Gynecol Cases Rev 3:082. 10.23937/2377-9004/1410082
Copyright: © 2016 Wiznitzer A, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Keywords
Amniotic fluid embolism, ECMO, Circulatory collapse, Pregnancy
Case Description
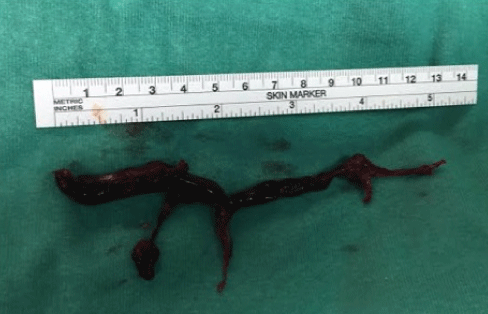
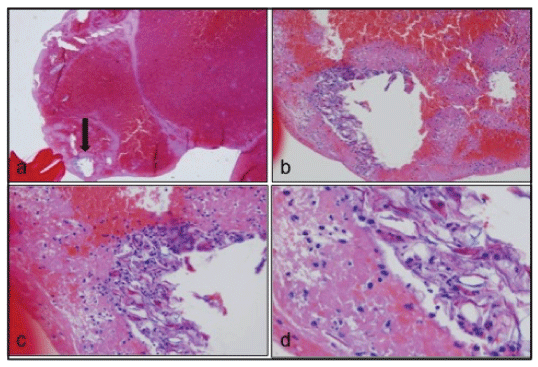
A 42-year-old gravida 1, 40-weeks' gestation, presented with rupture of membranes in labor. Her history and prenatal care were unremarkable except for obesity. On physical examination, blood pressure was 156/91 mmHg with no other signs of pre-eclamsia. Due to non-progression at active labor, cesarean section was performed with extended "top-up" epidural and delivery of a healthy boy. Immediately after removal of the placenta, the patient reported chest pain, and became unresponsive. ECG (electrocardiography) demonstrated asystole. The trachea was intubated, and cardiopulmonary resuscitation had begun, with closed chest compressions and administration of vasopressors. After 12 minutes of rigorous resuscitation spontaneous pulse returned. Abdomen was closed quickly with no active bleeding. Trans-esophageal echocardiography demonstrated decreased right ventricle contractility. Mild consumption coagulopathy was treated with coagulation factors. Two hours later, patient's vitals deteriorated and her blood pressure dropped despite continuous vasopressors use. At that time, as the woman was no longer hemodynamically stable, decision of re-laparotomy with hysterectomy was made followed by connection to venous-arterial extracorporeal oxygenation (VA-ECMO) support. The next day, after hemodynamic stabilization was achieved, CT angiography scan revealed a filling defect in the right pulmonary artery and open thoracotomy with excision of a 12-cm clot from the right pulmonary artery (Figure 1) was performed. After surgery, the patient's cardiac function gradually improved and she was weaned off the ECMO. Twenty-nine days later, she was discharged to rehabilitation center, neurologically intact. Histology of the thrombi revealed fetal debris that was compatible with amniotic fluid embolism (AFE) (Figure 2).
.
Figure 2: Histological examination of the thrombi. Histological examination of the thrombus removed from maternal pulmonary vasculature. (a) demonstrates small area suspected as fetal debris (arrow); (b-d) confirm amniotic fluid embolism showing squamous cells and fetal debris in the suspected area with magnification.
View Figure 2
Comment
AFE is thought to result from immune-mediated maternal response to fetal debris entering the pulmonary vasculature causing anaphylactoid like reaction [1]. Although rare, it is well known and feared by obstetricians [2,3] as it is one of the top five leading causes of direct obstetrics deaths in developed countries. Busardo et al. examined even different national registries and found that 12.8% (range 4.7-24.3%) of maternal deaths were caused by fatal AFE [4]. The diagnosis is essentially a clinical diagnosis of exclusion. The classical triad of respiratory distress, cardiovascular collapse and coagulopathy makes it easy to diagnose the classical form. In the atypical form, coagulopathy occurs in the absence of cardiopulmonary manifestations [5]. Currently, there is no confirmatory laboratory test for the diagnosis of AFE. TKH-2 antibody, sialyl-Tn (STN), tryptase and zinc coproporphyrin in maternal plasma was all previously linked to AFE [6-8]. The finding of amniotic fluid debris in the maternal pulmonary microvasculature is considered highly suggestive, especially when fetal squamous cells/debris and neutrophils are found in blood samples collected through the distal port of a pulmonary artery catheter [9,10]. However, this evidence, while supportive, is not definitive, as some investigators describe the amniotic fluid components to be commonly present in maternal circulation without clinical evidence of AFE syndrome [11-13].
In our case, AFE was diagnosed clinically close to the event and was supported by histological finding of fetal debris in the thrombi removed from the pulmonary trunk. The treatment of AFE is mainly supportive [1]. Extraordinary measures, including cardiopulmonary bypass and surgical removal of the large thrombi resulted in survival of our patient. The decision of hysterectomy enabled the connection to VA-ECMO with lower risk of bleeding while on anticoagulation. To the best of our knowledge, this is one of the few cases previously described [14,15] where combination of ECMO and pulmonary thromboembolectomy lead to survival in an almost inevitable death situation.
References
-
Conde-Agudelo A, Romero R (2009) Amniotic fluid embolism: an evidence-based review. Am J Obstet Gynecol 201: 445.
-
Say L, Chou D, Gemmill A, Tunçalp Ö, Moller AB, et al. (2014) Global causes of maternal death: a WHO systematic analysis. Lancet Glob Health 2: e323-333.
-
Clark SL, Hankins GD, Dudley DA, Dildy GA, Porter TF (1995) Amniotic fluid embolism: analysis of the national registry. Am J Obstet Gynecol 172: 1158-1167.
-
Busardo FP, Frati P, Karch SB, Fineschi V1 (2014) Epidemiological and clinical data of amniotic fluid embolism in forensic settings. Curr Pharm Biotechnol 14: 1189-1194.
-
Clark SL (2014) Amniotic fluid embolism. Obstet Gynecol 123: 337-348.
-
Kanayama N, Yamazaki T, Naruse H, Sumimoto K, Horiuchi K, et al. (1992) Determining zinc coproporphyrin in maternal plasma--a new method for diagnosing amniotic fluid embolism. Clin Chem 38: 526-529.
-
Oi H, Kobayashi H, Hirashima Y, Yamazaki T, Kobayashi T, et al. (1998) Serological and immunohistochemical diagnosis of amniotic fluid embolism. Semin Thromb Hemost 24: 479-484.
-
Nishio H, Matsui K, Miyazaki T, Tamura A, Iwata M, et al. (2002) A fatal case of amniotic fluid embolism with elevation of serum mast cell tryptase. Forensic Sci Int 126: 53-56.
-
Rhodes A, Cusack RJ, Newman PJ, Grounds RM, Bennett ED (2002) A randomised, controlled trial of the pulmonary artery catheter in critically ill patients. Intensive Care Med 28: 256-264.
-
Masson RG, Ruggieri J (1985) Pulmonary microvascular cytology. A new diagnostic application of the pulmonary artery catheter. Chest 88: 908-914.
-
Clark SL, Pavlova Z, Greenspoon J, Horenstein J, Phelan JP (1986) Squamous cells in the maternal pulmonary circulation. Am J Obstet Gynecol 154: 104-106.
-
Lee W, Ginsburg KA, Cotton DB, Kaufman RH (1986) Squamous and trophoblastic cells in the maternal pulmonary circulation identified by invasive hemodynamic monitoring during the peripartum period. Am J Obstet Gynecol 155: 999-1001.
-
Busardo FP, Gulino M, Di Luca NM, Vergallo GM, Pacchiarotti A, et al. (2014) Not only a clinical nightmare: amniotic fluid embolism in court. Curr Pharm Biotechnol 14: 1195-1200.
-
Sharma NS, Wille KM, Bellot SC, Diaz-Guzman E (2015) Modern use of extracorporeal life support in pregnancy and postpartum. ASAIO J 61: 110-114.
-
Shen HP, Chang WC, Yeh LS, Ho M (2009) Amniotic fluid embolism treated with emergency extracorporeal membrane oxygenation: a case report. J Reprod Med 54: 706-708.