Obstetrics and Gynaecology Cases - Reviews
Postpartum Low Back Pain: It is not always What You Think
Melissa Corso1*, Diane Grondin2 and Carol Ann Weis3
1Intern, BSc, MSc, Canadian Memorial Chiropractic College, Canada
2Assistant Professor, Graduate Education and Research Programs, Canadian Memorial Chiropractic College, Canada
3Researcher, Graduate Education and Research Programs, Canadian Memorial Chiropractic College, Canada
*Corresponding author: Melissa Corso, Intern, BSc, MSc, Canadian Memorial Chiropractic College, Canada, Tel: 416-482-2340, E-mail: mcorso@cmcc.ca
Obstet Gynecol Cases Rev, OGCR-3-079, (Volume 3, Issue 2), Case Report; ISSN: 2377-9004
Received: November 06, 2015 | Accepted: March 15, 2016 | Published: March 18, 2016
Citation: Corso M, Grondin D, Weis CA (2016) Postpartum Low Back Pain: It is not always What You Think. Obstet Gynecol Cases Rev 3:079. 10.23937/2377-9004/1410079
Copyright: © 2016 Corso M, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Background: Postpartum back pain is common, with up to 75% of women experiencing back pain immediately following birth. This pain can be moderately to severely debilitating to the new mom, affecting activities of daily living such as caring for her new born, sleep patterns, and otherhousehold activities. However, all low back pain is not created equal and we document a case study with an unusual cause of postpartum back pain.
Case report: A 37-year old female presented with right-sided low back pain that began 3 days following the birth of her child. Pain was 9 out of 10 on the visual analog scaleandmost activities of daily living were compromised. During the birth, the patient had an epidural at 9 cm to help progress to the last centimetre of dilation. Hospital records indicate there were no issues with the injection or injection site. At 3 days postpartum, the area of injection started to swell and a number of days later, a mass had formed. At about 5 weeks postpartum, the result of magnetic resonance imaging determined the cause of the patient's back pain.
Conclusion: Postpartum back pain may not actually be related to pregnancy itself or the act of delivering a child. There are other differential diagnoses that health care practitioners should consider when examining and diagnosing the new mom.
Introduction
Low back pain (LBP) and/or pelvic girdle pain (PGP) has a prevalence of 20-90% in the pregnant population, while a small number of women may suffer from a combination of both pains [1-9]. While PGP is typically more common and intense during pregnancy, LBP is more intense and common in the postpartum period [10]. In fact, up to 75% of women who suffer from pregnancy-related back pain may continue to have pain after giving birth [1-3,11-13]. While the majority of cases resolve within 6 months postpartum [14], 40% may continue to experience pain beyond 6 months [15]. For those with a history of LBP during pregnancy, LBP seems to decrease over the postpartum period. However, women who experience LBP or PGP at 3 months postpartum were found to be at higher risk for persistent or chronic LBP [14]. Of these women, only 6% recover within 6-18 months after giving birth [14].
Researchers have been unable to identify etiologic factors relating to postpartum LBP and PGP [8], and the simple act of having a baby may not be the only cause of pain at this time. The literature has reported rare cases of sacral stress fractures during and after pregnancy [16,17], postpartum pyogenic sacroiliitis [17], and osteomyelitis of the pubic symphisis [18] as postpartum causes of LBP or PGP. In addition, a more common cause of postpartum low back or pelvic pain includes pregnancy-related and lactation associated spinal osteoporosis [19,20]. Common differential diagnoses to consider in this population include, but are not limited to: arthritis of the spine or hip, sciatica or osteitis pubis, lumbar disc pathology and spondylolisthesis. Other less commondifferential diagnoses include urinary tract infection, lumbar stenosis, femoral venous thrombosis, osteomyelitis, cauda equina syndrome or ruptured symphysis pubis. The purpose of this paper is to demonstrate an atypical cause of postpartum back pain and its debilitating nature that could have resulted in a much worse outcome had the patient not been persistent in determining the cause of the pain.
Case Report
A 37-year-old female presented with right-sided low back pain at the L3-4 level that began 3 days after giving birth. The patient reported that the pain was excruciating and continued to progress until it was rated 9/10 on the visual analog scale (VAS). Over the course of 7 days, the painbecame bilateral and was described as a constant ache that was sharp with certain activities. The painintermittently “wrapped” around the hip and gluteal region especially on the right side. The patient reported that standing upright was painful and assuming a flexed posture was relieving. Activities of daily living were compromised; the patient was only able to walk short distances at a slow pace andcould lift a maximum of 10 lbs for a short period of time. Therefore, she was unable to properly care for her newborn. Sitting was uncomfortable; however it was better than standing. When she did sit, she could only tolerate minimal pressure on her back, as leaning against a backrest aggravated her condition. Position of relief was lying supine on the floor; however she reported the pain was still very intense in this position. She had difficulty leaving the house due to the pain. A systems review revealed that she was also experiencing extreme bilateral breast pain that was worse with nursing.
Prior to and during pregnancy
There was no history of LBP or PGP prior to or during her pregnancy. Prior to pregnancy she was very active; yoga 3 days a week, brisk walks 4 days a week, bike riding 2 days a week and canoeing once a week. This was her first pregnancy and she (and her baby) was experiencing a healthy singleton pregnancy. At delivery, they discovered that she had hydroamnios, but this did not affect the birth or the baby.
Labour
The patient delivered at 41 weeks after 2 days of early labour and 20 hours of active labour. She anticipated a fully natural, un-medicated birth; assisted by a midwife and doula at a community hospital. The patient coped as well as can be expected during labour; she utilized positional changes, a birthing ball and other support methods to manage her labour. The baby, who was big, was head down and posteriorly positioned so it was surprising thatthe patient did not experience any back pain or back labour. The labouring process was long and, after 20 hours of active labour, she was 9 cm dilated. There was uncertainty as to whether or not the patient would fully dilate so, at the midwife's suggestion, the patient elected to have an epidural to achieve the last centimeter of dilation. Failing that, the other option (and least desirable to the patient) was to have a caesarean section (C-section).
According to the hospital records, there were no issues associated with the administration of the epidural. The patient was asked to remain still, in a flexed position during the injection.The anaesthesiologist found the injection site easily and proceeded with one puncture. The patient gave birth to a healthy baby boy, weighing 9 lbs 10 oz and measuring 54.5 cm in length. According to the neonatal records, the baby's Apgar score and glucose levels were within normal levels. No other interventions were required during the delivery.
Immediately following birth
Three days following delivery the LBP began. The patient reported that her lower back felt swollen to the touch and she noticed some swelling at the level she was experiencing pain. This coincided with the injection site of the epidural. The area of complaint was not red or hot, nor was there any broken skin. As stated above, the pain was dull and achy but sharp with certain movements and rated as 9/10 on the VAS. Over the course of 2-3 days extension became restricted and the patient maintained a flexed position. The patient also reported feeling physically fatigued following the birth of her child. The patient was under the care of a midwife for 6 weeks after the birth. The midwife followed up with the patient at regular intervals for general post-partum care and newborn check-ups. The back pain was one of several concerns raised by the patient during the visits as she was a new mom who was also having extreme bilateral breast pain and difficulty nursing. The midwife assured the patient that LBP following labour is normal, as is being fatiguedso the main priority for the patient became decreasing the breast pain and feeding her new born. The midwife did recommend the patient see a chiropractor and massage therapist to help relieve her back pain.The patient had a new family doctor (who she had not yet met) lined up for when she was no longer under the care of the midwife.
LBP treatments
The patient attempted to alleviate the pain by seeking different forms of treatment.
Massage therapy: During 2 home visits at days 7 and 11 after birth, a registered massage therapist (RMT) performed light massage to the affected area, with only mild relief of symptoms and a rating of 7/10 following treatment.
Chiropractic care: Eight days following the birth, a chiropractic colleague, who has previously treated the patient, provided a house call. The patient reported to the chiropractor that her pain was between the bottom of the twelfth rib and top of the iliac crest, specifically at the site of her injection. Upon visual examination, the chiropractor told the patient that there was diffuse swelling slightly above, at the site of and slightly below the site of pain. There was no associated redness or heat. Range of motion (ROM) was examined for the lumbar spine and it was determined that there were restrictions at the site of pain with extension; rotation and lateral flexion to the right at L3-L4 and L4-L5, all are indications for performing a spinal adjustment in the area of complaint. The attending chiropractor determined she had mechanical low back pain as a result of her delivery and performed a bilateral lumbar roll at the site of pain. The patient experienced temporary mild relief of pain, 7/10 on the VAS.
Naturopathic care: With the assistance of her husband, at 18 and 20 days after the birth she visited 2 Naturopathic Doctors (ND). The first visit with the first ND, who is also an International Board Certified Lactation Consultant, was to help with the breast issue she was experiencing. While there, the patient asked the NDto take a look at her back; the ND found limited ROM in flexion and extension and hypertonicity of the muscles surrounding the epidural site. The first ND diagnosed her with back pain due to inflammation as a result of epidural injection. Treatment included acupuncture around the sacral foramina and ashi points around the epidural site. Relief following this treatment was mild (8/10) and temporary.
The patient visited a second ND two days later specifically for her back pain. ROM was still restricted and painful on extension, so she maintained a slightly forward flexed position and limited on lateral flexion bilaterally with pain on the left side. The ND performed a straight leg raise, which was within normal limits (greater than 70 degrees) and did not cause any pain. Treatment also included acupuncture with little relief (VAS score of 8/10).
Self-care: Included ice, periodic use of over the counter Tylenol and Advil, topical anti-inflammatory cream, use of a back rest, rest and hot showers. At 6-weeks postpartum she attempted light whole body stretching and core exercises for 1 hour per day, 5 days per week.
Approximately 1 month following birth: Approximately 3 to 4 weeks after giving birth, the patient reported improvement (7/10), but progress was very slow and she was still having trouble walking and standing. The patient requested that the midwife help set up a follow up with a specialist who could examine her pain. The midwife suggested she could refer the patient back to the anaesthesiologist. As the patient had heard that the medical community does not normally link epidurals with LBP, the patient elected to not pursue that option for the time being.
At the 5 to 6 week mark the patient volunteered to be a subject for a study involving ultrasound assessment of lumbar and abdominal musculature immediately following pregnancy. Although her pain had decreased to a rating of 5/10, the patient was more concerned with the lump that had started to form at the site of injections as well as her inability to extend fully. With the assistance of her husband, she was able tomakeit to the academic institution performing the studyfor the required visit. During the ultrasound portion of the study, the ultrasound technician noticed a fluid-filled mass in the region of pain. It was suggested that she see a medical doctor, as her midwifery care was complete. She went to a walk-in clinic where her new family doctor practiced and was sent for blood work and an ultrasound the same day. Her medical doctor was concerned that there was an infection or cerebrospinal fluid leak so magnetic resonance imaging (MRI) was ordered and completed within 2 days. Upon review of the MRI, the radiologist and family doctor instructed the patient to go to the hospital immediately as they were concerned about osteomyelitis with possible epidural involvement.
Upon admittance to the hospital, the patient was a febrile and hemodynamically stable. There was a significant fluctuant and tender midline mass over the lumbar spine with no erythema. Her blood work (white blood cells, erythrocyte sedimentation rate and C-reactive protein) waswithin normal limits. While in the hospital, the patient was under the care of Internal Medicine in consultation with Infectious Disease and Musculoskeletal Radiology. Upon further review of the MRI at the hospital it was noted that there was a small superficial fluid collection between L3 and L4 with no evidence of epidural involvement (Figure 1). There was a local osteal reaction but her overall presentation was not consistent with osteomyelitis, as the patient did not have a fever and the blood work was normal.
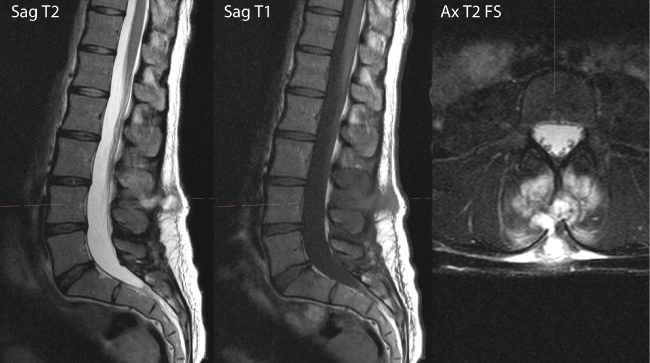
.
Figure 1: MRI 1 month postpartum. (a) T2-weighted sagittal magnetic resonance imaging (MRI) reveals a hyperintense lesion at L3-L4; (b) The same lesion, appeared hypointense in the T1-weighted MRI; (c) A fat supressed T2-weighted axial image of the same lesion.
View Figure 1
Diagnosis
In attempt to determine the composition of the mass and to remove any fluid, an ultrasound-guidedaspiration was performed. Results identified a complex inflammatory collection with a large amount of hyperemia. Only a small amount of bloody cloudy fluid could be obtained and was sent for Gram stain culture. No significant amount of fluid could be aspirated as the fluid was either very thick or was more phlegmonous. The diagnosis when the patient was discharged was a soft tissue infection, which came back positive for Staphylococcus aureus. Staphylococcus aureus is a common gram-positive coccus that typically grows in the nose and on the skin of 20-30% of healthy adults. Normally these bacteria are not harmful; however, when they penetrate the skin (as during a puncture or wound), they can lead to the formation of an abscess that ranges from mild to life threatening [21].
Treatment
The patient was treated with a course of oral antibiotics, Amoxi-clavulan. After approximately 7 days of antibiotic use the patient noticed a decrease in the size of the mass and pain intensity and, as a result, another round of antibiotics was prescribed. By the end of the second bout of antibiotics, the lump significantly reduced in size and was barely noticeable. A follow up MRI was conducted 8 weeks later to assess the resolution of the mass. It was determined at that time that there was a 2.2 × 0.8 mm phlegmon between L3-L4 spinous processes without evidence of osteomyelitis or discitis (Figure 2).
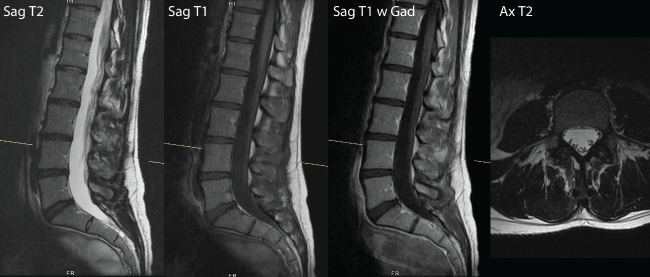
.
Figure 2: MRI 6 weeks following the start of antibiotic treatment reveals enhanced soft tissue between L3 and L4 spinous processes (at white line). (a) T2-weighted sagittal MRI; (b) T1-weighted sagittal MRI; (c) T2-weighted sagittal with contrast; (d) T2-weighted axial MRI.
View Figure 2
Post-treatment
At 12 weeks postpartum, the patient continues to have residual stiffness (2/10 on the VAS) and a decrease in ROM in lumbar extension by approximately 20%. She continues to progress with her rehabilitative exercises which target improving ROM and strengthening the transversus abdominis, multifidus, lumbar erector and gluteus minimus and medius muscles.
Concurrent health concerns
Within the first 3 days following birth, the patient experienced a fever for approximately 8 hours and developed bilateral breast pain the next morning. The midwife diagnosed the patient with mastitis and blocked ducts in the breasts; a common complaint of new mothers. Mastitisis an infection of the breast tissue that results in breast pain, swelling, warmth and redness (the latter 3 symptoms were not experienced by the patient). The ND who performed the acupuncture on the low back also treated the breast pain. Following the diagnosis of Staphylococcus aureus the ND suggested thatthe breast pain experienced by the patient may have been a stress reaction by the body. At 12 weeks postpartum the patient is no longer experiencing the breast pain.
Discussion
It is well known that back pain during the postpartum period is a common occurrence for new mothers [1-3,11-13,22,23]. Many attribute this increase in pain or, for some, new pain to the demands of motherhood, stress of labour and biomechanical and/or hormonal changes associated with pregnancy. As a result, many new moms are told to work around it, it will resolve on its own or to exercise to strengthen the back. Unfortunately, well-researched and effective treatments for postpartum back pain are scarce and women end up living with the pain. A health care practitioner should consider all of the differential diagnoses and keep them in mind when treating the new mother. This is a very rare case of atypical postpartum-related back pain involving soft tissue infection; other differential diagnoses included spinal epidural abscess (SEA) and osteomyelitis.
Osteomyelitis and SEA are very rare causes of back pain in the postpartum population. However, it is important to recognize their occurrence as they can lead to severe complications, such as bone necrosis, squamous cell carcinoma, amyloidosis, compression of the spinal cord and cauda equina syndrome [21,24,25]. Osteomyelitis is a bacterial or fungal infection of bone that may present as an acute infection, evolving over days to two weeks [21,24,26]. The most common infecting organism is Staphylococcus aureus [21,24,26]. Osteomyelitis may result due to hematogenous spread, local spread from contiguous infection or is associated with vascular insufficiency [27]. Risk factors include circulation disorders such as diabetes, open fracture or surgery, chronic soft tissue infection, immunocompromise and the use of IV drugs or catheters [21,24,26]. The patient in the current study had none of these risk factors, however during pregnancy and early postpartum period there is a modulation of the immune system reinforcing awareness of possible infection while safely allowing the fetus to grow [28]. Local signs and symptoms include acute onset of pain at the site of infection, redness, swelling, tenderness and delayed wound healing [22,25,27]. In the current case, a lump had formed but no redness was present at the site of infection.
In contrast, an epidural abscess is a contained purulent infection of the central nervous system, which can be either intracranial or spinal (extracranial) [28]. It is most commonly due to a bacterial infection, specifically Staphylococcus aureus, gram-positive bacteria that represents the major causative agent in SEA cases [25]. Although SEA is a very rare complication of epidural anesthesia [25], there are few case reports in the literature that report paraspinal abscess formation after spinal anesthesia for C-section [29-31]. When reviewing the literature, a case of mild back pain with purulent discharge from a swollen, erythematous puncture site, the patient did not present with fever [31]. In contrast, a patient who had a similar local low back presentation had severe debilitating back pain and a fever [30]. The third case had progressive back pain after birth and one episode of high fever associated with headache and malaise. It eventually progressed to local inflammation, erythema and purulent discharge [30]. All three of these cases were caused by Staphylococcus aureus infection [29-31].
SEAs tend to occur in the lumbosacral region and tend to be acute in nature [28]. However, the clinical presentation of SEA can be quite variable; the typical triad of fever, back pain and neurological deficit is not always present in patients [32-34]. Diagnosis can be delayed because the initial presentation may only be nonspecific back pain and half the cases are estimated to be misdiagnosed or have delayed diagnosis [35]. In general, the prevalence of SEA is approximately 0.2-2 cases/10,000 hospital admissions [25,36-39]. However, a systematic review by Reihaus et al. identified that 10 of 854 patients in the literature developed a SEA during pregnancy or the postpartum period and suggest it may be related to impairment of the immune defenses during the postpartum period [25]. They propose that SEA is a very rare complication of central nerve blocks; of 505,000 epidural anesthetics performed between 1982-1986, only one case of SEA was reported [25]. Skin infections were the primary source of infection [25]. Signs and symptoms include back pain, paresthesia, fever and neurological deficits [25,35,37,38]. As it progresses, signs and symptoms reflect spinal irritation, and orthopaedic testing including Lasegue's, Kernig's and Lhermitte's signs, Brudzinski reflex and neck stiffness become positive [25]. It is important to note that back pain is the most common symptom and occurs in 71% of patients [25]. Risk factors include spinal intervention, surgery, trauma, degenerative joint disease, chronic diseases that may impair immunity, hematogenous seeding of the spine, systemic infection or spread of local infection from soft-tissue infection, osteomyelitis, epidural anesthesia or nerve blocks [25,36-39]. SEA may first present with antecedent symptoms similar to discitis or osteomyelitis [36-38]. Therefore, when suspecting SEA it is important to consider these as differential diagnoses, and vice versa.
Conclusion
The patient in this case study was diagnosed with a subdermal soft tissue infection positive for Staphylococcus aureus at the site of the epidural at L3-L4. Other possible diagnoses had the condition progressed include ostemyelitis or SEA. Early diagnosis is a key factor in providing appropriate care to patients with a soft tissue infection, osteomyelitis or SEA. While in most cases epidural administration is not associated with back pain, it is important to have these diagnoses on a differential list in postpartum women presenting with acute onset of back pain.
Acknowledgements
We would like to thank the institution's radiologist, for preparing the figures for the manuscript and we would like to thank the ultrasound technician involved in the study, for suggesting the patient obtain proper imaging.
References
-
Stapleton DB, MacLennan AH, Kristiansson P (2002) The prevalence of recalled low back pain during and after pregnancy: a South Australian population survey. Aust N Z J Obstet Gynaecol 42: 482-485.
-
Turgut F, Turgut M, CetinA Yahin M (1998) A prospective study of persistent back pain after pregnancy. Eur J Obstet Gynecol Reprod Biol 80: 45-48.
-
Líndal E, Hauksson A, Arnardóttir S, Hallgrímsson JP (2000) Low back pain, smoking and employment during pregnancy and after delivery - a 3-month follow-up study. J Obstet Gynaecol 20: 263-266.
-
Ansari NN, Hasson S, Naghdi S, Keyhani S, Jalaie S (2010) Low back pain during pregnancy in Iranian women: Prevalence and risk factors. Physiother Theory Pract 26: 40-48.
-
Malmqvist S, Kjaermann I, Andersen K, Økland I, Brønnick K, et al. (2012) Prevalence of low back and pelvic pain during pregnancy in a Norwegian population. J Manipulative Physiol Ther 35: 272-278.
-
Gutke A, Ostgaard HC, Oberg B (2006) Pelvic girdle pain and lumbar pain in pregnancy: a cohort study of the consequences in terms of health and functioning. Spine 31: E149-E155.
-
Ostgaard HC, Andersson GB, Karlsson K (1991) Prevalence of back pain in pregnancy. Spine (Phila Pa 1976) 16: 549-552.
-
Bastiaanssen JM, de Bie RA, Bastiaenen CH, Essed GG, van den Brandt PA (2005) A historical perspective on pregnancy-related low back and/or pelvic girdle pain. Eur J Obstet Gynecol Reprod Biol 120: 3-14.
-
Katonis P, Kampouroglou A, Aggelopoulos A, Kakavelakis K, Lykoudis S, et al. (2011) Pregnancy-related low back pain. Hippokratia 15: 205-210.
-
Ostgaard HC, Roos-Hansson E, Zetherström G (1996) Regression of back and posterior pelvic pain after pregnancy. Spine (Phila Pa 1976) 21: 2777-2780.
-
Bastiaenen CH, de Bie RA, Vlaeyen JW, Goossens ME, Leffers P, et al. (2008) Long-term effectiveness and costs of a brief self-management intervention in women with pregnancy-related low back pain after delivery. BMC Pregnancy Childbirth 8: 19.
-
Breen TW, Ransil BJ, Groves PA, Oriol NE (1994) Factors associated with back pain after childbirth. Anesthesiology 81: 29-34.
-
Gutke A, Lundberg M, Östgaard HC, Öberg B (2011) Impact of postpartum lumbopelvic pain on disability, pain intensity, health-related quality of life, activity level, kinesiophobia, and depressive symptoms. Eur Spine J 20: 440-448.
-
Ostgaard HC, Andersson GB (1992) Postpartum low-back pain. See comment in PubMed Commons below Spine (Phila Pa 1976) 17: 53-55.
-
Mogren IM (2008) Physical activity and persistent low back pain and pelvic pain post partum. BMC Public Health 8: 417.
-
Speziali A, Tei MM, Placella G, Chillemi M, Cerulli G (2015) Postpartum Sacral Stress Fracture: An Atypical Case Report. Case Rep Orthop 2015: 704393.
-
Millwala F, Chen S, Tsaltskan V, Simon G (2015) Acupuncture and postpartum pyogenic sacroiliitis: a case report. J Med Case Rep 9: 193.
-
Dunk RA, Langhoff-Roos J (2010) Osteomyelitis of the pubic symphysis after spontaneous vaginal delivery. BMJ Case Rep 2010.
-
Terzi R, Terzi H, Özer T, Kale A (2014) A rare cause of postpartum low back pain: pregnancy- and lactation-associated osteoporosis. Biomed Res Int 2014: 287832.
-
Gunduz B, Erhan B, Saridogan M, Elbasi N, Ozdogan H (2010) Postpartum Spinal Osteoporosis: An Unusual Cause of Low Back Pain. Turk J Rheumatol 25: 47-49.
-
Calhoun JH, Manring MM (2005) Adult osteomyelitis. Infect Dis Clin North Am 19: 765-786.
-
Schytt E, Lindmark G, Waldenström U (2005) Physical symptoms after childbirth: prevalence and associations with self-rated health. BJOG 112: 210-217.
-
Russell R, Groves P, Taub N, O'Dowd J, Reynolds F (1993) Assessing long term backache after childbirth. BMJ 306: 1299-1303.
-
Berbari EF, Steckelberg JM, Osmon DR (2015) Osteomyelitis In: Bennett J, Dolin R, Blaser M, eds. Mandell, Douglas, and Bennett's Principles and Practice of Infectious Diseases. 8th ed. New York, NY: Saunders; 2015: 1318-1327.e2.
-
Reihsaus E, Waldbaur H, Seeling W (2000) Spinal epidural abscess: a meta-analysis of 915 patients. Neurosurg Rev 23: 175-204.
-
Hatzenbuehler J, Pulling TJ (2011) Diagnosis and management of osteomyelitis. Am Fam Physician 84: 1027-1033.
-
Lew DP, Waldvogel FA (2004) Osteomyelitis. Lancet 364: 369-379.
-
Mor G, Cardenas I (2010) The immune system in pregnancy: a unique complexity. Am J Reprod Immunol 63: 425-433.
-
Huang YY, Zuo Z, Yuan HB, Tsou MY, Chen MT, et al. (2005) A paraspinal abscess following spinal anaesthesia for caesarean section and patient-controlled epidural analgesia for postoperative pain. Int J Obstet Anesth 14: 252-255.
-
Chiang HL, Chia YY, Chen YS, Hung CC, Liu K, et al. (2005) Epidural abscess in an obstetric patient with patient-controlled epidural analgesia--a case report. Int J Obstet Anesth 14: 242-245.
-
Yang YW, Chen WT, Chen JY, Lee SC, Chang Y, et al. (2011) Bacterial infection in deep paraspinal muscles in a parturient following epidural analgesia. Acta Anaesthesiol Taiwan 49: 75-78.
-
Joshi SM, Hatfield RH, Martin J, Taylor W (2003) Spinal epidural abscess: a diagnostic challenge. Br J Neurosurg 17: 160-163.
-
Prendergast H, Jerrard D, O'Connell J (1997) Atypical presentations of epidural abscess in intravenous drug abusers. Am J Emerg Med 15: 158-160.
-
Davis DP, Wold RM, Patel RJ, Tran AJ, Tokhi RN, et al. (2004) The clinical presentation and impact of diagnostic delays on emergency department patients with spinal epidural abscess. J Emerg Med 26: 285-291.
-
Huff JS (2015) Spinal epidural abscess clinical presentation. Medscape.
-
Pradilla G, Ardila GP, Hsu W, Rigamonti D (2009) Epidural abscesses of the CNS. Lancet Neurol 8: 292-300.
-
Darouiche RO (2006) Spinal epidural abscess. N Engl J Med 355: 2012-2020.
-
Shah NH, Roos KL (2013) Spinal epidural abscess and paralytic mechanisms. Curr Opin Neurol 26: 314-317.
-
Grewal S, Hocking G, Wildsmith JA (2006) Epidural abscesses. Br J Anaesth 96: 292-302.





