Obstetrics and Gynaecology Cases - Reviews
Robotic Assisted Repair of Bilateral Fallopian Tube Prolapse after Vaginal Hysterectomy
Ruben J. Barrera -Vera1,2*, Kimberley Chiu1,2, Perry Cohen3, Victoria Chernyak4 and Nicole S. Nevadunsky1,2
1Division of Gynecologic Oncology, Department of Obstetrics & Gynecology and Women's Health, Albert Einstein College of Medicine, Montefiore Medical Center, Bronx, NY, USA
2Albert Einstein Cancer Center, Albert Einstein College of Medicine, Bronx, NY, USA
3Department of Pathology, Albert Einstein College of Medicine, Montefiore Medical Center, USA
4Department of Radiology, Albert Einstein College of Medicine, Montefiore Medical Center, USA
*Corresponding author: Ruben J. Barrera-Vera, Montefiore Medical Center, Albert Einstein College of Medicine, Department of Obstetrics, Gynecology and Women's Health, 3332 Rochambeau Avenue, Bronx, New York 10467, USA, Tel: 718 -920-4794, Fax: 718-920-6313, E-mail: nnevadun@montefiore.org
Obstet Gynecol Cases Rev, OGCR-3-072, (Volume 3, Issue 1), Case Report; ISSN: 2377-9004
Received: December 14, 2015 | Accepted: January 27, 2016 | Published: January 30, 2016
Citation: Barrera-Vera RJ, Chiu K, Cohen P, Chernyak V, Nevadunsky NS (2016) Robotic Assisted Repair of Bilateral Fallopian Tube Prolapse after Vaginal Hysterectomy. Obstet.
Gynecol Cases Rev 3:072. 10.23937/2377-9004/1410072
Copyright: © 2016 Barrera-Vera RJ, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Background: Fallopian tube prolapse into the vagina is a rare clinical presentation, and only approximately 100 cases have been reported in the literature to date. To our knowledge, bilateral prolapse after hysterectomy has not been described and only unilateral presentations have been reported.
Case: We report the case of a 45 year-old female with bilateral fallopian tube prolapse six years after vaginal hysterectomy, who had complaint of chronic pelvic pain beginning after the procedure. The patient was treated with opioid pain medications for severe pain for several years and had been evaluated by several providers who performed examination by direct visualization and Papanicolaou smear of the vaginal cuff but failed to identify the prolapse. After several consultations for pelvic pain she was sent to Gynecologic Oncology when the ultrasound showed cystic structures involving the adnexa bilaterally, with the differential given of dermoid versus tuboovarian complex and torsion. When evaluated in the Gynecologic Oncology clinic the clinical exam was consistent with prolapsed fallopian tube, however, preoperative biopsy of the presumed tube showed squamous epithelium. The patient was treated with robotic assisted bilateral removal of the fallopian tubes and had complete resolution of pelvic pain within six weeks recovery period.
Conclusion: Fallopian tube prolapse is a rare anatomical but surgically correctable cause for pelvic pain. Preoperative biopsy of fallopian tubes prolapsed into the vagina may be misleading; this type of histopathological change has previously been reported and should not guide treatment decision making, when the clinical suspicion for fallopian tube prolapse on visual inspection is high. Furthermore, patients should be preoperatively counseled for potentially bilateral adnexal surgery/adnexectomy, and there may be value to intraperitoneal evaluation when considering vaginal only surgical approach.
Keywords
Fallopian, Tube, Prolapse, Hysterectomy, Bilateral, Pelvic, Pain, Robotic
Introduction
Hysterectomy is the most frequent major surgical procedure performed in gynecology. Fallopian tube prolapse into the vaginal vault is a rare but known reported complication of hysterectomy, estimated to occur in approximately 0.1% of procedures, although the true incidence of this complication is difficult to estimate, as many cases are either unreported or unrecognized. It is three times more frequent after vaginal hysterectomy [1]. In 1902, Pozzi described for the first time the occurrence of fallopian tube prolapse in two patients following hysterectomy [2]. Presenting symptoms include dyspareunia, vaginal discharge, vaginal bleeding, and a vaginal mass suspicious for malignancy. Patients may present with abdominal or pelvicpain, which is usually unilateral and reproducible by grasping the prolapsed tube. Urinary symptoms, tubo-ovarian abscess, herpetic salpingitis and peritonitis due to vaginoperitoneal fistula have also been described [3,4]. Visual examination of the vaginal vault may demonstrate a red, granular, or polypoid mass that resembles proliferative vault granulation tissue [5]. Definitive diagnosis is often made with excisional biopsy, although cellular atypia induced by chronic inflammation may pose a diagnostic pitfall [6-8]. Surgical excision is the preferred treatment.
Case
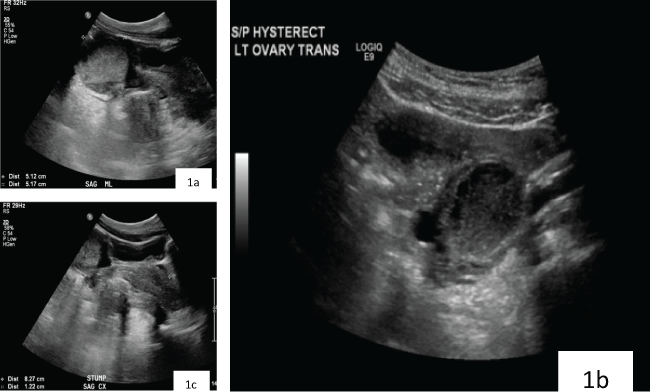
We present the case of a 45 year-old woman with a positive smoking history and family history of arthritis, ovarian cancer and lung cancer, who had vaginal hysterectomy in 2009 for a fibroid uterus. The patient recalled experiencing severe pelvic pain from the immediate postoperative period, and recounted that she walked up several flights of stairs during her recovery period. Before being referred to our facility, she had been seen by multiple providers for pelvic pain, and was prescribed opioid medications as well as atypical anti-depressants without resolution of symptoms. Two years after her hysterectomy she had an ultrasound examination that showed a 3.6 × 2.7 × 4.9 cm left ovary with fluid debris level as well as a right ovary that measured 4.0 × 2.8 × 4.0 cm and contained a 3.1 × 2.0 × 2.5 cm sonolucency. Papanicolaou smear at that time showed atypical squamous cells of uncertain significance, and anucleated and benign glandular cells present. Repeated ultrasound for persistent pelvic pain was performed in 2015 which showed a 5.2 cm mass consistent with the lesion seen previously in the left adnexa, a 5.4 × 5.0 × 5.3 cm mass with complex cystic components and rim of solid stroma in the right adnexal region, as well as 3.6 × 1.9 × 2.7 cm vaginal cuff or cervical remnant (Figure 1a, Figure 1b and Figure 1c).
.
Figure 1: a) Ultrasound of right ovary showing complex cystic structure; b) Ultrasound of left ovary showing complex cystic structure; c) Possible vagina/cervical stump remnant.
View Figure 1
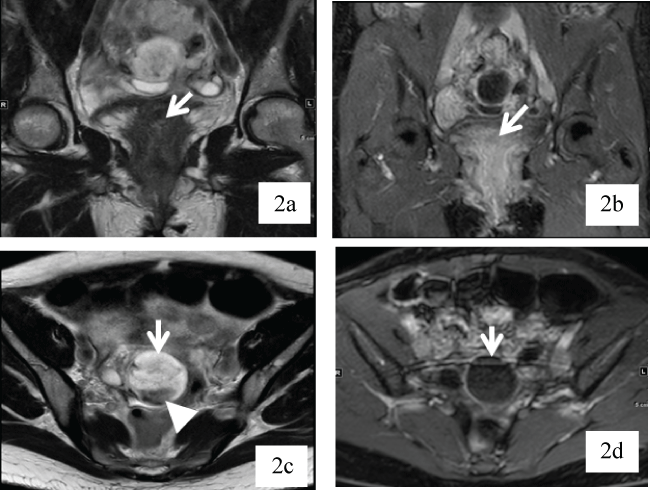
Recommendation was made for pelvic magnetic resonance imaging which showed a cluster of cysts in the right adnexa measuring approximately 4.7 × 5.7 × 4.3 cm, with the largest cyst measured 4.2 × 3.6 × 4.1 cm and containing a dependent hemorrhagic/proteinaceous component. It was impossible to determine on imaging whether the lesion represented a single multiloculated cyst or multiple cysts within the right ovary. Hydrosalpinx was noted on the left as well as several follicles within the left ovary. No abnormalities were noted at or near the vaginal cuff (Figure 2a, Figure 2b, Figure 2c and Figure 2d).
.
Figure 2: a) Coronal T2-weighted image through posterior vagina demonstrates subtle signal abnormality which in retrospect corresponds to the lesion found on the physical examination; b) Coronal post-contrast T1 weighted image through the level similar to (2a) demonstrating no discernable signal abnormality; c and d) Axial T2-weighted (c) and post-contrast T1-weighted (d) images demonstrate a T2-hyperintense non-enhancing lesion (long arrow) with dependent sediment (short arrow) consistent with hemorrhagic cyst.
View Figure 2
Her tumor CEA tumor marker was found to be elevated at 5.8 ng/mL, however the remainder of tumor markers was normal, CA19-9 of 8.9 U/mL, CA125 of 10.8 U/mL and AFP of 3.4 ng/mL, and the patient was referred for Gynecologic Oncology consultation.
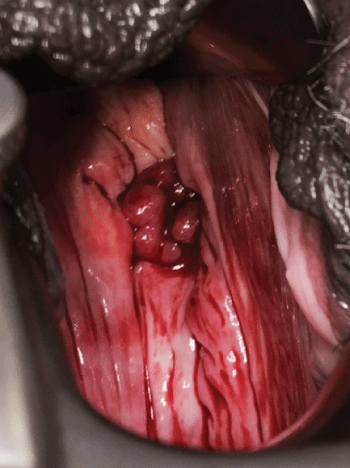
Upon presentation to Gynecologic Oncology the patient was in moderate distress and endorsed a history of intractable pelvic pain that was uncontrolled by medications. The pain caused her unremitting discomfort and depression. On an extensive symptom review, the patient reported having hadcolicky abdominal pain, located predominantly in the right lower abdominal quadrant, intermittent and not related to a particular activity or time. She denied fevers, chills or sweating, nausea or vomiting, cough or breathing complaint. During exacerbations, the pain would reflect to the contralateral side, rating it as 3/10 baseline, and 10/10 during acute attacks. Dyspareunia was reported as well. No vaginal discharge or change in urinary or bowel habits was described. Physical examination was in all ways negative except for a tender reddish polypoid mass protruding from the vaginal cuff with no associated dischargeon speculum examination. Manipulation of the tissue during physical exam reproduced the pain. To visual inspection the tissue appeared to be a 2-3 cm fallopian tube protruding into the vaginal cuff (Figure 3).
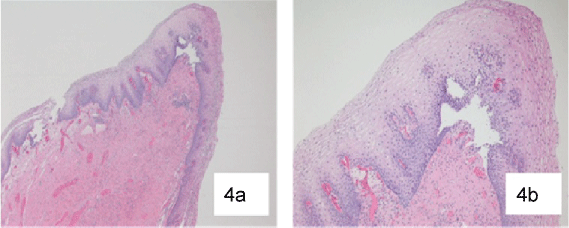
Papanicolaou smear was performed which resulted in normal findings, and biopsy of the lesion resulted squamous epithelium with no significant histopathological findings (Figure 4a and Figure 4b).
.
Figure 4: a) (40x) Vaginal mass biopsy shoeing benign unremarkable squamous mucosa; b) (100x) Vaginal mass biopsy.
View Figure 4
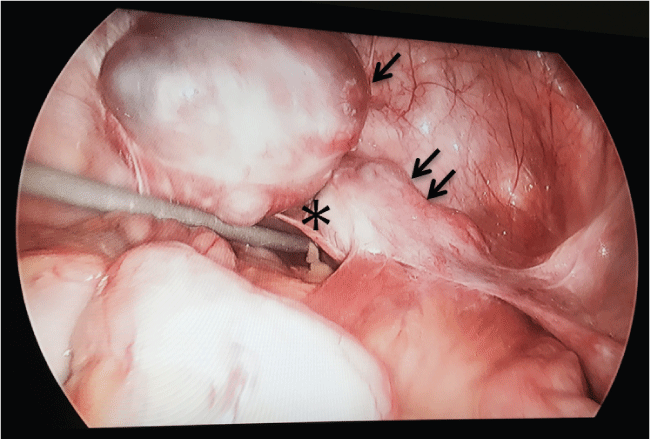
Based on clinical exam the patient was counseled and consented for robotic assisted removal of presumed prolapsed Fallopian tube. Operative findings included adnexal structures protruding towards the vaginal vault with several cystic structures noted along the right fallopian tube Figure 5.
.
Figure 5: Intraabdominal view of the prolapse. (Right tubal complex [single arrow], left tubal complex [double arrow], vaginal cuff [asterix]).
View Figure 5
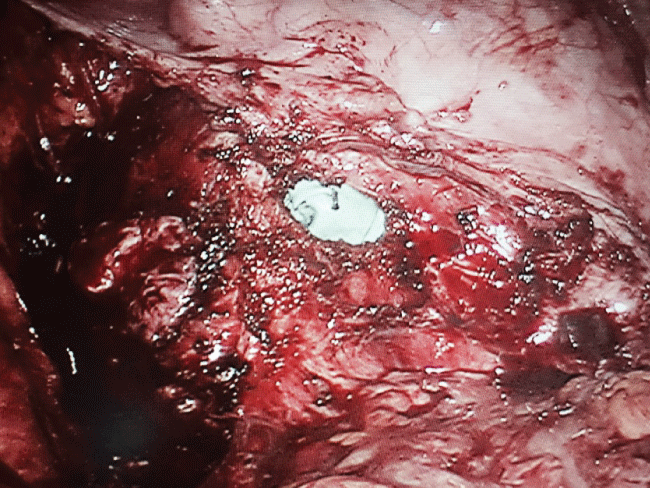
No free fluid was present and no fistula was demonstrated. The tubes were completely resected and no intra-operative complications were encountered, Figure 6. The angles of the vaginal vault were taken separately, and the rest of the vault was sutured continuously with vicryl stitches.
She was discharged uneventfully on postoperative day #1. Surgical specimen demonstrated a right hemorrhagic cyst with no evidence of malignancy and left hemorrhagic corpus luteum cysts of the ovary. Both fallopian tubes showed no histopathologic abnormality. Her immediate post-operative recovery was uneventful. One week later, she was reexamined in the outpatient clinic, and she reported improved pelvic pain, with complete resolution by the 6th week post-procedure.
Discussion
Chronic pelvic pain affects approximately 1 in 7 women. It accounts for approximately 10 percent of all ambulatory referrals to a gynecologist. Its complex natural history and sometimes unclear etiology poses a diagnostic challenge to clinicians. It is a common indication for diagnostic and therapeutic surgery, accounting for approximately 20 percent of all hysterectomies performed for benign disease and at least 40 percent of all gynecological laparoscopies performed annually in the United States. Common causes of chronic pelvic pain include chronic pelvic inflammatory disease, endometriosis, interstitial cystitis, irritable bowel syndrome, pelvic floor myalgia and other neurologic syndromes.
Fallopian tube prolapse is a rare and underreported cause of chronic pelvic pain, usually accompanied by dyspareunia, leucorrhea and/orblood loss, although some patients may remain asymptomatic. It has been reported to be more common after vaginal hysterectomy than abdominal hysterectomy, with an approximate incidence ratio of 3:1 [9,10]. Development of hematoma or abscess at the vaginal apex and incomplete closure of the vaginal vault after hysterectomy are predisposing factors for prolapsed [11]. Other surgical procedures in the abdominal cavity may cause prolapse of the female reproductive organs, including the fallopian tubes [12]. Intraoperative methods have been described to prevent fallopian tube prolapse after hysterectomy, including fixation of adnexa into the pelvic wall and complete peritonealization, meticulous closure of the peritoneum and the vagina separately, prevention of hematoma formation by carefully achievement hemostasis prior to vault closure and avoiding fixing the vault to the corneal pedicles [13]. The intended approach for surgical correction (vaginal versus abdominal), must be assessed individually based on the patient's comorbidities and preference [14,15]. Of interest, one series of 17 cases of fallopian tube prolapse after hysterectomy reported a 41% rate of spontaneous disappearance of the prolapsed tissue with no intervention in women with previously diagnosed tubal prolapsed [16]. Application of silver nitrate and cryotherapy has been recommended therapies for prolapse of a small amount of tissue only at the vaginal cuff.
Imaging diagnostic studies may pose a diagnostic pitfall, even with the use of advanced modalities such as pelvic MRI and biopsy of the prolapsed tissue. Vaginal cuff cytology can demonstrate squamous or glandular cells, often with superimposed granulation changes or reactive atypia secondary to chronic inflammation [17-19]. Histopathology remains the only means of definitive diagnosis. Resemblance of the sampled tissue to certain malignant entities has been described [20-22]. Clinicians should be advised that in the setting of a high clinical suspicion based on visual examination of the vaginal vault, attempts at confirmatory studies including biopsy may confuse treatment decision making. Additionally, although the prolapse appeared to be unilateral on physical exam, both tubes and ovaries were involved. While preoperative imaging suggested bilateral pathology in this case, clinicians may consider that a potentially unilateral appearing case on physical exam, may be involve both fallopian tubes and require bilateral adnexal surgery. This may be of particular concern to a patient who is hormonally premenopausal. The authors encourage clinicians to provide thorough preoperative counseling regarding potential adnexal surgery and adnexectomy, even in the presence of unilateral pathology. This may provide the surgeon with the ability to manipulate or remove the adnexa when warranted, reducing the need for a re-intervention later in time.
To the best of our knowledge, this is the first reported case of a fallopian tube prolapse in bilateral presentation repaired robotically. Our patient had a classic presentation of fallopian tube prolapse, complicated with severe chronic pelvic pain unresponsive to typical pain medications. The rapid resolution of pain after surgical removal of the fallopian tubes and closure of the vaginal cuff reinforced the suspected etiology of the pain. It is unclear why the patient had elevation in her CEA (5.8 mcg/L). Causes of elevation in CEA include malignancies of the breast, pancreas, thyroid and lung, as well as benign conditions including cigarette smoking, mucinous cystadenoma of ovary or appendix, cholecystitis, liver cirrhosis, diverticulitis, inflammatory bowel disease, pancreatitis and pulmonary infections. The upper limit for CEA in smokers is 5.5 mcg/L. It is possible that smoking or peritoneal irritation secondary to fallopian tube prolapse may have caused this slight elevation [23].
The patient in the above noted case refused adnexectomy at the time of her original surgery secondary to avoid onset of menopausal symptoms despite a positive family history of ovarian cancer. The Society of Gynecological Oncology (SGO) released a series of recommendations in November 2013, regarding the use of salpingectomy after hysterectomy for ovarian cancer prevention [24]. The SGO recommendation was forrisk-reducing salpingectomy for women at average risk of ovarian cancer, at the time of abdominal or pelvic surgery, hysterectomy or in lieu of tubal ligation, however, conversion to laparotomy was not included in this recommendation if minimally invasive techniques were unsuccessful to remove both fallopian tubes in their entirety. Walker et al. also recommended oophorectomy only for women at increased risk of ovarian cancer, such as those carrying BCRA 1 and/or 2 mutations [25]. It is unknown whether our patient would have accepted bilateral salpingectomy if counselled, as her original hysterectomy occurred in 2009 before the SGO recommendation was published.
References
-
Piacenza JM, Salsano F (2001) Post-hysterectomy fallopian tube prolapse. Eur J Obstet Gynecol Reprod Biol 98: 253-255.
-
Pozzi M (1902) Hernie de la trompe dans le vagin au niveau d'un cicatrice operatoire d'hysterectomie vaginale. C R Soc Obstet Gynecol Pediatr Pans 4: 255-257.
-
Lefrancq T, Orain I, Michalak S, Hourseau M, Fetissof F (1999) Herpetic salpingitis and fallopian tube prolapse. Histopathology 34: 548-550.
-
Fox R, Kassab A, Sankar A (2008) Ascending peritonitis associated with Fallopian tube prolapse following hysterectomy. J Obstet Gynaecol 28: 456.
-
Noor S, Halimi M, Faiz NR, Sadaf F, Akhtar P, et al. (2004) Fallopian tube prolapse after abdominal hysterectomy. J Ayub Med Coll Abbottabad 16: 78-79.
-
Song YS, Kang JS, Park MH (2005) Fallopian tube prolapse misdiagnosed as vault granulation tissue: a report of three cases. Pathol Res Pract 201: 819-822.
-
Varnholt H, Otis CN, Nucci MR, Johari VP (2005) Fallopian tube prolapse mimicking aggressive angiomyxoma. Int J Gynecol Pathol 24: 292-294.
-
Kucuk M (2002) Laparoscopic management of fallopian tube prolapse masquerading as adenocarcinoma of the vagina in a hysterectomized woman. BMC Womens Health 2: 2.
-
Ouldamer L, Caille A, Body G (2013) Fallopian tube prolapse after hysterectomy: a systematic review. PLoS One 8: e76543.
-
Ramin SM, Ramin KD, Hemsell DL (1999) Fallopian tube prolapse after hysterectomy. South Med J 92: 963-966.
-
Moustafa M, Elgonaid WE, Massouh H, Beynon WG (2008) Evaluation of closure versus non-closure of vaginal vault after vaginal hysterectomy. J Obstet Gynaecol 28: 791-794.
-
Rosenthal DM, Cheung VY (2005) Fallopian tube prolapse after laparoscopic resection of pelvic endometriosis. Obstet Gynecol 105: 808-809.
-
Patil A, Parulekar SV, Fernandes G (2014) PosthysterectomyFimbrial Prolapse. JPGO 1.
-
Candiani GB, Candiani M (1996) Posthysterectomy fallopian tube herniation. A report of two cases. J Reprod Med 41: 915-920.
-
Hernandez CR, Howard FM (1998) Management of tubal prolapse after hysterectomy. J Am Assoc Gynecol Laparosc 5: 59-62.
-
Fan QB, Liu ZF, Lang JH, Sun DW, Leng JH, et al. (2006) Clinical analysis of fallopian tube prolapse after hysterectomy. Zhonghua Fu Chan Ke Za Zhi 41: 449-451.
-
Bedaiwy MA, Kho MF, Patel P, Melton T (2011) Laparoscopic management of transcervical fallopian tube prolapse. JSLS 15: 393-395.
-
Nasir N, Desai M, Marshall J, Gupta N (2013) Prolapsed fallopian tube: cytological findings in a ThinPrep liquid based cytology vaginal vault sample. Diagn Cytopathol 41: 146-149.
-
Wolfendale M (1980) Exfoliative cytology in a case of prolapsed fallopian tube. Acta Cytol 24: 545-548.
-
Michal M, Rokyta Z, Mejchar B, Pelikan K, Kummel M, et al. (2000) Prolapse of the fallopian tube after hysterectomy associated with exuberant angiomyofibroblastic stroma response: a diagnostic pitfall. Virchows Arch 437: 436-439.
-
Wheelock JB, Schneider V, Goplerud DR (1985) Prolapsed fallopian tube masquerading as adenocarcinoma of the vagina in a postmenopausal woman. Gynecol Oncol 21: 369-375.
-
Anastasiades KD, Majmudar B (1983) Prolapse of fallopian tube into urinary bladder, mimicking bladder carcinoma. Arch Pathol Lab Med 107: 613-614.
-
Sajid KM, Parveen R, Durr-e-Sabih, Chaouachi K, Naeem A, et al. (2007) Carcinoembryonic antigen (CEA) levels in hookah smokers, cigarette smokers and non-smokers. J Pak Med Assoc 57: 595-599.
-
(2013) SGO Clinical Practice Statement: Salpingectomy for Ovarian Cancer Prevention." SGO Clinical Practice Guidelines. Society of Gynecologic Oncology.
-
Walker JL, Powell CB, Chen LM, Carter J, Bae Jump VL, et al. (2015) Society of Gynecologic Oncology recommendations for the prevention of ovarian cancer. Cancer.