Obstetrics and Gynaecology Cases - Reviews
Ovarian Stromal Hyperthecosis and Hyperandrogenemia Refractory to Hormonal Suppression
Gregory M Gressel1, Natalia Buza2, Shirley M McCarthy3 and Pinar H Kodaman1*
1Department of Obstetrics, Gynecology and Reproductive Sciences, Yale University School of Medicine, New Haven, USA
2Department of Pathology, Yale University School of Medicine, New Haven, USA
3Department of Diagnostic Radiology, Yale University School of Medicine, New Haven, USA
*Corresponding author: Pinar H Kodaman, Department of Obstetrics, Gynecology and Reproductive Sciences, Yale University School of Medicine, 333 Cedar Street- FMB307. New Haven, CT 06520, USA, Tel: 203-764-5866, E-mail: pinar.kodaman@yale.edu
Obstet Gynecol Cases Rev, OGCR-2-058, (Volume 2, Issue 5), Case Report; ISSN: 2377-9004
Received: June 26, 2015 | Accepted: September 24, 2015 | Published: September 26, 2015
Citation: Gressel GM, Buza N, McCarthy SM, Kodaman PH (2015) Ovarian Stromal Hyperthecosis and Hyperandrogenemia Refractory to Hormonal Suppression. Obstet Gynecol Cases Rev 2:058. 10.23937/2377-9004/1410058
Copyright: © 2015 Gressel GM, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Background: Ovarian stromal hyperthecosis is an uncommon disorder reported only in case reports and small case series. Patients typically present with menstrual irregularity and virilization.
Case: A 67 year-old postmenopausal patient presented complaining of increased libido, hirsutism and male pattern hair loss. Laboratory investigation revealed elevated testosterone levels. Pelvic imaging including ultrasound and MRI demonstrated bilaterally enlarged ovaries. Administration of a GnRH antagonist failed to suppress androgen levels. After bilateral oophorectomy, pathology demonstrated bilateral ovarian stromal hyperthecosis. Shortly after surgery, the patient's hyperandrogenemia and symptoms resolved.
Conclusion: This is a rare report of a patient, who presented with virilization and hyperandrogenemia refractory to suppression with a GnRH antagonist due to ovarian stromal hyperthecosis. Given the overlapping concerns of potential occult ovarian cancer in patients with hyperandrogenemia and virilization, we suggest that providers consider oophorectomy rather than hormonal suppression in postmenopausal patients or in women who have completed childbearing.
Introduction
Ovarian stromal hyperthecosis (OSH) is an uncommon clinical entity. According to the 2014 WHO classification of gynecologic tumors, it has been documented in one-third of patients older than age 55 years of age in autopsy studies [1]. This disorder usually presents in the second or third decade of life with menstrual irregularity as well as signs of virilization. Presentation in the post-menopausal period is less common, but has been reported. The true incidence and etiology of this condition are unknown. Treatment involves medical therapy including hormonal suppression or surgical management, such as ovarian wedge resection or oophorectomy.
Case
A 67 year-old Caucasian woman, gravida 2 para 2 presented to our clinic for evaluation of symptoms of virilization. The patient had been menopausal since age 53 and for approximately 1 year, had noted undesired increased libido and hirsutism. Although she was not sexually active with her husband, she experienced frequent and unprovoked spontaneous orgasms. She also reported hair growth on her arms, shoulders, chin and upper lip as well as recession of her hairline and thinning of hair on her scalp. The patient's medical history was remarkable for recently diagnosed subclinical hyperthyroidism, osteoporosis, obesity, as well as Meniere's disease, hypertension and chronic obstructive pulmonary disease. She also had a long-standing history of type 2 diabetes mellitus on oral hypoglycemics with a baseline hemoglobin A1C value of 6.2%. Her obstetrical history was significant for 2 terms, uncomplicated vaginal deliveries. Her gynecologic history included late onset of menarche at the age of 16 and a history of life-long oligomenorrhea. Other than a twenty-pack year smoking history, she reported no other significant medical comorbidities.
On evaluation, the patient was noted to have normal vital signs with a body mass index of 36.6 kg/m2. She appeared hirsute with facial and lower abdominal hair growth as well as male pattern alopecia. She also exhibited cervicodorsal protuberance ("buffalo hump") and clitoromegaly, above the upper limit for her age. Laboratory assessment demonstrated elevated free (12.0 pg/mL) and total (115 ng/dL) testosterone, but normal 17-hydroxyprogesterone (39 ng/dL), dehydroepiandrosterone (57 mcg/dL), and its sulfate (36 mcg/dL). Prolactin (10.6 ng/mL), estradiol (18 pg/mL), luteinizing hormone (43.1 mIU/mL) and follicular stimulating hormone (48.5 mIU/mL) were all within normal limits for a postmenopausal woman. Thyroid stimulating hormone levels were very low (< 0.005 uU/mL), and the patient was started on methimazole for hyperthyroidism. Additional laboratory values,such as glucose tolerance testing and insulin levels, were unavailable.
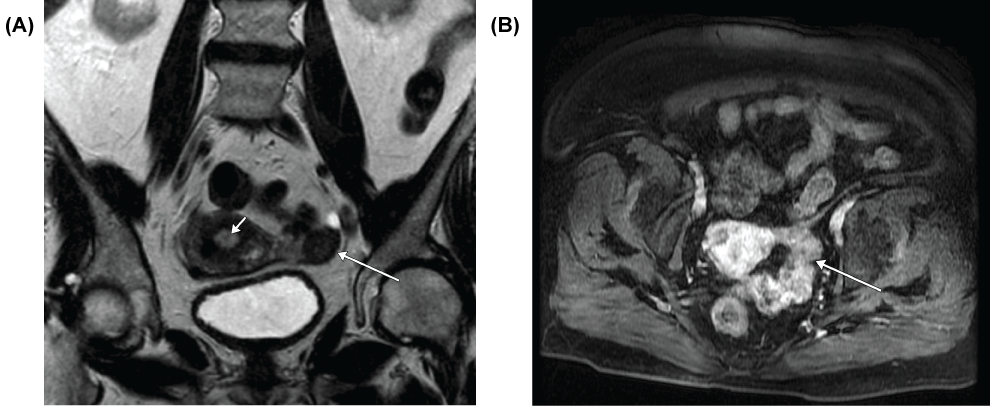
The patient's workup included a transvaginal ultrasound demonstrating a uterus measuring 3.7 × 6.4 × 4.7 cm, with right ovary measuring 1.74 × 2.27 × 2.42 cm and her left ovary measuring 1.89 × 2.26 × 2.24 cm. Given the increased ovarian volume for a postmenopausal woman (normal postmenopausal average volume of 2.6 ± 2.0 cm3) [2], a MRI was performed, which showed a thickened endometrial stripe of 9 mm with bilaterally enlarged ovaries and stromal enlargement (Figure 1). Endometrial biopsy showed atrophic endometrium with an endometrial polyp.
.
Figure 1: MRI- Coronal T2 weighted scan (A) shows a diffusely hypointense (dark), mildly enlarged left ovary (long arrow); note endometrial thickening (short arrow). Axial post IV gadolinium scan (B) demonstrates mild enhancement of the ovary (arrow).
View Figure 1
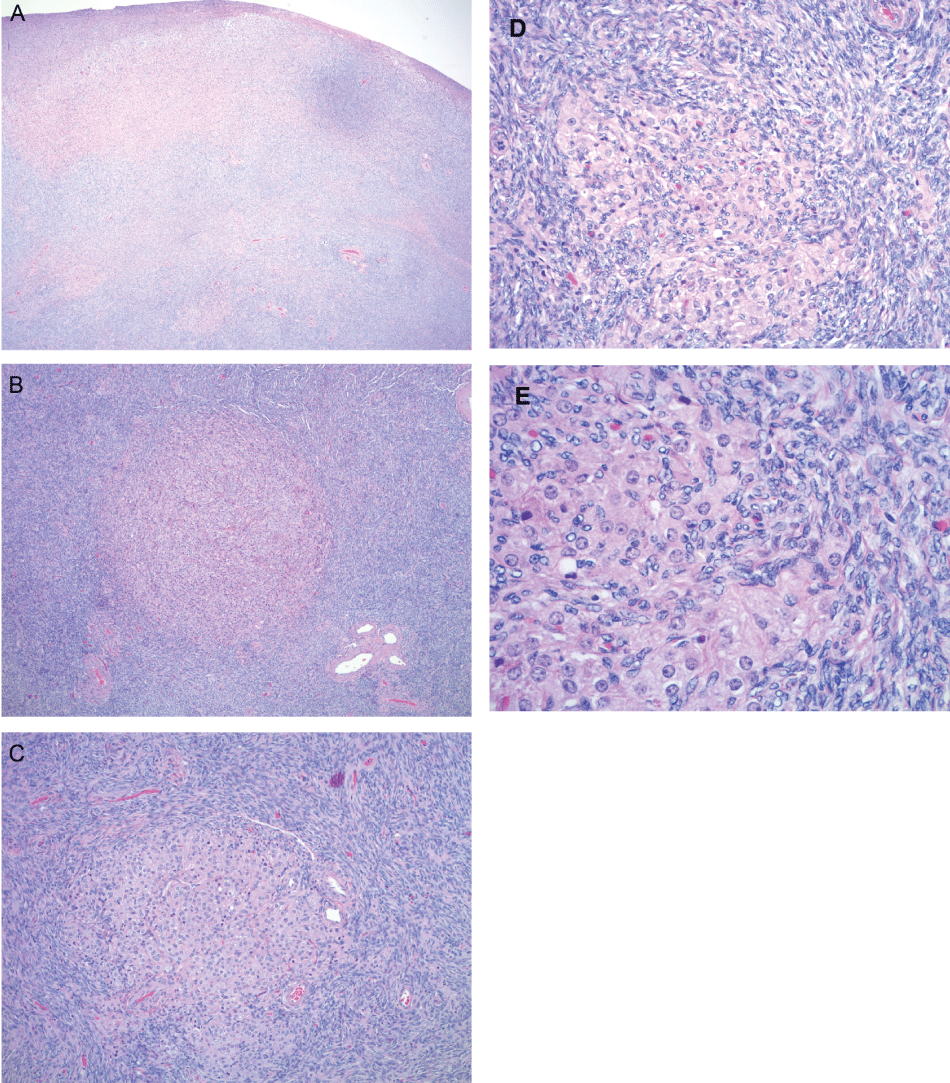
Potential treatment options were reviewed, including hormonal suppression as well as surgery. The patient received a dose of 250 mcg of the GnRH antagonist Ganirelex acetate subcutaneously, which failed to suppress her peripheral androgen levels (free testosterone 9.3 pg/mL and total testosterone 94 ng/dL one day after treatment). She underwent a laparoscopic-assisted vaginal hysterectomy with bilateral salpingo-oophorectomy. Frozen section pathology returned consistent with endometrial polyp and grossly unremarkable ovaries. Final pathology demonstrated an 88 gram uterus with multiple intrauterine leiomyomata and atrophic endometrium. The ovaries measured 3 cm in largest dimension bilaterally, and on microscopic examination showed bilateral nodular stromal hyperthecosis (Figure 2). Immediately after surgery, the patient's free (5.0 pg/mL) and total (27 ng/dL) testosterone levels fell to normal limits and at her one month follow up visit, she stated that her libido had dramatically decreased and the spontaneous orgasms had resolved. Repeat androgen levels were further decreased and in the normal postmenopausal range (free testosterone 0.8 pg/mL, total testosterone 8 ng/dL).
.
Figure 2: Pathology- Microscopic images show ovarian stromal hyperplasia (A) with small clusters and nodules of luteinized cells (B,C,D, and E). The luteinized cells in images B, C, D, and E are characterized by large eosinophilic cytoplasm and small, uniform, round nuclei. (Hematoxilin-Eosin stain, Original magnification A: 20×, B: 40×, C: 100×, D: 200×, E: 400×).
View Figure 2
Discussion
The differential diagnosis of hyperandrogenemia includes pituitary, ovarian, and adrenal sources of androgen production. Among ovarian pathologies, polycystic ovarian syndrome (PCOS) is the most common cause of hirsutism and hyperandrogenism. In contrast to PCOS, patients with OSH will usually present with signs of frank masculinization rather than uncomplicated hirsutism or dermatologic changes [3].
The first reported case of OSH was presented in 1943 by Dr. Fraenkel, who described a 32 year old female with virilization and ovarian stromal thickening [4]. OSH was not recognized as a clinical syndrome until 1949 when Culiner and Shippel described virilization in a patient with characteristic features of OSH. Their group defined the disorder as virilization resulting from bilaterally enlarged ovaries, hyperplasia and luteinization of the theca interna cells [5]. Current pathological definition of OSH includes presence of luteinized cells - singly or in small nests and nodules measuring less than 1 cm - typically associated with stromal hyperplasia [1].
Little is known about the true incidence or etiology of this disorder. According to the 2014 WHO classification of gynecologic tumors, it has been documented in one-third of patients older than 55 years of age in autopsy studies (although a reference is not cited in the report) [1]. The available literature regarding OSH consists of case reports and small case series. Patients are usually in their second or third decade of life and exhibit menstrual irregularities or oligomenorrhea. Virilization, such as baldness, facial hair growth, voice changes and clitoral hypertrophy may occur [3]. Patients with OSH produce up to eight times as much testosterone and three times as much androstenedione as patients without ovarian pathology [6]. The degree of virilization a patient exhibits closely correlates with testosterone production; however, OSH has been reported in patients without frank signs of virilization [7]. Some women with OSH have high peripheral levels of circulating estrogen, which may be a result of peripheral aromatization of androgen rather than an intrinsic ovarian process [8]. Of note, these elevated estrogen levels can result in endometrial overgrowth, including hyperplasia, and thus, abnormal uterine bleeding in the setting of OSH. Our patient's endometrial lining was thickened despite low estradiol levels and workup with endometrial biopsy preoperatively revealed a polyp and benign pathology.
The excess androgen production seems to arise from the luteinized stromal cells of the enlarged ovary [7]. Some investigators have suggested that enzymatic defects in these cells may result in increased androgen secretion [8-10]. However, other studies have demonstrated that hyperandrogenemia results from the increased volume of stromal tissue itself rather than abnormal biochemical function of the ovary [11]. Some groups have shown that administration of a GnRH agonist can suppress circulating androgen levels, suggesting that OSH may not be autonomous, but instead may rely on gonadotropin secretion [12-14].
The sonographic features of OSH are variable but the ovaries can be normal or increased in size and typically exhibit a round shape [15]. On color Doppler, no areas of increased vascularity are seen [16]. The first MRI case report described symmetric ovarian enlargement, homogeneous T2 hypointensity, and mild enhancement post contrast [17]. One other case report described bilateral ovarian masses that showed isointensity to myometrium on both T1 and T2 weighted sequences [18].
This patient's differential diagnosis as suggested by her MRI findings includes ovarian fibromas, however, in this entity, there is partial sparing of the ovaries with visualization of the follicles and a thick rind of hypointense tissue [17,19,20]. Ovarian fibromas are also T2 hypointense and minimally enhance, but are well-defined masses and typically unilateral [21]. Her differential diagnosis also includes a potential hilus cell tumor which is often small and overlooked. However, in this case, the slice-thickness on the T2 weighted scan was 5 mm, and on the post contrast scan it was 4.8 mm. A hilus cell tumor would demonstrate a higher (brighter) signal with greater enhancement on the T2 weighted scan. Both ovaries were serially sectioned for pathology at 3 mm intervals and submitted entirely for microscopic evaluation. No hilus cell tumor was present microscopically in either ovary after careful microscopic analysis.
Here we present an unusual case of a postmenopausal woman with virilization and hyperandrogenemia secondary to OSH, which failed hormonal suppression with a GnRH antagonist, a drug which competes with natural GnRH for binding to GnRH receptors, thus suppressing ovarian hormonal function. In this patient, we used a GnRH antagonist to demonstrate gonadotropin-independent androgen production by her ovaries. It is difficult to define "normal ranges" of total serum testosterone levels in postmenopausal women. Many laboratories list normal levels up to 70-80 ng/dL; however publications have reported normal levels between 30-40 ng/dL in premenopausal women and between 20-30 ng/dL in postmenopausal women [22]. Our patient had total testosterone levels as high as 115 ng/dL, and levels did not decrease after administration of a GnRH antagonist (pre and post GnRH antagonist testosterone was 86 ng/ml and 94 ng/dL, respectively).
One potential mechanism for failure of GnRH antagonism in this clinical scenario is the patient's insulin resistance. Although her type 2 diabetes was relatively well controlled on oral hypoglycemic agents with an estimated average serum glucose value of 142.7 mg/dL, hyperinsulinemia may have acted synergistically with luteinizing hormone (LH) to stimulate her ovaries. Because we have no pre- or post-operative quantitative measurement of serum insulin levels, we cannot further evaluate this hypothesis; however, if this patient's hyperandrogenemia were due to a combined effect of LH and elevated insulin levels, one would still expect a modest decrease in testosterone with the use of GnRH antagonist, which immediately abrogates release of LH from the ovary.
Another possible explanation regarding why Ganirelix failed to suppress ovarian testosterone production is that the single dose of 250 mcg may not have been sufficient to significantly suppress ovarian testosterone production. A GnRH agonist could also have been used to assess the gonadotropin dependence of ovarian androgen production; however, in this case, given the concern for possible occult malignancy in this rapidly virilizing, symptomatic postmenopausal woman, use of the quick acting GnRH antagonist was preferred with progression to surgery once a significant effect of the antagonist therapy was not seen.
To the best of our knowledge, this is the first report of a patient with OSH who failed treatment with GnRH antagonist therapy. Although we are not able to completely elucidate the mechanism for failed ovarian response to GnRH antagonism, our demonstration of independent ovarian androgen production was concerning enough for a cancerous process to prompt surgical intervention. Given the overlapping concerns of potential occult ovarian cancer in patients with hyperandrogenemia and virilization, we suggest that providers consider oophorectomy rather than hormonal suppression in postmenopausal patients or in women who have completed childbearing.
References
-
Kurman RJ, Carcangiu ML, Herrington CS, Young RH, IARC (2014) WHO Classification of Tumors of Female Reproductive Organs. (4th edn), World Health Organization, Washington.
-
Fleischer AC, McKee MS, Gordon AN, Page DL, Kepple DM, et al. (1990) Transvaginal Sonography of Postmenopausal Ovaries with Pathologic Correlation. J Ultrasound Med 9: 637-644.
-
Bardin CW, Lipsett MB, Edgcomb JH, Marshall JR (1967) Studies of testosterone metabolism in a patient with masculinization due to stromal hyperthecosis. N Engl J Med 277: 399-402.
-
Fraenkel L (1943) Thecoma and hyperthecosis of the ovary. J Clin Endocrinol Metab 3: 557-559.
-
Culiner A, Shippel S (1949) Virilism and theca-cell hyperplasia of the ovary; a syndrome. J Obstet Gynaecol Br Emp 56: 439-445.
-
Aiman J, Edman CD, Worley RJ, Vellios F, MacDonald PC (1978) Androgen and estrogen formation in women with ovarian hyperthecosis. Obstet Gynecol 51: 1-9.
-
Sasano, H., Fukunaga M, Rojas M, Silverberg SG (1989) Hyperthecosis of the Ovary. Clinicopathologic Study of 19 Cases with Immunohistochemical Analysis of Steroidogenic Enzymes. Int J of Gynecol Pathol 8: 311-320.
-
Wilson E, Greene JW Jr (1978) Androgen and estrogen formation in women with ovarian hyperthecosis. Obstet Gynecol 51: 105-106.
-
Abraham GE, Buster JE (1976) Peripheral and Ovarian Steroids in Ovarian Hyperthecosis. Obstet Gynecol 47: 581-586.
-
Nagamani, M. Urban RJ (1999). Increased Expression of Messenger Ribonucleic Acid Encoding Cytochrome P450 Cholesterol Side-Chain Cleavage and P450 17alpha-Hydroxylase Enzymes in Ovarian Hyperthecosis. Fertility & Sterility 71: 328-333.
-
Belisle S, Lehoux JG, Benard B, Ainmelk Y (1981) Ovarian hyperthecosis: in vivo and in vitro correlations of the androgen profile. Obstet Gynecol 57: 70S-5S.
-
Pascale MM, Pugeat M, Roberts M, Rousset H, Dechaud H, et al. (1994) Androgen suppressive effect of GnRH agonist in ovarian hyperthecosis and virilizing tumours. Clin Endocrinol (Oxf) 41: 571-576.
-
Krug E, Berga SL (2002) Postmenopausal hyperthecosis: functional dysregulation of androgenesis in climacteric ovary. Obstet Gynecol 99: 893-897.
-
Rajput R, Bhansali A, Singh R (2008) Ovarian hyperthecosis and response to antiandrogens: An uncommon presentation of a common disorder. J Obstet Gynaecol 28: 249-250.
-
Brown DL, Henrichsen TL, Clayton AC, Hudson SB, Coddington CC 3rd, et al. (2009) Ovarian stromal hyperthecosis: sonographic features and histologic associations. J Ultrasound Med 28: 587-593.
-
Rousset P, Gompel A, Christin-Maitre S, Pugeat M, Hugol D, et al. (2008) Ovarian hyperthecosis on grayscale and color Doppler ultrasound. Ultrasound Obstet Gynecol 32: 694-699.
-
Rosenkrantz AB, Popiolek D, Bennett GL, Hecht EM (2009) Magnetic resonance imaging appearance of ovarian stromal hyperplasia and ovarian hyperthecosis. J Comput Assist Tomogr 33: 912-916.
-
Fujii S, Kiyokawa T, Tsukihara S, Senda T, Tahara T, et al. (2009) Magnetic resonance imaging findings of ovarian stromal hyperthecosis. Acta Radiol 50: 954-957.
-
Takeuchi M, Matsuzaki K, Sano N, Furumoto H, Nishitani H (2008) Ovarian fibromatosis: magnetic resonance imaging findings with pathologic correlation. J Comput Assist Tomogr 32: 776-777.
-
Bazot M, Salem C, Cortez A, Antoine JM, Darai E (2003) Imaging of ovarian fibromatosis. AJR Am J Roentgenol 180: 1288-1290.
-
Troiano RN, Lazzarini KM, Scoutt LM, Lange RC, Flynn SD, et al. (1997) Fibroma and fibrothecoma of the ovary: MR imaging findings. Radiology 204: 795-798.
-
Goodman NF, Cobin RH, Ginzburg SB, Katz IA, Woode DE, et al. 2011. American Association of Clinical Endocrinologists Medical Guidelines for Clinical Practice for the Diagnosis and Treatment of Menopause. Endocr Pract 17: 1-25.





