Journal of Sleep Disorders and Management
Sleep Pattern during Pregnancy and Maternal Depression: Study of Aube Cohort
Flora Bat-Pitault1,2*, Christine Deruelle2, Sophie Flori3,4, Véronique Porcher-Guinet5,6, Camille Stagnara4, Aurore Guyon5,6, Sabine Plancoulaine7,8, Joëlle Adrien9, David Da Fonseca1,2, Hugues Patural3,4 and Patricia Franco5,6
1Child and Adolescent Psychopathology Unit, Salvator Hospital, Public Assistance-Marseille Hospitals, France
2Institut de Neurosciences de la Timone, CNRS, Aix-Marseille University, France
3Pediatric and neonatal reanimation, Pôle Mère et Enfant. Hôpital Nord, France
4University Jean Monnet, Saint-Etienne, France
5Pediatric Sleep Unit, Mère-Enfant Hospital, University Lyon1, France
6Integrative Physiology of Brain Arousal System, CRNL, University Lyon1, France
7INSERM, Center for Research in Epidemiology and Population Health, France
8Univ Paris-Sud, France
9Centre de recherche de l'institut du cerveau et de la moelle épinière, faculté de médecine Pierre-et-Marie-Curie, hôpital de la Pitié-Salpêtrière, France
*Corresponding author:
Flora Bat-Pitault, Service de Pédopsychiatrie, Child and Adolescent Psychopathology Unit, Salvator Hospital, Public Assistance-Marseille Hospitals, University Aix-Marseille II, 249 Boulevard Sainte-Marguerite, 13009 Marseille, France, Tel: 33-491745844, Fax: 33-491745845, E-mail: flora.bat@ap-hm.fr
J Sleep Disord Manag,
JSDM-1-005, (Volume 1, Issue 1),
Original Article
Received: August 10, 2015: Accepted: September 24, 2015: Published: September 26, 2015
Citation: Bat-Pitault F, Deruelle C, Flori S, Porcher-Guinet V, Stagnara C, et al. (2015) Sleep Pattern during Pregnancy and Maternal Depression: Study of Aube Cohort. J Sleep Disord Manag 1:005
Copyright: © 2015 Bat-Pitault F. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Objectives:The aim of this study was to compare sleep disturbances during pregnancy in women with a lifetime diagnosis of major depressive disorder (MDD) and controls and to determine whether the sub group of postpartum major depression (PPMD) women exhibits a specific pattern of disturbances.
Study design:217 women recruited in childbirth in the maternity participated in the survey with 34 included in the MDD group. 17 among the MDD group were diagnosed PPMD. Sleep characteristics of women were assessed before and during pregnancy with self-administered questionnaires and depressive symptoms after delivery were screen with the hospital anxiety depression scale (HAD). Diagnosis of depression was performed according to DSM-IV criteria during a semi-structured interview done by phone.
Results:Women in the MDD group presented more difficulties in falling asleep (P = 0.030), nocturnal awakening (P = 0.021) and sleep deprivation (P = 0.048) during the first trimester, more pain during sleep (P = 0.025) and shorter sleep duration (P = 0.012) during the second trimester than control women. They, moreover exhibited more early awakening during the second (P = 0.037) and third (P = 0.013) trimesters with more morning sleepiness during the first (P = 0.044) and second (P = 0.034) trimesters of gestation. Among these sleep alterations several, particularly in the second trimester, seemed to be associated with PPMD and not with MDD outside postpartum.
Conclusions:This survey revealed greater sleep disturbances during pregnancy in vulnerable women to MDD compared with control women. Interestingly, several of these disturbances were selectively reported in the sub group of PPMD only. These outcomes could provide biomarkers for a better detection of high-risk of PPMD women and the development of more specific strategies to improve sleep during pregnancy in order to better prevent PPMD.
Keywords
Depression, Pregnancy, Sleep, Postpartum depression, Biomarker
Introduction
Major depressive disorder (MDD) is a frequent disease in the general population (16.2% lifetime) with a serious socio-economic burden for society [1,2]. Its prevalence is higher in women during the childbearing years [3] and particularly during the postnatal period with an estimated range from 7.5% to 33.1% [4-10] and a risk of recurrence of 25% [11]. Because of its adverse outcomes on maternal-infant interactions, attachment disorders and child development [12-16], these depressive disorders occurring during post partum (Postpartum major depression (PPMD)) represents a major mental health problem.
A lot of data about MDD show that sleep is disturbed before [17], during [18] and often after an acute episode [19]. In the MDD period, a specific sleep pattern of abnormalities has been very well-known for many years. There is a reduction in REM latency and slow-wave sleep time as well as an increase in sleep latency, wakefulness and sleep fragmentation [20]. These sleep specificities are still currently explored after mood normalization [19]. The exact link sleep abnormalities and mood disorders remains however to be specified. Some recent findings tend to indicate that perturbations of sleep, insomnia in particular, could be a risk factor of MDD [17] emergence and MDD relapse. Also patients with mood disorders could share a pattern of vulnerability to mood and sleep disturbances [21] and the genetic factors that could be responsible for this association are currently under study [21,22].
Sleep disturbances had also been consistently reported during pregnancy [23-25] such as variations in sleep duration and sleepiness, increase in nocturnal awakenings [26], snoring [27] and restless legs syndrome [25], and increase, decrease or stabilization in apnea and hyponea [23]. Hormonal modifications during gestation, the mechanical effects of an enlarged uterus, backache and fetal movement are involved in these sleep changes [23]. Several studies have also reported sleep deprivation during pregnancy and its negative maternal and fetal outcomes [28]. These disturbances in sleep characteristics progressively increase during pregnancy [23-25].
However, if sleep seems to be disturbed in both pregnancy and MDD, few data are available on the sleep pattern of MDD during pregnancy and the postpartum period [29].
Karacan et al. [30] and Coble et al. [26] reported sleep recording abnormalities in depressed pregnant women and especially decreased REM sleep. In 2007, Field et al. [31] described greater sleep alterations during pregnancy in depressed women by visual analogue scales. Goyal et al. [32], in 124 primiparas, presented an association between higher depressive symptoms and difficulty in falling asleep, more daytime sleepiness and early awakening. In 2011, Okun et al. [33], using a questionnaire, found in depressed pregnant women more fragmented sleep with longer sleep latencies, longer periods of nocturnal wakefulness and poorer sleep efficiency during the second half of pregnancy, and sleep deprivation at 20 weeks of gestation. The sleep pattern of pregnant women with a vulnerability of mood disorder (i.e. a history of MDD or PPMD) has also not been clearly described because only a few studies have explored this topic. A 1994 study by Coble et al. [26] comparing the EEG sleep of women with and without a history of depression, reported an increased time in bed and sleep time in early pregnancy, increased wakefulness in late pregnancy and more sleep disturbances in women with a history of MDD. In 2007, Jomeen et al. [34] found that high scores at the Pittsburg Sleep Quality Inventory (PSQI) in late pregnancy were associated with an increase of depressive symptoms in the postpartum period. However, Okun et al. [35] did not confirm this result in women at risk of depression. In 2003, Wolfson et al. [36] reported with sleep diaries in thirty-eight first-time mothers, more total sleep time, later rising times and more time napping at the end of pregnancy in women with higher depressive symptoms in postpartum. Some polysomnography data have, in high-risk women, shown different sleep abnormalities linked to postpartum depression and in particular, modifications in REM sleep [26,37].
Taken together these previous studies showed that the links are strength between sleep alterations during pregnancy and depressive symptoms and vulnerability. Many questions persist, however, about the links between sleep disturbances and mood during the pre and post-natal periods: Are sleep abnormalities during pregnancy associated with depressive symptoms in postpartum? Do women with a vulnerability to MDD or PPMD have a specific sleep pattern during pregnancy? And, could we characterize among vulnerable women to MDD sleep abnormalities during pregnancy associated with an increased risk of postpartum depression?
In this study, we hypothesized that high-risk depressed women (called hereafter MDD group) have greater sleep disturbances in pregnancy than non-depressed women and that sleep abnormalities during pregnancy could represent a specific marker for postpartum depression.
Materials and Methods
Participants
From September 2009 to September 2011, we recruited in the AuBE cohort (Autonomic Baby Evaluation) 302 women whose infants were born in the maternity ward of Saint-Etienne (France) [38]. This cohort was constituted to study the maturation of autonomic nervous system and sleep in children between 0 and 24 months and the relationship between maternal mood and sleep during pregnancy and postpartum. We included all consenting mothers in the study whose children met the inclusion criteria and had no exclusion criteria. Inclusion criteria at birth were: term or preterm infants with a postnatal corrected age of 37 weeks and signed parental consent. Exclusion criteria were: preterm infants with a postnatal corrected age inferior to 37 weeks, familial dysautonomy, Riley-Day syndrome, Allgrove syndrome, congenital central hypoventilation syndrome, congenital heart malformation, congenital brain stem abnormalities, Pierre Robin syndrome, disorder of cardiac rhythm, cardiac and pulmonary medication and all medications with an action on the autonomic nervous system, general analgesia in the two weeks before the inclusion. At 24 months of follow-up, 85 mothers had decided to stop the study and only 217 mothers remained in the cohort and constituted the final sample of the current study.
Procedures
Diagnosis of maternal depression: All the mothers included in this study filled in the HAD (Hospital Anxiety Depression scale) [39] to assess their depression and anxiety symptoms at 0,6,12,18 and 24 months after delivery [40]. The HAD is a self-administered questionnaire with 14 items designed to screen the anxiety and depression by eliminating somatic symptoms may distort evaluations. They also filled in a questionnaire about their history of MDD. The mothers with a HAD depression score > 8 at any time or with a positive personal history of MDD were contacted by phone for a semi-structured interview using the Mini Instrumental Mental Interview (MINI) [41] to confirm the diagnosis of MDD (in the past or after the postpartum period) or the diagnosis of PPMD according to DSM-IV criteria. We used a broad definition of PPMD limited to the 12 months after delivery. Mothers were separated into two groups: MDD and control women. Within the MDD group, two subgroups were constituted and separately analyzed: the MDOPP (women with major depression outside of the post-partum period) and PPMD (women with major depression within the post-partum period). A mean HAD depression score and a mean HAD anxiety score were also assessed to determine the level of each mother's depression and anxiety symptoms during the postpartum period (between birth and 12 months).
Evaluation of mothers' sleep during pregnancy: On inclusion, mothers completed a form about their sleep before and in the first, second and third trimesters of pregnancy. This form had questions on total sleep time, nightmares, snoring, the need for naps, sleepiness and sleep disorders such as obstructive sleep apnea syndrome, restless legs syndrome or insomnia and their management. We collected medical and demographic data about the mother health, pregnancy and conditions of childbirth that could be linked to sleep alterations during pregnancy. These data included weight before and at the end of pregnancy, height, tobacco and coffee consumption, marital and socioeconomic status, means of her baby delivery and the term at birth.
Statistical analysis
Statistical analysis was performed using SPSS 16.0. Student's t-test was done to compare the two groups (MDD and control; and MDOPP and PPMD). A χ2 test or Fisher's exact test was used if the number of subjects was fewer than five. An analysis of correlation with Pearson's coefficient was used to examine the links between depressive symptoms and sleep duration. A multivariate analysis determined for each sleep disturbance during pregnancy if it was linked to maternal postpartum depression or to MDD in another period or to both. Logistic regressions were conducted to calculate odds ratios (ORs) with 95% confidence intervals (ICs) to illustrate the strength and significance of associations.
Results
Demographic and clinical features of mothers
Of the 217 mothers participating in the study, 45 (20.7%) smoked, 0 (0%) consumed alcohol, 119 (55%) drank coffee, 188 (86.6%) took one or more medications during pregnancy and 5 (2.3%) had antidepressant medication. They were 30.9 (± 4.5) years old, had 2 (± 1.3) gestations, a body mass index (BMI) before pregnancy of 23.1 (± 3.2) kg/m2 and a weight gain during pregnancy of 12.2 (± 4.9) kg. On delivery, 48 (22.1%) had cesareans and 65 (29.9%) had premature rupture of membranes. Of the infants, there were 121 (55.8%) males, 8 (3.7%) twins and 11 (5%) preterm with gestational age of 34.3 (± 3.7) weeks. For the cohort, the weight at birth was of 3142.8 (± 562.9) g and the Apgar score at 5 min of 9.8 (± 0.9).
Maternal MDD
The self-administering questionnaires screened 50 mothers with a positive history of MDD or with HAD depressive symptoms > 8 at one point of evaluation. The semi-structured interview using the MINI confirmed 34 mothers (15.7%) with a diagnosis of MDD (MDD group) composed of 17 mothers (7.8%) with a diagnosis of major depression outside of the postpartum period (MDOPP) and 17 mothers (7.8%) with a postpartum depression occurring during the study (PPMD). Among the PPMD mothers, 6 had one or more past MDD. The 183 mothers (84.3%) remaining women constituted the control group.
The mean HAD depression score was significantly higher in the MDD than in the control group (t = -7.12, P < 0.001) with a maximum score for the PPMD group of 7.0 ± 2.3 compared to the MDOPP group (4.3 ± 1.9), P < 0.001. The mean HAD anxiety score was also significantly higher in the MDD than in the control group (P < 0.001) but did not differ between the PPMD and MDOPP (P > 0.05). The only significant differences in clinical or demographic data between the MDD and control groups concerned the coffee consumption that was greater in MDD than in controls (P = 0.019) and the greater number of employed women in the control than in the MDD group (P = 0.020) (Table 1).
![]()
Table 1: Demographic and clinical characteristics of the sample by groups of depression.
View Table 1
Sleep pattern during pregnancy
Of the 217 pregnant women, 60 (27.7%) had nightmares, 85 (39.2%) had pain during sleep and 63 (29.0%) snored with a significant increase of symptoms from the first to the third trimester of pregnancy. We observed that 166 (76.5%) needed naps, 150 (69.1%) had daytime sleepiness and 101 (46.5%) had morning sleepiness with a significant reduction in symptoms during the second trimester and a maximum in the last trimester. 2.3% had answered to present clinical signs of obstructive sleep apnea syndrome and 31.3% of restless legs syndrome.
Nine percent complained from insomnia, 46.2% from difficulties in falling asleep, 74.2% nocturnal awakening and 42.4% from early morning awakening.
More interesting, our results showed a significant increase in the frequency of these sleep disorders during pregnancy with a maximum of symptoms during the third trimester (Figure 1). The mean sleep duration per night before pregnancy was 7.7 (± 1.1) hours with an increase in the first trimester and a decrease over time: 8.0 (± 1.4) hours in the first trimester, 7.7 (± 1.4) hours in the second trimester and 7.2 (± 2.0) hours in the third trimester of pregnancy (Figure 2). Eleven percent of the mothers were sleep deprived (less than 7 hours per night) in the first trimester, 20.6% in the second trimester and 40.5% in the third trimester of gestation. We found no association between sleep deprivation throughout pregnancy and preterm birth, occurrence of preeclampsia, gestational diabetes or cesarean sections.
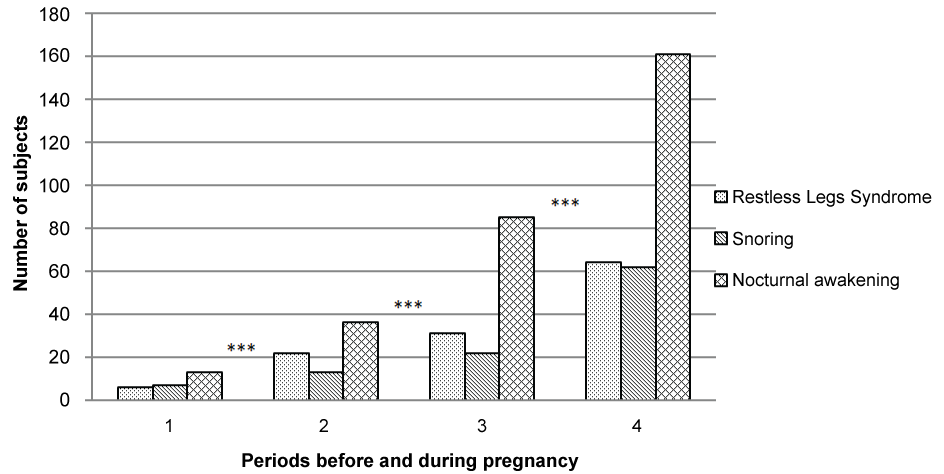
.
Figure 1: Evolution of Restless Legs Syndrome, Snoring and Nocturnal awakening during pregnancy.
1: Before pregnancy, 2: First trimester of pregnancy, 3: Second trimester of pregnancy,
4: Third trimester of pregnancy. *** P < 0.001.
View Figure 1
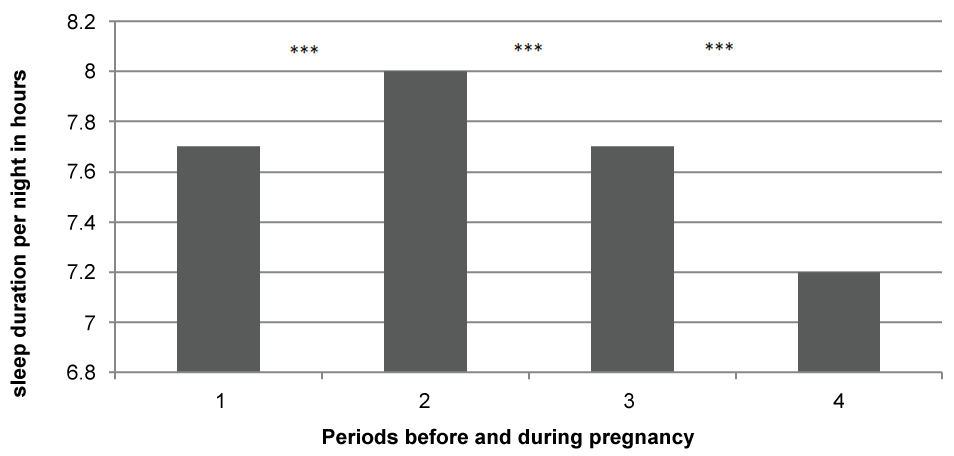
.
Figure 2: Evolution of women's sleep duration per night before and during pregnancy.
1: Before pregnancy, 2: First trimester of pregnancy, 3: Second trimester of pregnancy,
4: Third trimester of pregnancy. *** P ≤ 0.001.
View Figure 2
Sleep pattern during pregnancy and postpartum depressive symptoms in women
We used the mean HAD depression score to determine the links between postpartum depressive symptoms and sleep particularities during pregnancy for our entire cohort. We found significantly more depressive symptoms in the mothers with nocturnal awakening in the first (t = -2.042; P = 0.048) and second trimesters (t = -3.352; P = 0.001), early morning awakening in the second (t = -2.355; P = 0.027) and the third trimesters (t = -2.168; P = 0.032) and pain during sleep (t = -2.975; P = 0.003) in the second trimester of pregnancy. A significant increase in postpartum depressive symptoms is also linked to snoring (t = -2.206; P = 0.029) and apnea (t = -2.428; P = 0.016) in the third trimester of gestation, daytime sleepiness in the second (t = -3.747; P = 0.000) and the third trimesters (t = -1.968; P = 0.050) and morning sleepiness in the first (t = -2.329; P = 0.021), second (t = -3.855; P = 0.000) and third trimesters of pregnancy (t = -2.474; P = 0.014). Shorter sleep duration in the second (r = -0.255; P = 0.001) and third trimesters (r = -0.152; P = 0.049), and sleep deprivation (less than 7 hours per night) during pregnancy are also associated with higher mean HAD depression scores (Figure 3).
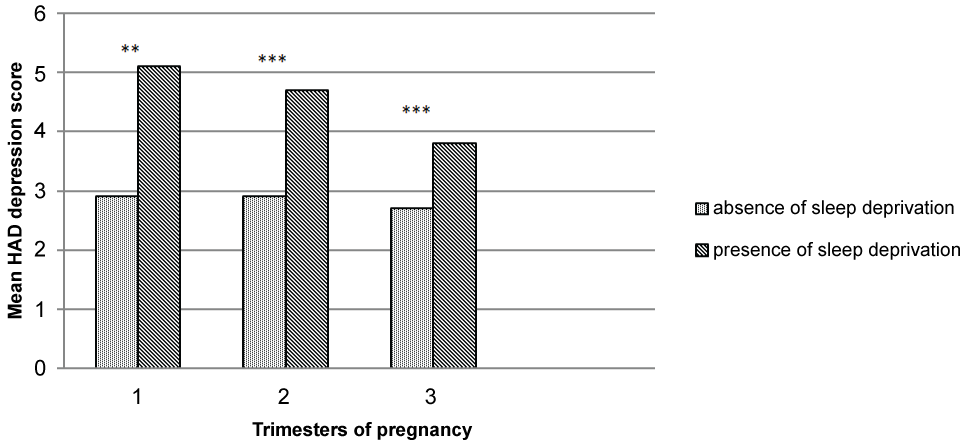
.
Figure 3: Postpartum depressive symptoms and sleep deprivation during pregnancy.
1: First trimester of pregnancy, 2: Second trimester of pregnancy, 3: Third trimester of pregnancy.
** P < 0.01; *** P ≤ 0.001
View Figure 3
Sleep pattern during pregnancy and maternal MDD
We first compared the sleep patterns of pregnant women with past or postpartum episodes of MDD and controls (Table 2). MDD mothers had a shorter sleep duration during the second trimester of pregnancy (t = 2.54; P = 0.012) with sleep deprivation (fewer than 7 hours per night) in the first trimester (P = 0.048), more nocturnal awakening in the first trimester (χ2 = 5.35; P = 0.021), more early morning awakening in the second (P = 0.038) and third trimesters (χ2 = 6.24; P = 0.013), more pain during the sleep in the second trimester (P = 0.025) and more morning sleepiness in the first (χ2 = 4.06; P = 0.044) and second trimesters of pregnancy (P = 0.034). They also had more nightmares (χ2 = 4.19; P = 0.041) and more difficulties in falling asleep (χ2 = 4.73; P = 0.030) than control women. Moreover, multivariate analysis revealed an increased probability to develop PPMD if the women had nocturnal awakening in the first trimester (P = 0.036), early morning awakening (P = 0.027), pain during sleep (P = 0.016) and morning sleepiness in the second trimester (P = 0.031). Interestingly, reported sleep disturbances were not significantly associated with MDD outside postpartum but were significantly associated with PPMD (Table 3).
![]()
Table 2: Women's sleep disturbances during pregnancy by group of depression.
View Table 2
![]()
Table 3: Impact of MDD outside pregnancy and PPMD in the "MDD group" sleep disturbances.
View Table 3
Discussion
This study compared sleep patterns of pregnant women with or without an history of depression by using, for the first time to our knowledge, depression diagnosis according to DSM-IV criteria both and explore specific sleep anomalies linked to PPMD.
First of all, the sleep alterations and their aggravation during pregnancy observed in our cohort are in accordance with previous findings in the literature [23-25]. Restless legs syndrome, nocturnal awakenings and snoring were multiplied respectively 4, 11 and 8-fold between the period prior to pregnancy and the third trimester [23]. As in Hedman et al. [23], our findings showed that sleep duration began to decrease significantly from the second trimester of pregnancy and not later. They also revealed that apnea did not constitute a problem before and throughout the pregnancy probably because of the low percentage of obese women in our cohort. Finally, as Guilleminault et al. [42] reported in 2000, daytime sleepiness increased during the first trimester but contrary to these authors, it decreased during the second trimester in our study.
Secondly, some sleep alterations that women have during pregnancy can be associated with postpartum depressive symptoms. Like Chang et al. [28], we observed a relationship between a reduction in the duration of sleep throughout the pregnancy and an increase in postpartum depressive symptoms. Likewise, more postpartum depressive symptoms were noted in women who presented an increase in apnea during the third trimester, an increase in nocturnal and early morning awakening as well as morning and daytime sleepiness throughout the pregnancy. These latest results are in agreement with those of Goyal et al. [32] in 124 primiparas followed up during the last month of pregnancy until three months postpartum but are in contradiction with those of Wolfson et al. [36] regarding the duration of sleep and early awakening. Finally, maternal snoring during the third trimester was confirmed as being associated with an increase in postpartum depressive symptoms, but contrary to O'Brien et al. [27], we did not find that it was associated with an increase in diagnosed depression.
More than a mere increase in depressive symptoms, certain sleep anomalies in pregnant women are significantly associated with lifelong diagnoses of depression according to DSM-IV criteria. This is the case for reduction of the duration of sleep and nocturnal awakening during the first trimester of pregnancy and for reduction of the duration of sleep during the second trimester. These results appear to suggest that a decrease in sleep duration at the beginning of pregnancy, a moment when the need for sleep increases, could underscore a vulnerability to depression or a sequela of a past depressive episode. In the same manner, the elevated frequency of nightmares and difficulties sleeping during pregnancy in the "depressed" group could be explained as the effects of a prior depressive episode [19] or as the prodomal state of postpartum depression (PPMD) [43] or an association of both. The pattern of sleep anomalies during pregnancy that we describe as being associated with a diagnosis of lifelong depression differs from that found by Coble et al. [26] in 14 women with a personal history of depression [26]. However, like this author, we are reporting sleep alterations that occurred very early in the pregnancies of these women at risk and in particular for the duration of sleep. Moreover, it should be noted that nocturnal awakening during the first trimester of pregnancy and early awakening, pain during sleep and morning sleepiness during the second trimester are linked to an increase in the probability of developing postpartum depression (PPMD) and not to the fact of having only one major depressive episode outside postpartum (MDOPP). These results suggest that the impact of a personal history of depression on the pregnant women's sleep is lower than a future episode of postpartum depression (PPMD).
The pattern of sleep that we have just described could therefore represent a prodromal state of PPMD or a marker for vulnerability to PPMD. Finally, the strong association that exists between PPMD and bipolarity could lead to the hypothesis that these sleep abnormalities during pregnancy could be interpreted more broadly as a risk factor for bipolarity in association with the circadian dysregulation that is described in this pathology [44,45]. Knowledge of these abnormal sleep patterns should make it possible to better define strategies to improve sleep during pregnancy (bright light exposure, behavioral therapy for insomnia, manipulation of the sleep/wake rhythm, interpersonal and social rhythm therapy) in order to prevent PPMD [46,47].
The results of this study should be considered in the light of some limitations.
First of all, this study only used subjective sleep data within the limits of self-reporting, notably regarding sleep duration. Several authors have already reported that pregnant women sleep approximately 30 minutes less than they declare [28].
Second, the retrospective collection of maternal sleep data during pregnancy may involve recall biases. This is however unlikely since sleep changes abnormalities reported in the current study are perfectly concordant with those described in previous studies conducted on similar populations [19,23-25,28,32].
Third, the women in our cohort had potentially few medical complications during pregnancy because our exclusion criteria were broad in terms of infant pathology and prematurity. It is therefore logical to hypothesize that these pregnant women could have better sleep than the general population. In this context, it is reassuring to observe for the generalization of our results, that the sleep disturbances during pregnancy that we have described are in agreement those described in other studies [23-25].
Fourth, among the 217 women in our cohort, only 34 had a diagnosis of MDD according to DSM-IV criteria and only 17 of them developed PPMD. This low percentage of depression in our cohort is probably in part due to the selection bias induced by the long duration of the study (2 years). We could therefore hypothesize that given their symptoms, more depressed women refused to participate in the study or left before the end. While it is true that the low number of depressed women decreased the power of our results, the fact that the results related to the depressive symptoms of our entire cohort are in agreement with those of depressed women is in favor of their reliability. Of course, future studies with larger cohorts will be needed to confirm our results.
Fifth, some women who developed PPMD during follow-up had personal histories of depression before pregnancy. In that case, it is impossible to totally eliminate the impact of past depression on sleep pattern during pregnancy that is specifically associated with PPMD. However, the absence of an association between sleep anomalies during pregnancy and a major depressive episode outside of the postpartum period (MDOPP) supports the hypothesis of a PPMD-specific sleep pattern during pregnancy.
In conclusion, this study of 217 participants made it possible to describe the sleep patterns of women during pregnancy and confirmed certain anomalies found in previous studies. This is the first study that has attempted to characterize both sleep alterations in pregnant women with diagnosis of a lifelong major depressive episode and a sleep pattern that constitute a prodromal state or a vulnerability marker of PPMD. Our results could have a certain number of implications in terms of PPMD prevention in pregnant women and should enable the development of more specific strategies that aim to improve sleep to diminish PPMD. These results should be confirmed by longitudinal studies of large cohorts of women at great risk of MDD in order to better differentiate the sleep pattern of pregnant women presenting a history of depression from that of women who only have PPMD during the follow-up and who could therefore present a prodromal state or a risk factor for PPMD and perhaps even bipolarity.
References
-
Kessler RC, Berglund P, Demler O, Jin R, Koretz D, et al. (2003) The epidemiology of major depressive disorder: results from the National Comorbidity Survey Replication (NCS-R). JAMA 289: 3095-3105.
-
Bat-Pitault F, Da Fonseca D, Cortese S, Le Strat Y, Kocher L, et al. (2013) The sleep macroarchitecture of children at risk for depression recruited in sleep centers. Eur Psychiatry 28: 168-173.
-
Hasin DS, Goodwin RD, Stinson FS, Grant BF (2005) Epidemiology of major depressive disorder: results from the National Epidemiologic Survey on Alcoholism and Related Conditions. Arch Gen Psychiatry 62: 1097-1106.
-
Milgrom J, Gemmill AW, Bilszta JL, Hayes B, Barnett B, et al. (2008) Antenatal risk factors for postnatal depression: a large prospective study. J Affect Disord 108: 147-157.
-
Le Strat Y, Dubertret C, Le Foll B (2011) Prevalence and correlates of major depressive episode in pregnant and postpartum women in the United States. J Affect Disord 135: 128-138.
-
Gavin NI, Gaynes BN, Lohr KN, Meltzer-Brody S, Gartlehner G, et al. (2005) Perinatal depression: a systematic review of prevalence and incidence. Obstet Gynecol 106: 1071-1083.
-
S Banti, M Mauri, A Oppo, Borri C, Rambelli C, et al (2011) From the third month of pregnancy to 1 year postpartum. Prevalence, incidence, recurrence, and new onset of depression. Results from the Perinatal Depression - Research & Screening Unit study. Comprehensive Psychiatry 52: 343-351.
-
Austin MP, Hadzi-Pavlovic D, Priest SR, Reilly N, Wilhelm K, et al. (2010) Depressive and anxiety disorders in the postpartum period: how prevalent are they and can we improve their detection? Arch Womens Ment Health 13: 395-401.
-
Muneer A, Minhas FA, Tamiz-ud-Din Nizami A, Mujeeb F, Usmani AT (2009) Frequency and associated factors for postnatal depression. J Coll Physicians Surg Pak 19: 236-239.
-
A Gaillard, Y Le Strat, L Mandelbrot, Keita H, Dubertret C (2014) Predictors of postpartum depression: Prospective study of 264 women followed during pregnancy and postpartum. Psychiatry Res 215: 341-346.
-
Wisner KL, Perel JM, Peindl KS, Hanusa BH, Piontek CM, et al. (2004) Prevention of postpartum depression: a pilot randomized clinical trial. Am J Psychiatry 161: 1290-1292.
-
Logsdon MC, Wisner KL, Pinto-Foltz MD (2006) The impact of postpartum depression on mothering. J Obstet Gynecol Neonatal Nurs 35: 652-658.
-
Hayes LJ, Goodman SH, Carlson E (2013) Maternal antenatal depression and infant disorganized attachment at 12 months. Attach Hum Dev 15: 133-153.
-
Beck CT (1995) The effects of postpartum depression on maternal-infant interaction: a meta-analysis. Nurs Res 44: 298-304.
-
Grace SL, Evindar A, Stewart DE (2003) The effect of postpartum depression on child cognitive development and behavior: a review and critical analysis of the literature. Arch Womens Ment Health 6: 263-274.
-
Field T (2010) Postpartum depression effects on early interactions, parenting, and safety practices: a review. Infant Behav Dev 33: 1-6.
-
Perlis ML, Giles DE, Buysse DJ, Tu X, Kupfer DJ (1997) Self-reported sleep disturbance as a prodromal symptom in recurrent depression. J Affect Disord 42: 209-212.
-
Reynolds CF 3rd, Kupfer DJ (1987) Sleep research in affective illness: state of the art circa 1987. Sleep 10: 199-215.
-
van Mill JG, Vogelzangs N, van Someren EJ, Hoogendijk WJ, Penninx BW (2014) Sleep duration, but not insomnia, predicts the 2-year course of depressive and anxiety disorders. J Clin Psychiatry 75: 119-126.
-
Armitage R (1995) Microarchitectural findings in sleep EEG in depression: diagnostic implications. Biol Psychiatry 37: 72-84.
-
S Utge, E Kronholm, T Partonen, Soronen P, Ollila HM, et al. (2011) Shared genetic background for regulation of mood and sleep: association of GRIA3 with sleep duration in healthy Finnish women. Sleep 34: 1309-1316.
-
Etain B, Milhiet V, Bellivier F, Leboyer M (2011) Genetics of circadian rhythms and mood spectrum disorders. Eur Neuropsychopharmacol 21 4: 676-682.
-
Hedman C, Pohjasvaara T, Tolonen U, Suhonen-Malm AS, Myllyla VV (2002) Effects of pregnancy on mothers' sleep. Sleep Med 3: 37-42.
-
Mindell JA, Jacobson BJ (2000) Sleep disturbances during pregnancy. J Obstet Gynecol Neonatal Nurs 29: 590-597.
-
Pien GW, Schwab RJ (2004) Sleep disorders during pregnancy. Sleep 27: 1405-1417.
-
Coble PA, Reynolds CF 3rd, Kupfer DJ, Houck PR, Day NL, et al. (1994) Childbearing in women with and without a history of affective disorder. II. Electroencephalographic sleep. Compr Psychiatry 35: 215-224.
-
O'Brien LM, Owusu JT, Swanson LM (2013) Habitual snoring and depressive symptoms during pregnancy. BMC Pregnancy Childbirth 13: 113.
-
Chang JJ, Pien GW, Duntley SP, Macones GA (2010) Sleep deprivation during pregnancy and maternal and fetal outcomes: is there a relationship? Sleep Med Rev 14: 107-114.
-
Park EM, Meltzer-Brody S, Stickgold R (2013) Poor sleep maintenance and subjective sleep quality are associated with postpartum maternal depression symptom severity. Arch Womens Ment Health 16: 539-547.
-
Karacan I, Williams RL, Hursch CJ, McCaulley M, Heine MW (1969) Some implications of the sleep patterns of pregnancy for postpartum emotional disturbances. Br J Psychiatry 115: 929-935.
-
Field T, Diego M, Hernandez-Reif M, Figueiredo B, Schanberg S, et al. (2007) Sleep disturbances in depressed pregnant women and their newborns. Infant Behav Dev 30: 127-133.
-
Goyal D, Gay CL, Lee KA (2007) Patterns of sleep disruption and depressive symptoms in new mothers. J Perinat Neonatal Nurs 21: 123-129.
-
Okun ML, Kiewra K, Luther JF, Wisniewski SR, Wisner KL (2011) Sleep disturbances in depressed and nondepressed pregnant women. Depress Anxiety 28: 676-685.
-
Jomeen J, Martin CR (2007) Replicability and stability of the multidimensional model of the Edinburgh Postnatal Depression Scale in late pregnancy. J Psychiatr Ment Health Nurs 14: 319-324.
-
Okun ML, Hanusa BH, Hall M, Wisner KL (2009) Sleep complaints in late pregnancy and the recurrence of postpartum depression. Behav Sleep Med 7: 106-117.
-
Wolfson AR, Crowley SJ, Anwer U, Bassett JL (2003) Changes in sleep patterns and depressive symptoms in first-time mothers: last trimester to 1-year postpartum. Behav Sleep Med 1: 54-67.
-
Frank E, Kupfer DJ, Jacob M, Blumenthal SJ, Jarrett DB (1987) Pregnancy-related affective episodes among women with recurrent depression. Am J Psychiatry 144: 288-293.
-
H Patural, S Flori, V Pichot, Franco P, Pladys P, et al. (2014) Autonomic Nervous System: A Biomarker of Neurodevelopmental Comportment - the AuBE Study. J Clin Trials 4.
-
A Zigmond, R. Snaith (1983) The Hospital Anxiety and Depression Scale. Acta Psychiatr Scand 67: 361-370.
-
Bocquet C, Deruelle P (2014) What psychometric scales can be used to assess psychologic distress of pregnant women? J Gynecol Obstet Biol Reprod (Paris) 43: 587-592.
-
D Sheehan, Y Lecrubier, K Sheehan, Amorim P, Janavs J, et al. (1998) The Mini Instrumental Neuropsychiatric Interview (M.I.N.I): the development and validation of a structured diagnostic psychiatric interview for DSM-IV and ICD-10. J Clin Psychiatry 20: 22-33.
-
Guilleminault C, Querra-Salva M, Chowdhuri S, Poyares D (2000) Normal pregnancy, daytime sleeping, snoring and blood pressure. Sleep Med 1: 289-297.
-
S Nakajima, Y Inoue, T Sasai, Okajima I, Komada Y, et al. (2014) Impact of frequency of nightmares comorbid with insomnia on depression in Japanese rural community residents: a cross-sectional study. Sleep Med 15: 371-374.
-
Milhiet V, Boudebesse C, Bellivier F, Drouot X, Henry C, et al. (2014) Circadian abnormalities as markers of susceptibility in bipolar disorders. Front Biosci (Schol Ed) 6: 120-137.
-
Azorin JM, Angst J, Gamma A, Bowden CL, Perugi G, et al. (2012) Identifying features of bipolarity in patients with first-episode postpartum depression: findings from the international BRIDGE study. J Affect Disord 136: 710-715.
-
H Khazaie, M Ghadami, D Knight, Emamian F, Tahmasian M (2013) Insomnia treatment in the third trimester of pregnancy reduces postpartum depression symptoms: a randomized clinical trial. Psychiatry Res 210: 901-905.
-
Kempler L, Sharpe L, Bartlett D (2012) Sleep education during pregnancy for new mothers. BMC Pregnancy Childbirth 12: 155.





