Journal of Musculoskeletal Disorders and Treatment
The Chondral Tissue and PRP. Theory to Support the Use
Marcus Vinicius Danieli*
Department of Knee Surgery, UNIORT.E Hospital, Brazil
*Corresponding author:
Marcus Vinicius Danieli, MD, PHD, Department of Knee Surgery, UNIORT.E Hospital, Av Higienópolis 2600, Guanabara, Londrina, Paraná, Brazil, Tel: 86050-482, E-mail: mvdanieli@hotmail.com
J Musculoskelet Disord Treat, JMDT-2-020, (Volume 2, Issue 3), Short Review
Received: June 07, 2016; Accepted: September 09, 2016; Published: September 12, 2016
Citation: Danieli MV (2016) The Chondral Tissue and PRP. Theory to Support the Use. J Musculoskelet Disord Treat 2:020.
Copyright: © 2016 Danieli MV. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
The hyaline cartilage structure is very complex, with few cells, and without blood and lymphatic vessels or nerves. This makes the healing potential very limited. Knee cartilage injuries are very common, and its treatment is a major challenge. Surgical options available nowadays like chondroplasty, microfractures, mosaicplasty and autologous chondrocyte transplantation still doesn't have satisfactory results, mainly in long term. Platelet-Rich Plasma (PRP) has been used in orthopedics since the 90's in order to stimulate tissue healing, because of its potential to concentrate platelet derived growth factors in the target place. The goal of the PRP application is to stimulate a better healing environment. PRP has been used in cartilage to treat osteoarthritis and to support treatment techniques for chondral injuries. However, the literature is still doubtful regarding the surgical results with PRP application.
Chondral Tissue Structure
The hyaline cartilage covers the joint's surfaces and has the ability to support intense and repeated compression and tensile forces. This complex tissue has the function of providing a flat surface with low friction, shock absorption, and reduce the peak pressure of the subchondral bone. Consists of chondrocytes and the extracellular matrix [1,2].
The matrix is composed primarily of type II collagen and glycosaminoglycans, as hyaluronic acid and chondroitin sulfate. It has important functions like: protect the chondrocytes from mechanical loads; stock of cytokines and growth factors; to determine type, concentration and diffusion rate of nutrients; and act as cell signal transducer [2]. The matrix deformation produces mechanical, electrical and chemical stimuli affecting chondrocytes function [2].
Articular cartilage has no blood vessels, lymphatics or nerves. Chondrocytes nutrition and metabolites transport are made by diffusion through the extracellular matrix and depend on the synovial fluid [1,2]. These are spheroidal cells without cell to cell contact and are only 1-5% of the total cartilage volume [2]. They are responsible for the synthesis of type II collagen, aggregative proteoglycans, non-collagenous proteins, structuring and maintenance of specialized matrix and synthesis of matrix degradation enzymes. This cell has a highly individual metabolic activity; however, due to the low number of cells, the total activity is reduced. Their survival is subject to low oxygen concentration, therefore it depends on the anaerobic metabolism; in addition, the mechanical loads also influences the cell function [2].
Collagen constitutes 10 to 20% of hyaline cartilage, and type II composes 90-95% of the total. It provides an important tensile strength [2].
Proteoglycans are protein molecules of polysaccharides which are 10 to 20% of cartilage tissue and provide a compressive strength. The proteoglycan subunits are called glycosaminoglycans [2].
The cartilage is composed of four main layers: the superficial layer, thinner, with flat ellipsoids cells, parallel to the articular surface and covered with a thin synovial fluid layer (called lamina splendens). Chondrocytes of this zone synthesize a high concentration of collagen and few proteoglycans, which makes the high water concentration of this zone. The parallel collagen fibrils provide greater tensile strength. This zone also has filter function for macromolecules, thus, protects the cartilage from the synovial immune system [2]. This layer preservation is critical to protect the underlying zones [3]. Below the superficial layer there is the transitional zone (or intermediate), characterized by low cell density, with spheroidal cells, abundant extracellular matrix and high concentration of agrecans, with obliquely oriented collagen fibers, resisting primarily to compressive forces [2,3]. Next comes the radial zone, formed by perpendicular cells with spheroidal shape. This layer contains a large diameter collagen fibers and a great concentration of proteoglycans [2]. And lastly, the calcified zone, which shows a small volume of cells imbibed in calcified matrix, with low metabolic activity. It is noted that this layer has type X collagen synthesis, responsible by the structure and shock absorption of the subchondral bone [2].
The Chondral Healing
A typical tissue response to injury follows the classic sequence of necrosis, inflammation, repair and scar remodeling. The vascular phase is the most important part of the healing, and cartilage, being avascular, has its healing capacity hampered [2]. However, the necessary forces to cause an acute chondral injury must be larger than those required to cause a femoral fracture [4]. An osteochondral lesion (which affects the cartilage until the subchondral bone) causes bleeding, with fibrin clot formation and activation of the inflammatory response. Then, occurs growth factors release that stimulates vascular invasion, undifferentiated cells migration and synthetic activity of cells. After two weeks, some mesenchymal cells take the form of chondrocytes, and begin to synthesize matrix with type II collagen and high concentration of proteoglycans. These cells produce hyaline-like cartilage zones. Six to eight weeks after injury, the repair tissue contains many similar to chondrocytes cells, with new matrix, but the tissue is rarely equal to the original cartilage [4,5]. This tissue, even when similar to hyaline cartilage, differs biochemical and biomechanically from normal [5], because it is mainly made of type I collagen, which is more resistant to tensile than compressive forces [6]. When the injury doesn't reach the subchondral bone, the bleeding is limited, and the scar tissue, when present, will be of poorer quality in most cases [2].
Literature suggests that the chondral and osteochondral repair will be complete in six weeks or less, but the remodeling continues for months or even years. Therefore, the minimum time for a post-treatment analysis is about four to six months, and methods showing promising initial results need a new evaluation after six months [4,5].
In most cases, the repair tissue begins to lose proteoglycan matrix over the time, with fragmentation and fibrillation, increasing the collagen content and decrease of cells concentration, after one year or less [4]. The remaining cells take the form of fibroblasts and the matrix became a dense collagen fibrils package. This tissue fragments and degrades. Its evolution is worse according to the age of patient and the size of the lesion [4].
The incidence of chondral injuries is very high, especially in the knee. In a study of 993 consecutive knee arthroscopies, Aroen, et al. [7] found cartilage changes in 66% of patients, with 11% of total chondral injuries (113 patients), more severe in medial femoral condyle (43%) and patella (23%). Shelbourne, et al. [8], in a study of 2770 ACL reconstructions, found 125 chondral injuries, classified as Outerbridge 3 or 4, and these patients had lower subjective scores in the postoperative period than those who had no chondral injuries.
The main goal of the surgical treatment of chondral injuries is to promote the repair of this tissue, to eliminate pain and to restore joint function, preventing or, perhaps, slowing the progression to osteoarthritis [9].
The PRP
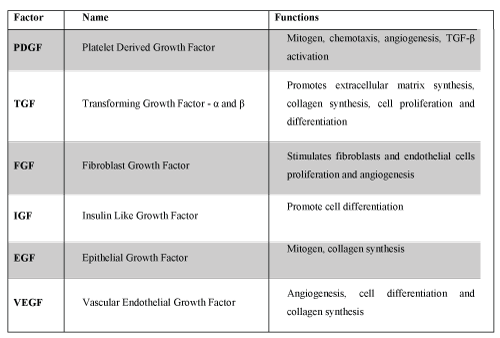
To increase the healing capacity, growth factors can be placed in order to enhance the inflammatory phase and the matrix proliferation; a scaffold can be introduced to serve as a structure for cell migration and concentration of active cytokines; or stem cells could be delivered at the site [10]. Based on this principle, since the 90's, began in orthopedics the use of Platelet-Rich Plasma - PRP. With a better understanding of tissues healing it was observed that many natural components present in blood, like fibrin, vitronectin, platelet-derived growth factor and TGF-β, when increased in their concentration, could change and accelerate the healing process [11]. These components stimulate cell proliferation, chemotaxis, cell differentiation, debris removing, angiogenesis, synthesis and deposition of extracellular matrix. Platelets have over than 30 bioactive proteins in their alpha granules, many with crucial role in homeostasis and wound healing. Examples of these proteins (Figure 1): Platelet Derived Growth Factor (PDGF), Transforming Growth Factor-beta (TGF-β), Platelet Factor-4, Interleukin-1, Platelet Derived Angiogenic Factor, Vascular Endothelial Growth Factor (VEGF), Platelet Derived Endothelial Growth Factor, Epithelial Growth Factor, Insulin Like Growth Factor (IGF), Osteocalcin, Osteonectin, Fibrinogen, Vitronectin, Fibronectin and Thrombospondin-1 [11].
The PDGF is a potent mitogen for connective tissue cells. TGF-β has morphogenic action and is also involved in collagen synthesis. IGF-1 appears to be critical for survival, growth and cell metabolism, and the cooperative action of the VEGF induces proliferation and endothelial cell migration, starting the angiogenic response [12].
PRP in defined as the plasma portion with higher concentration of platelets [13]. Increasing the number of these cells will increase the complement fractions, secreted proteins concentration and also the growth factors presents in the platelets alpha granules. Thus, the PRP shows capacity for recruitment and proliferation of mesenchymal stem cells and endothelial cells [11].
Some articles mentioned that PRP should have 2 to 2.5 times higher concentration of platelets when compared to normal plasma [14], others state that the dosage must be 3 to 5 times higher [15]. However, there are several ways of presenting and obtaining (homemade or commercial) with different concentrations and nomenclatures [7,8,16,17]. Furthermore, the concentration of active cytokines may vary from each person, independent of platelet concentration [15].
PRP is obtained in a simple way, with low cost and with virtually no risk to the patient. The technique consists of collecting a blood sample, anticoagulated in vitro with citrate-that inhibits the ionized calcium and prevents the coagulation cascade formation. After that the blood is centrifuged, once or twice, according to the protocol [15,18]. The first centrifugation separates the red and white blood cells from the Platelet-Rich Plasma. The second centrifugation separates the Platelet-Rich Plasma, called, in this case, Platelet Concentrate (PC) and the Platelet-Poor Plasma (PPP) (Figure 2) [18]. Prior to application, PRP may be activated to release the alpha granules content. This activation can be done by thrombin (bovine or human), CaCl2 (calcium chloride), synthetic collagen or contact with the collagen of the tissue where it will be applied. After activation, 70% of the growth factors will be released within the first ten minutes and the remaining within one hour. However small amounts will still be produced and released during the life of the platelet (for 8 to 10 days) [15,18].

.
Figure 2: PRP preparation (single and double centrifugation).
A: Collecting the blood sample; B: Result of the first centrifugation = Platelet-Rich Plasma (first layer), Buffy Coat (thin white layer) and Red Blood Cells; C: Enlarged image for a better view of the layers; D: Result of the second centrifugation = the upper half is the Platelet-Poor Plasma and the bottom is the Platelet Concentrate (Source: personal files).
View Figure 2
The most widely activation used is CaCl2, because of its low cost, higher availability and virtually no side effects, although doubts about its activation potential still remains. Thrombin would be the best activator, but has the limitation of stimulate animmune response and the transmission risk of prion diseases. Collagen is an attractive alternative because of their intrinsic involvement in the coagulation cascade and the use as a biomaterial, but it has a higher cost [19]. Activation by tissue collagen contact is suggested as a safe way, with low cost and more physiological [20,21].
PRP and the Literature
There are evidences suggesting that PRP could have some effect when applied in ACL reconstruction, tendon injuries and tendinopathy treatment, like epicondylitis, in addition to already show strong evidence in the treatment of knee osteoarthritis [10,12,22-24]. Recently, it was also observed PRP's ability to reduce pain and to provide antibacterial and antifungal effect [12]. Patients with osteoarthritis showed functional and pain improvement with PRP treatment, especially under 60 years old [18]. In this pathology there isn't a conclusion regarding the number of applications, activation type and the platelet concentration. Studies suggest that PRP application leads to a better functional scores when compared to placebo and hyaluronic acid, with this results lasting for up to 12 months, with a peak of effect about 6 months [25,26]. The use in knee arthroplasty shows less blood loss [18] and less pain [27].
In chondral tissue, PRP may increase the glycosaminoglycans concentration and type II collagen synthesis, in addition to reduce the chondral degradation [23]. Also can induce chondrogenesis of mesenchymal stem cells and promote proliferation, differentiation and adhesion of chondrocytes [23]. Additionally, the PRP clot can serve as a scaffold for chondrocytes and mesenchymal stem cells migration to the injured tissue [18]. Also shows biological glue function, enhance of the joint homeostasis and restoration of intra articular hyaluronic acid [10]. Recently, it has been shown that PRP has a strong and important action in synoviocytes [28], improving the synovial fluid quality and decreasing inflammation and pain [29] and also, improving the quality and the synthesis of the surface zone protein, with better joint lubrication [30]. These PRP effects depends on the local inflammatory response, so literature indicates not to use anti-inflammatory drugs for up to 14 days after its application [18].
It has already been shown that the type of PRP preparation should be adapted, depending on the tissue where it will be applied [31,32]. By doing this, its effect can be optimized. It is known that there are PRPs with higher leukocytes concentrations and these, containing pro-inflammatory and catabolic active cytokines such as interleukins and matrix metalloproteinases (MMPs), may not have good effect in the articular environment [13,15,31-36].
Oh, et al. [15] shows that the PDGF and VEGF concentration are directly proportional to platelet concentration, which did not occur with TGF-β and FGF. Besides that, the home made systems showed the lowest concentration of MMP-9 (catabolic protein). The PRP action shows an inverse relationship to the size of chondral injury and the patient's age [13,37].
Kon, et al. [37] also suggests that even if PRP has no effect on the chondral structure or in the degenerative joint disease progression, it may, however, influence the articular homeostasis, reduce synovial tissue hypertrophy and modulate cytokines. Therefore, leads to clinical improvement in patients with cartilage injuries, even temporarily.
Conclusion
Given these effects, it is necessary several researches to define the best use for PRP. We have to reach a consensus on the best formulation for each tissue, the better platelet concentration, activation form and the presence or absence of leukocyte. Recent literature has to be clear and detailed about the PRP preparation methods, the platelet concentration, the activation and leukocytes concentration. I believe that this product has a promising role in the tissue healing, especially as an adjuvant to surgery, and the articular cartilage is an important target.
References
-
da Costa AJF, de Oliveira CRGCM, Leopizzi N, Amatuzzi MM (2001) O uso da matriz óssea desmineralizada na reparação de lesões osteocondrais. Estudo experimental em coelhos. Acta Ortop Bras 9: 27-38.
-
Bhosale AM, Richardson JB (2008) Articular cartilage: structure, injuries and review of management. Br Med Bull 87: 77-95.
-
Alford JW, Cole BJ (2005) Cartilage Restoration, Part 1. Basic Science, Historical Perspective, Patient Evaluation, and Treatment Options. Am J Sports Med 33: 295-306.
-
Buckwalter JA (2002) Articular Cartilage Injuries. Clin Orthop Relat Res 21-37.
-
de Souza TD, Del Carlo RJ, Viloria MIV (2000) Avalição histológica do processo de reparação da superfície articular de coelhos. Cienc Rural 30: 439-444.
-
Saw KY, Hussin P, Loke SC, Azam M, Chen HC, et al. (2009) Articular Cartilage Regeneration With Autologous Marrow Aspirate and Hyaluronic Acid: An Experimental Study in a Goat Model. Arthroscopy 25: 1391-1400.
-
Aroen A, Loken S, Heir S, Alvik E, Ekeland A, et al. (2004) Articular Cartilage Lesions in 993 Consecutive Knee Arthroscopies. Am J Sports Med 32: 211-215.
-
Shelbourne KD, Jari S, Gray T (2003) Outcome of Untreated Traumatic Articular Cartilage Defects of the Knee: a natural history study. J Bone Joint Surg Am 85: 8-16.
-
Guilak F, Butler DL, Goldstein SA (2001) Functional Tissue Engineering. The Role of Biomechanics in Articular Cartilage Repair. Clin Orthop Relat Res S295-S305.
-
Lopez-Vidriero E, Goulding KA, Simon DA, Sanchez M, Johnson DH (2010) The Use of Platelet-Rich Plasma in Arthroscopy and Sports Medicine: Optimizing the Healing Environment. Arthroscopy 26: 269-278.
-
Mehta S, Watson JT (2008) Platelet Rich Concentrate: Basic Science and Current Clinical Applications. J Orthop Trauma 22: 432-438.
-
Sánchez M, Anitua E, Orive G, Mujika I, Andia I (2009) Platelet-Rich Therapies in the Treatment of Orthopaedic Sports Injuries. Sports Med 39: 345-354.
-
Mascarenhas R, Saltzman BM, Fortier LA, Cole BJ (2015) Role of Platelet-Rich Plasma in Articular Cartilage Injury and Disease. J Knee Surg 28: 3-10.
-
Hart R, Safi A, Komzák M, Jajtner P, Puskeiler M, et al. (2013) Platelet-rich plasma in patients with tibiofemoral cartilage degeneration. Arch Orthop Trauma Surg 133: 1295-1301.
-
Oh JH, Kim W, Park KU, Roh YH (2015) Comparison of the Cellular Composition and Cytokine-Release Kinetics of Various Platelet-Rich Plasma Preparations. Am J Sports Med 43: 3062-3070.
-
Alford JW, Cole BJ (2005) Cartilage Restoration, Part 2. Techniques, Outcomes and Future Directions. Am J Sports Med 33: 443-460.
-
Haleem AM, Singergy AA, Sabry D, Atta HM, Rashed LA, et al. (2010) The Clinical Use of Human Culture-Expanded Autologous Bone Marrow Mesenchymal Stem Cells Transplanted on Platelet-Rich Fibrin Glue in the Treatment of Articular Cartilage Defects: A Pilot Study and Preliminary Results. Cartilage 1: 253-261.
-
Foster TE, Puskas BL, Mandelbaum BR, Gerhardt MB, Rodeo SA (2009) Platelet-Rich Plasma: From Basic Science to Clinical Applications. Am J Sports Med 37: 2259-2272.
-
Harrison S, Vavken P, Kevy S, Jacobson M, Zurakowski D, et al. (2011) Platelet Activation by Collagen Provides Sustained Release of Anabolic Cytokines. Am J Sports Med 39: 729-734.
-
Arnoczky SP, Shebani-Rad S (2013) The Basic Science of Platelet-Rich Plasma (PRP): What Clinicians Need to Know. Sports Med Arthrosc 21: 180-185.
-
Mishra A, Harmon K, Woodall J, Vieira A (2012) Sports Medicine Applications of Platelet Rich Plasma. Curr Pharm Biotechnol 13: 1185-1195.
-
Vavken P, Sadoghi P, Murray MM (2011) The Effect of Platelet Concentrates on Graft Maturation and Graft-Bone Interface Healing in Anterior Cruciate Ligament Reconstruction in Human Patients: A Systematic Review of Controlled Trials. Arthroscopy 27: 1573-1583.
-
Smyth NA, Murawski CD, Fortier LA, Cole BJ, Kennedy JG (2013) Platelet-Rich Plasma in the Pathologic Processes of Cartilage: Review of Basic Science Evidence. Arthroscopy 29: 1399-1409.
-
Del Torto M, Enea D, Panfoli N, Filardo G, Pace N, et al. (2015) Hamstrings anterior cruciate ligament reconstruction with and without platelet rich fibrin matrix. Knee Surg Sports Traumatol Arthrosc 23: 3614-3622.
-
Campbell KA, Saltzman BM, Mascarenhas R, Khair MM, Verma NN, et al. (2015) Does Intra-Articular Platelet-Rich Plasma Injection Provide Clinically Superior Outcomes Compared With Other Therapies in the Treatment of Knee Osteoarthritis? A Systematic Review of Overlapping Meta-analyses. Arthroscopy 31: 2213-2221.
-
Marmotti A, Rossi R, Castoldi F, Roveda E, Michielon G, et al. (2015) PRP and articular cartilage: a clinical update. Biomed Res Int 2015: 542502.
-
Guerreiro JPF, Danieli MV, Queiroz AO, Deffune E, Ferreira RR (2015) Plasma Rico em Plaquetas (PRP) aplicado na artroplastia total do joelho. Rev Bras Ortop 50: 186-194.
-
Sundman EA, Cole BJ, Karas V, Della Valle C, Tetreault MW, et al. (2014) The Anti-inflammatory and Matrix Restorative Mechanisms of Platelet-Rich Plasma in Osteoarthritis. Am J Sports Med 42: 35-41.
-
Cugat R, Cuscó X, Seijas R, Álvarez P, Steinbacher G, et al. (2015) Biologic Enhancement of Cartilage Repair: The Role of Platelet-Rich Plasma and Other Commercially Available Growth Factors. Arthroscopy 31: 777-783.
-
Sakata R, McNary SM, Miyatake K, Lee CA, Van den Bogaerde JM, et al. (2015) Stimulation of the Superficial Zone Protein and Lubrication in the Articular Cartilage by Human Platelet-Rich Plasma. Am J Sports Med 43: 1467-1473.
-
Cavallo C, Filardo G, Mariani E, Kon E, Marcacci M, et al. (2014) Comparison of Platelet-Rich Plasma Formulations for Cartilage Healing. An in Vitro Study. J Bone Joint Surg Am 96: 423-429.
-
Filardo G, Kon E, Roffi A, Di Matteo B, Merli ML, et al. (2015) Platelet-rich plasma: why intra-articular? A systematic review of preclinical studies and clinical evidence on PRP for joint degeneration. Knee Surg Sports Traumatol Arthrosc 23: 2459-2474.
-
Anitua E, Sánchez M, Aguirre JJ, Prado R, Padilla S, et al. (2014) Efficacy and Safety of Plasma Rich in Growth Factors Intra-Articular Infiltrations in the Treatment of Knee Osteoarthritis. Arthroscopy 30: 1006-1017.
-
Sundman EA, Cole BJ, Fortier LA (2011) Growth Factor and Catabolic Cytokine Concentrations Are Influenced by the Cellular Composition of Platelet-Rich Plasma. Am J Sports Med 39: 2135-2140.
-
Boswell SG, Schnabel LV, Mohammed HO, Sundman EA, Minas T, et al. (2014) Increasing Platelet Concentrations in Leukocyte-Reduced Platelet-Rich Plasma Decrease Collagen Gene Synthesis in Tendons. Am J Sports Med 42: 42-49.
-
Braun HJ, Kim HJ, Chu CR, Dragoo JL (2014) The Effect of Platelet-Rich Plasma Formulations and Blood Products on Human Synoviocytes: Implications for Intra-Articular Injury and Therapy. Am J Sports Med 42: 1204-1210.
-
Kon E, Mandelbaum B, Buda R, Filardo G, Delcogliano M, et al. (2011) Platelet-Rich Plasma Intra-Articular Injection Versus Hyaluronic Acid Viscosupplementation as Treatments for Cartilage Pathology: From Early Degeneration to Osteoarthritis. Arthroscopy 27: 1490-1501.