Journal of Family Medicine and Disease Prevention
Impact of Physics in Family Health Care: Fluorescence Spectroscopy
Buttigieg JP1*, Helmerson K2 and Coventry BJ3
1Faculty of Medicine, Nursing and Health Sciences, Monash University, Australia
2School of Physics and Astronomy, Monash University, Australia
3School of Medicine, University of Adelaide, Australia
*Corresponding author: Jason Buttigieg, Clinical Psychologist, M Psych (Clinical), M Psych (Neuro), Post Grad Cert (Forensic & Counselling), Teaching Staff, Faculty of Medicine, Nursing and Health Sciences, Monash University, Wellington Road, Clayton, Victoria, 3168, Australia, Tel: +61-3-9905-3968, Fax: +61-39905-3948, E-mail: Jason.buttigieg@monash.edu
J Fam Med Dis Prev, JFMDP-3-051, (Volume 3, Issue 1), Opinion; ISSN: 2469-5793
Received: November 01, 2016 | Accepted: January 27, 2017 | Published: January 31, 2017
Citation: Buttigieg JP, Helmerson K, Coventry BJ (2017) Impact of Physics in Family Health Care: Fluorescence Spectroscopy. J Fam Med Dis Prev 3:051. 10.23937/2469-5793/1510051
Copyright: © 2017 Buttigieg JP, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Keywords
Health care, Fluorescence spectroscopy
Introduction
New technologies in physics are developing and modes of medical care are poised to shift rapidly. Healthcare provision provides family physicians with a complex set of challenges ranging from mastering the early detection and prevention of disease to treating a diverse range of illnesses. For example, in 2016, it was predicted that more than 8.3 million people were expected to die from cancer while cancer accounts for about 13% of all deaths worldwide [1]. One technology that is greatly advancing our understanding of the origins of disease is fluorescence spectroscopy. Fluorescence spectroscopy is also known as fluorometry or spectrofluorometry, and is basically where a beam of light, usually ultraviolet light, excites electrons in molecules of a substance to emit a spectrum of light which can then be analysed. Fluorescence spectroscopy represents the recent development in non-invasive imaging techniques that enables the visualization of molecular events underlying cellular processes in live cells [2]. Although microscopic objects can be readily manipulated at the cellular level, additional physiological insight at the macromolecular level is likely to be gained by manipulation of cells in vivo, which is being pioneered.
Fluorescence spectroscopy has been shown to detect a range of diseases, like tumors, which are easily detected by identifying altered fluorescence properties with respect to the fluorescence of ordinary tissue [3]. Fluorescence spectroscopy aids family physicians by potentially providing them with access to a more accurate assessment and diagnosis of disease manifestation across several medical conditions including: Crohn's disease and ulcerative colitis; chronic infections, such as tuberculosis, and AIDS; hematologic malignancies, including Hodgkin's disease and renal cell carcinoma; or diseases like breast cancer.
Applications of Fluorescence Spectroscopy
In order to diagnose diseases like cancer at an early stage, a small number of molecules need to be carefully studied so that their role in protein aggregation (changes to protein conformation or structure that potentially cause disease) can be accurately determined. This presents a challenge for fluorescence spectroscopy, since it has several limitations. Firstly, high doses of radiation used to create images associated with fluorescence spectroscopy can cause notable thermal damage to cell samples and a process called photo bleaching [4]. Photo bleaching is a typically permanent, photochemical alteration of the fluorescence efficiency of molecules, which occurs when the sample is exposed to light- used to examine the movement of molecules inside cells and membranes that helps define disease characteristics-for prolonged periods. Thus, the inexperienced bio-technician could inadvertently damage tissue samples if they fail to protect samples from over-exposure during single molecule fluorescence spectroscopy analysis. Secondly, it is a challenge to detect a small number of molecules for analysis from background noise (i.e., any sound other than the sound being monitored).
Fortunately, these problems with fluorescence spectroscopy can be minimized. When using a very small number of molecules, the dose of radiation is minimal. Moreover, if trapped cells have good heat conductance (due to adequate water content), molecular destruction could be reduced [2]. Ensuring photo bleaching is contained to a single region or area will also serve as a control and limit fluorescence loss due to photo-damage, as compared to fluorescence loss due to the photo bleaching, of a molecule inside cells or within membranes [5]. Optical trapping using optical tweezers provides one possible method of containing photo bleaching and can also aid with the isolation of molecules from background noise.
Single Molecule Fluorescence Spectroscopy Using Optical Trapping and Optical Tweezers
Single molecule fluorescence spectroscopy provides an interface between physics, chemistry and biomedical applications for the noninvasive diagnosis of various medical conditions, with optical trapping using 'optical tweezers' enhancing fluorescence spectroscopy. A detailed description of fluorescence spectroscopy using 'optical trapping' and optical tweezers is provided by [6]. Optical trapping originated in the 1980s [4] and is a method used in physics to trap small particles, so that the mechanics of small particle interactions/processes can be studied [4]. Optical trapping utilizes optical tweezers. Optical tweezers are capable of manipulating nanometer to micron-sized dielectric particles by exerting small forces, which can act like 'tweezers', via a highly focused laser beam. The beam is typically focused by passing it through a high numerical aperture microscope objective. The narrowest point of the focused beam contains a very strong electric field gradient. This electric field gradient allows optical tweezers to trap particles, even within a living cell. Biological research has shown how optical tweezers can trap red blood cells in mice to better study live cell dynamics in animals [2]. Thus single molecule fluorescence spectroscopy using optical trapping and optical tweezers creates a very sensitive instrument that can be used to isolate molecules from background noise and reduce the consequences of photo bleaching. Such a method thereby makes it capable of the in vivo study of single molecules. Figure 1 provides an illustration of single molecule detection using optical trapping of hydrosomes-optically trappable aqueous nano droplets that allow for the encapsulation and study of single molecules [7].
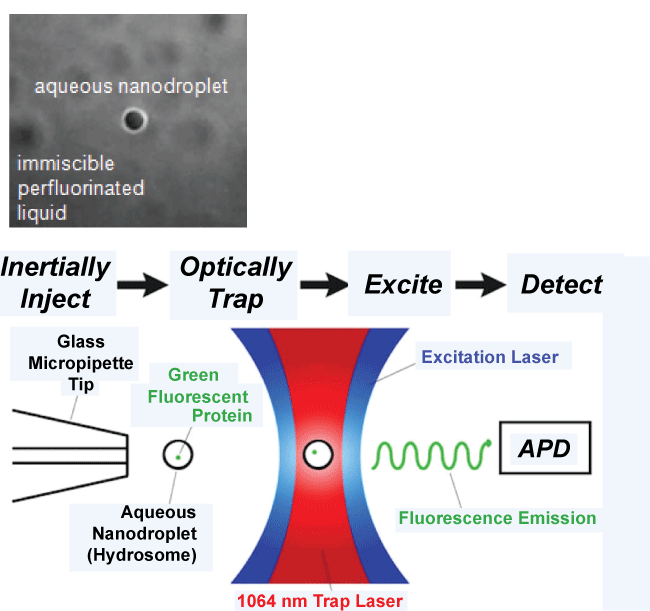
.
Figure 1: Single molecule detection of an aqueous nanodroplet (hydrosome) using optical tweezers. Avalanche photodiode (APD) is a sensitive semiconductor electronic device that converts light to electrical signal [7].
View Figure 1
Optical trapping of 'hydrosomes', microscopic water droplets encapsulating and confining single molecules, enables the effective isolation and immobilization of single molecules without lipid membranes or surface attachment, thus, providing for the study of single molecule activity. Figure 2 shows typical fluorescence signals from the single molecules [8].
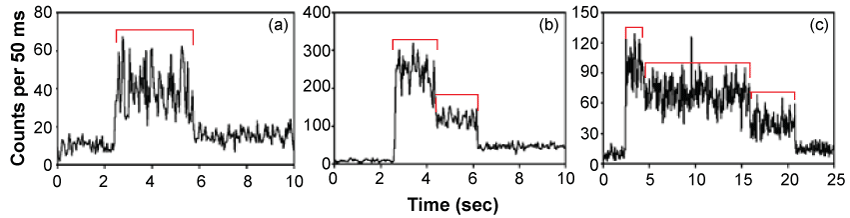
.
Figure 2: Detection of single, dual and several molecules.
Fluorescence signals over time from molecules contained in optically trapped aqueous nanodroplets. The change in signals indicates the presence of (a) one molecule, (b) two molecules and (c) three molecules [8].
View Figure 2
DNA and the proteins [9,10] and enzymes that interact with it, are commonly studied using single molecule fluorescence spectroscopy. Once a single molecule is trapped, the behavior of the molecule can be studied by the change in the emission spectra of fluorophores (fluorescence signals) due to either variations in the distance between fluorophores using Fluorescence Resonance Energy Transfer (FRET) or alterations in the orientations of the fluorophore relative to the polarized incident light [4].
Conclusion
The method of single molecule fluorescence spectroscopy offers access to the analysis of the functional dynamics of enzyme activity and therefore cell dynamics, in vivo. Moreover, as this technology improves, the analysis of smaller numbers of molecules will lower laboratory costs, thereby improving overall cost-efficacy. This analytical information is becoming more readily available to family physicians so that they can better determine the appropriate course of action in diagnosing and in selecting suitable pharmacological approaches for improved management of diseases. Fluorescence spectroscopy is one example of where careful application of such technologies arising from physics ensures family physicians will not only have access to better fundamental disease diagnostics, but will be able to likely more carefully tailor therapeutic approaches in an effort to improve clinical outcomes.
Acknowledgements
Jason Buttigieg would like to thank Monash University School of Physics and Astronomy, for helping develop Jason's concept of using physics to study situational factors like mind-body interactions at a macromolecular scale. This work helps Jason strive to map specific atomic signatures of mind-body functions. Jason also thanks Professor Brendon Coventry for mentoring Jason in the field of Medicine as well as Dr. Christopher Duffy and Rebecca Altmann for their meaningful contributions with the development of this manuscript.
References
-
World Health Organization (2016) World Health Statistics. Monitoring health for the SDGs.
-
Zhong MC, Wei XB, Zhou JH, Wang ZQ, Li YM (2013) Trapping red blood cells in living animals using optical tweezers. Nat Commun 4: 1768.
-
Dramicanin T, Dramicanin M (2016) Using Fluorescence Spectroscopy to Diagnose Breast Cancer. Chapter 12. Applications of Molecular Spectroscopy to Current Research in the Chemical and Biological Sciences.
-
Lakowicz J (2010) Principals of Fluorescence Spectroscopy. Third Edition, Springer.
-
Ishikawa-Ankerhold HC, Ankerhold R, Drummen GP (2012) Advanced fluorescence microscopy techniques--FRAP, FLIP, FLAP, FRET and FLIM. Molecules 17: 4047-4132.
-
Neuman KC, Block SM (2004) Optical trapping. Rev Sci Instrum 75: 2787-2809.
-
National Institute of Standards and Technology (NIST), USA.
-
Reiner JE, Crawford AM, Kishore RB, Goldner Lori S, Helmerson K, et al. (2006) Optically trapped aqueous droplets for single molecule studies. Applied Physics Letters 89: 1-3.
-
Jagannathan B, Marqusee S (2013) Protein folding and unfolding under force. Biopolymers 99: 860-869.
-
Lachmann HJ, Goodman HJ, Gilbertson JA, Gallimore JR, Sabin CA, et al. (2007) Natural history and outcome in systemic AA amyloidosis. N Engl J Med 356: 2361-2371.





