International Journal of Surgery Research and Practice
Computed Tomographic Quantification of Thoracic Aortic Wall Calcification as a Predictor of Clinical Outcomes in Patients Undergoing Coronary Artery Bypass Grafting
Se Jin Oh1, Kwang Nam Jin2, Jae-Sung Choi1*, Hyeon Jong Moon1, Yong Won Seong1 and Jeong Sang Lee1
1Department of Thoracic and Cardiovascular Surgery, SMG-SNU Boramae Medical Center, Seoul National University College of Medicine, Seoul, Korea
2Department of Radiology, SMG-SNU Boramae Medical Center, Seoul National University College of Medicine, Seoul, Korea
*Corresponding author: Jae-Sung Choi, MD, Ph.D, Department of Thoracic and Cardiovascular Surgery, Seoul National University College of Medicine, SMG-SNU Boramae Medical Center, 20, Boramae-ro 5-gil, Dongjak-gu, Seoul 156-707, Korea, Tel: 82 028702293, Fax: 82 02 8703866, E-mail: turejsreal@hanmail.net
Int J Surg Res Pract, IJSRP-3-041, (Volume 3, Issue 1), Research Article; ISSN: 2378-3397
Received: March 22, 2016 | Accepted: June 07, 2016 | Published: June 09, 2016
Citation: Oh SJ, Jin KN, Jae-Sung C, Moon HJ, Seong YW, et al. (2016) Computed Tomographic Quantification of Thoracic Aortic Wall Calcification as a Predictor of Clinical
Outcomes in Patients Undergoing Coronary Artery Bypass Grafting. Int J Surg Res Pract 3:041. 10.23937/2378-3397/1410041
Copyright: © 2016 Oh SJ, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Background: We evaluated computed tomographic (CT) measurement of thoracic aortic wall calcification as a predictor of the postoperative clinical outcomes in patients who underwent coronary artery bypass grafting (CABG).
Methods: Between 2009 and 2013, a total of 100 patients with isolated CABG underwent chest CT or total aorta CT angiography preoperatively. Using dedicated calcium measuring software, we measured thoracic aortic calcification score (TACS) on CT images. A high TACS was defined as a TACS above the 75th or greater percentile. Postoperative clinical outcome variables were analyzed.
Results: The mean TACS was 3892.4 ± 5580.0, and the median and 75th percentile values of TACS were 1965.0 and 5408.8, respectively. The number of patients with a high TACS was 25, andpostoperative outcome variables were detected in 43 patients. Multivariate analysis showed that preoperative use of intra-aortic balloon pump (p = 0.048), peripheral arteriopathy (p = 0.047) and a TACS above the 75th percentile (p = 0.011) were independent risk factors of postoperative mortality and cardiovascular complications.
Conclusions: Severe thoracic aortic wall calcification significantly correlated with poor clinical outcome. Therefore, CT quantification of thoracic aortic wall calcification may be a useful option for predicting early postoperative prognosis in patients undergoing isolated CABG.
Keywords
Coronary artery bypass surgery, Computed tomography, Vascular disease, Calcification
Introduction
Thoracic aortic wall calcification is associated with coronary and valvular calcification, and has been recognized as an independent predictor of increased prevalence of cardiovascular events [1,2]. Moreover, there have been several reports that have demonstrated a significant correlation between severe thoracic aortic wall calcification and cardiovascular risk factors in a large population-based cohort [3-6]. The aim of this study was to evaluate thoracic aortic wall calcification quantified by preoperative computed tomography as a predictor of clinical outcomes in patients who underwent coronary artery bypass grafting (CABG).
Materials and Methods
The study protocol was reviewed by the Institutional Review Board of our hospital and approved as a minimal risk retrospective study that did not require patient consent.
Patients
This study included a total of 100 patients (mean age, 66.2 ± 8.9 years; M:F = 61:39) who underwent isolated CABG and preoperative CT work-up between March 2009 and June 2013. We retrospectively reviewed the medical records and preoperative chest CT angiography or total aorta CT angiography. Baseline demographic data and risk factors are shown in table 1. Hypertension, Dyslipidemia, and diabetes were acknowledged when the patients were taking corresponding medicine or had been medically diagnosed elsewhere. We defined preoperative chronic kidney disease (CKD) as CKD stage > 3 (estimated glomerular filtration rateusing the Modification of Diet in the Renal Disease equation < 60 mL/min/1.73 m2).
![]()
Table 1: Baseline demographic data and risk factors.
View Table 1
Using the dedicated calcium measuring software, we measured thoracic aortic calcification score (TACS) on CT images (detection range, above the left main coronary artery to the bottom of both ventricles). A high TACS was defined as a TACS above the 75th or greater percentile.
Surgical procedure
We performed either off-pump CABG or conventional CABG as surgical revascularization strategies in the study periods. Conventional CABG was mainly performed for complete revascularization and reasonable clinical outcomes during the 3 years of learning period, but we changed our operative strategy from conventional CABG to off-pump CABG to avoid the ascending aorta manipulation and the use of cardiopulmonary bypass after 2012 according to the improved technical skill.
Off-pump CABG: All operations were performed through a median sternotomy incision under normothermia and intraoperative monitoring methods, including Swan-Ganz catheterization and transesophageal echocardiography, were routinely used. Our off-pump revascularization strategies were the use of a composite graft based on the left internal thoracic artery (ITA) and the avoidance of aortic manipulation in most patients. Immediately after the side-arm conduit (right ITA, radial artery (RA), and saphenous vein (SV)) was harvested, it was anastomosed to the side of the left ITA to construct a Y-composite graft. After the Y-composite graft was constructed, in almost all the patients the left anterior descending coronary artery territory was revascularized first by using the left ITA, and the left circumflex coronary artery territory was then revascularized, followed by the right coronary artery territory. A sequential anastomotic technique was used for complete revascularization when more than 2 coronary arterial anastomoses were needed. Patients were given an initial dose of heparin (1.5 mg/kg) and periodic supplemental doses to maintain an activated clotting time of > 300 seconds.
On-pump CABG: The conventional cardiopulmonary bypass using the ascending aorta and right atrium was performed under moderate systemic hypothermia. Transesophageal echocardiography was used to evaluate postoperative cardiac function and the presence of significant atheroma on the ascending aorta. The aorta was cross-clamped, and myocardial protection was achieved with intermittent antegrade, or combined antegrade and retrograde infusion of histidine-tryptophan-ketoglutarate solution (HTK-Custodiol; Koehler Chemi, Alsbach-Haenlien, Germany).
Postoperative clinical events
We defined postoperative clinical events as at least one occurrence among 8 clinical outcome variables; major adverse cardiac and cerebral events (MACCE) (death, perioperative myocardialinfarction (PMI), stroke, and repeated revascularization), acute kidney injury (AKI), low cardiac output requiring intra-aortic balloon pump (IABP) and/or extracorporeal membrane oxygenation (ECMO), pulmonary complication, and atrial fibrillation. Operative mortality was defined as any death within 30 days, including deaths after hospital discharge. Postoperative AKI was defined as an increase of 50% in serum creatinine level form the preoperative value or a need for renal replacement therapy regardless of serum creatinine level. PMI was defined as the combination of new ST changes or Q waves, echocardiographic demonstration of new regional wall motion abnormality, and elevation of troponin level. Pulmonary complications included postoperative pneumonia or > 48 hours of prolonged ventilator support.
Thoracoabdominal CT angiography scan protocol
CT scan was performed on a 16-channel, multi-detector CT (MDCT) (Light-Speed, GE Healthcare) or a 64-channel MDCT (Brilliance; Philips Medical Systems, Cleveland, Ohio). Scans were obtained with or without contrast media enhancement. Images were acquired in the cranio-caudal direction. CT scan with contrast enhancement was performed from the carotid bulb level to the inguinal area. For contrast-enhanced CT images, 120 to 140 mL of iopamidol (Iopamiro 300; Bracco, Milan, Italy) was administered intravenously at a rate of 3.0 mL/s. Helical scan data were acquired using 16 × 1.5 mm, or 64 × 0.625 mm collimation, with a rotation speed of 0.5 or 0.42 seconds, a pitch of 1.11 to 1.25, and 120 kilovolt (peak). Effective milliampere-seconds ranged between 120 and 187, using the automatic tube current modulation technique. Transverse datasets were reconstructed with 1.2 mm thickness at 1.2 mm increments. Resultant images were transferred to a commercially available workstation (Aquarius, TeraRecon, San Mateo, Calif) for image analysis.
Quantification of thoracic aortic calcification score (TACS) on CT images
An experienced cardiothoracic radiologist evaluated non-contrast axial CT images by using a commercially available workstation (Aquarius; TeraRecon, San Mateo, Calif). An aortic wall calcification was quantified by using the calcium scoring software system, based on a threshold of 130 Hounsfield units (HU) [7]. Automatic quantification of aortic wall calcification was initiated with the radiologist's designation for calcification on each axial CT image (Figure 1). The overall score of calcification was recorded in the workstation. We measured wall calcifications in the ascending and descending thoracic aorta from the aortic root (above the left main coronary artery) to the level of the diaphragmatic crura (the bottom of both ventricles). Calcifications in the carotid or subclavian arteries were not included. And we did not measure wall calcification in abdominal aorta due to difficult and inaccurate differentiation between the aortic wall calcification and adjacent vertebrae on the measurement software.
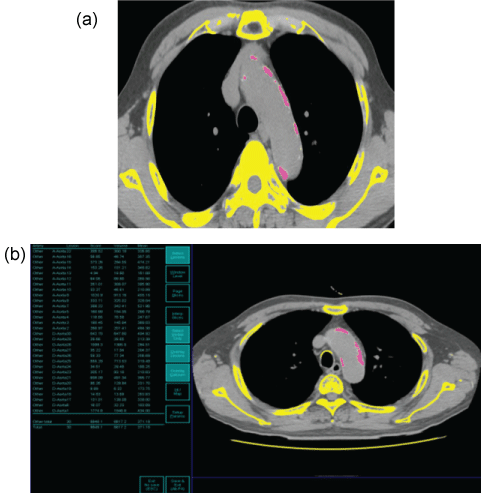
.
Figure 1: Semi-automatic quantification of aortic wall calcification.
(a) Manual selection of a calcification site from the entire thoracic aortic wall including bone;
(b) automatic measurement of aortic wall calcium score.
View Figure 1
Statistical analysis
Statistical analysis was performed with commercially available statistical software, SPSS version 20.0 (SPSS, Chicago, Illinois). Continuous variables are expressed as the mean ± standard deviation, median and ranges, or proportions. Comparisons between the 2 groups were made using the chi-square test and Fisher's exact test for categorical variables and the Student t test for continuous variables. Simple and multiple linear regression analysis were used. For multiple regression analysis, variables with a P value of less than 0.1 in simple linear regression analysis were selected. The body surface area (BSA) was used for analysis as a covariate to avoid the variation in scores according to patient height. The percentiles of TACS score were usedto determine the relative standing of an individual in our population. The 75th percentile was the third quartile. All statistical tests were 2-tailed, and a P value of less than 0.05 was considered statistically significant.
Results
Patient and operative characteristics
Preoperative BMI and left ventricular ejection fraction (EF) on echocardiography were 24.2 kg/m2 and 55.8%, respectively. Twenty-six patients had left main coronary artery disease on preoperative coronary angiography. Of all the 100 patients, 38 underwent off-pump CABG and 9 needed preoperative use of IABP. The number of distal anastomoses and different grafts used per patient were 2.9 ± 0.9 and 2.1 ± 0.4, respectively. In off-pump CABG group, left ITA was used for revascularization in all patients except one in whom the right ITA was used for revascularization of the left anterior descending coronary artery due to proximal left ITA stenosis. Other conduits used included SV (60.5%, 23/38), right ITA (31.6%, 12/38), and RA (21.1%, 8/38). In on-pump CABG group, left ITA was used in 96.8% (60/62), SV in 67.7% (42/62), RA in 20.9% (13/62), and right ITA in 14.5% (9/62). Mean cardiopulmonary bypass time in on-pump CABG group was 170.2 ± 62.3 min.
Analysis of TACS
The mean TACS was 3892.4 ± 5580.0, and the median and 75th percentiles of TACS were 1965.0 and 5408.8, respectively. The number of patients with a high TACS was 25. There was no significant difference in risk factors between the high and low TACS groups except age (p = 0.001) and hypertension (p = 0.018). Most of patients in the high TACS group had a hypertension. Moreover, high TACS group showed higher logistic EuroSCORE (p = 0.013) (Table 2).
![]()
Table 2: Comparison of risk factors between the high and low TACS groups.
View Table 2
Postoperative clinical outcomes: MACCE and other morbidities
The MACCE were detected in 14 patients. Among them, two operative motalities occurred resulting from sudden cardiac arrest with PMI and intractable ventricular fibrillation after surgery. Moreover, there were 3 strokes and 7 PMIs. Repeated revascularization including bypass graft revision or percutaneous coronary intervention was needed in 4 patients. Postoperative clinical events were detected in 43 patients. Most of the complications were atrial fibrillation (n = 31) and pulmonary complications (n = 8). Seven patients showed postoperative low cardiac output requiring intra-aortic balloon pump (IABP) and/or extracorporeal membrane oxygenation (ECMO). AKI developed in 6 patients.
Predictors of postoperative clinical events
Univariate analysis showed that age, preoperative use of IABP, peripheral arteriopathy, and a high TACS were significant predictors of clinical events (Table 3). Multivariate analysis revealed that the preoperative use of IABP (odds ratio, 3.6, p = 0.048), peripheral arteriopathy (odds ratio, 3.6, p = 0.047) and a high TACS (odds ratio, 3.3, p = 0.011) were independent risk factors forclinical events, meaningpostoperative mortality and cardiovascular complications (Table 4).
![]()
Table 3: Comparison of risk factors between the high and low TACS groups.
View Table 3
![]()
Table 4: Predictors of postoperative clinical outcomes (multivariate).
View Table 4
Discussion
Thoracic aortic wall calcification reflecting the atherosclerotic process on arterial wall has been shown to be a risk factor for atherosclerosis and to increase the risk of cardiovascular and cerebrovascular events [3-6,8]. Moreover, descending thoracic aortic calcification (DTAC) has been found to be a strong predictor of coronary artery calcification in the multi-ethnic study of atherosclerosis (MESA) [1]. A previous study of stable angina pectoris patients has demonstrated that there is a significant correlation between thoracic aortic calcification and the presence of coronary artery calcification as expressed by coronary calcium score, and aortic valve and mitral annulus calcifications [2]. In that study, during 4, 5 to 6 years of follow-up, patients with thoracic aortic calcification had a higher incidence of cardiovascular events in a multivariable analysis, and the severity of calcification was higher in patients with any event than in those without. Therefore, thoracic aortic calcification along with coronary artery and valvular calcifications was associated with an increased risk of death and cardiovascular disease in symptomatic patients [2]. In addition, thoracic aorta calcification has been recognized as an independent predictor of an increased prevalence of cardiovascular events, including hypertension and myocardial infarction [2,9]. In a large population-based cohort of the MESA, a significant correlation between severe thoracic aortic wall calcification and increased coronary heart disease events was founded in women independent of coronary artery calcium [10].
Although numerous studies have demonstrated the predictive value of thoracic aorta calcification in several imaging techniques for future cardiovascular disease in the non-cardiac surgery population, there have been few studies evaluating the relationship between thoracic aorta calcification as measured by CT and clinical outcomes in cardiac surgery patients. Therefore, in thepresent study,we first evaluated thoracic aortic wall calcification quantified on CT scans as a predictor of clinical outcomes in patients undergoing CABG.
As an advance in diagnostic imaging techniques, unrequested information on coronary and aortic calcification collected from routine diagnostic chest CT can predict future cardiovascular events in a clinical care population with fatal and non-fatal cardiovascular disease [11]. Moreover, a recent increase in preoperative chest CT or CT angiography investigations in patients undergoing elective cardiac surgery has led to an increase in unexpected information not related to the clinical indication. Especially in coronary artery disease patients requiring CABG, preoperative CT work-upsprovide numerous abnormal findings of clinical relevance, including the aorta and peripheral vascular wall calcification/stenosis, pulmonary and mediastinal diseases, and skeletal osteoporosis. Therefore, we have routinely performed preoperative chest CT or total aorta CT angiography in CABG patients, and have examined whether subclinical thoracic aortic wall calcification and aortic wall calcium is associated with postoperative clinical outcomes. We used 16-channel, multi-detector CT (MDCT) or 64-channel MDCT, and the scans were obtained with or without contrast media enhancement.It has been reported that inter-scan reproducibility of measurement of coronary calcium using images from MDCT is excellent, irrespective of slice thickness and type of calcium parameter [12,13]. Therefore, it can be acceptable to measure calcium score using two different channel MDCT scanners.
Previous studies showed the variable distribution of TACS. One study demonstrated that a total of 69% of subjects had Agatston TACS of 0; 5% 1 to 9; 12% 10 to 99; 8% 100 to 399; and 7% ≥ 400 in a total of 2,303 asymptomatic adults (mean age 55.7 years, 38% female) with CT scans [14]. In another study, overall 2,538/4,025 (63.1%) participants revealed thoracic aortic wall calcification (median Agatston-score: 17.5, lower quartile 0, upper quartile 124.7) in a general unselected population [15]. In our study, we included specific population with old age and known coronary artery disease. To our knowledge, there is no reference for the threshold of the high TACS for the population. Therefore, we arbitrarily defined the threshold of high TACS for our study population by the use of the percentiles of the TACS distribution as the study by Kalsch et al. [15].
There have been several studies indicating aortic calcification increases arterial stiffness [16-19]. Roos et al. [16] have demonstrated that atherosclerosis in the descending thoracic aorta as measured by DTA thickening and calcium score on the CTA is associated with arterial stiffness as determined by carotid-femoral pulse wave velocity (PWV) in patients with diabetes mellitus. In multivariate analysis, they concluded that DTA calcium score is independently associated with arterial stiffness and that an increase in arterial stiffness by atherosclerosis is mediated and exacerbated by arterial calcification. Although arterial stiffness was not evaluated in the present study, it is thought that poor postoperative clinical outcomes in patients with severe aortic calcification may be associated with the progression of atherosclerosis and arterial stiffness. Diastolic myocardial dysfunction caused by chronic aortic stiffness can be worsened by variable factors associated with cardiac surgery, including cardiopulmonary bypass with hypothermia, transient ischemia/reperfusion injury during procedures, and heart manipulation/volume overload. It may aggravate the systolic and diastolic dysfunction and frequently induce arrhythmia after surgery. In our study, a high TACS was an independent risk factor for postoperative cardiovascular complications.
There are limitations to the current study that must be recognized. First, this study was not performed in a prospective manner and the number of patients enrolled was relatively small. However, a selective enrollment of 100 patients who underwent isolated CABG, and consistent CT protocols with quantification of TACS may compensate for that limitation. Second, the distribution of TACS showed wide range. Because we included subjects retrospectively, we did not make specific inclusion criteria. Third, we did not evaluate independent value of TACS or its additional value for traditional risk prediction methods. Having low statistical power, this work was preliminary report trying to reveal the usefulness of a new tool, thoracic aortic wall calcification, for the prediction of postoperative clinical outcomes. Further study will be necessary to reveal the clinical impact of the TACS.
Conclusions
In conclusion, severe thoracic aortic wall calcification was strongly associated with poor clinical results. Therefore, thoracic aortic wall calcification quantified on preoperative CT scans may be a useful predictor of early postoperative outcomes in patients undergoing isolated CABG.
Conflict of Interests
The authors declared no conflicts of interest with respect to the authorship or publication of this article.
Funding
The authors received no financial support for the research or authorship of this article.
References
-
Takasu J, Budoff MJ, O'Brien KD, Shavelle DM, Probstfield JL, et al. (2009) Relationship between coronary artery and descending thoracic aortic calcification as detected by computed tomography: the Multi-Ethnic Study of Atherosclerosis. Atherosclerosis 204: 440-446.
-
Eisen A, Tenenbaum A, Koren-Morag N, Tanne D, Shemesh J, et al. (2008) Calcification of the thoracic aorta as detected by spiral computed tomography among stable angina pectoris patients: association with cardiovascular events and death. Circulation 118: 1328-1334.
-
Iribarren C, Sidney S, Sternfeld B, Browner WS (2000) Calcification of the aortic arch: risk factors and association with coronary heart disease, stroke, and peripheral vascular disease. JAMA 283: 2810-2815.
-
Takasu J, Katz R, Nasir K, Carr JJ, Wong N, et al. (2008) Relationships of thoracic aortic wall calcification to cardiovascular risk factors: the Multi-Ethnic Study of Atherosclerosis (MESA). Am Heart J 155: 765-771.
-
Nicoll R, Henein MY (2014) The predictive value of arterial and valvular calcification for mortality and cardiovascular events. IJC Heart & Vessels 3: 1-5.
-
Allison MA, Hsi S, Wassel CL, Morgan C, Ix JH, et al. (2012) Calcified atherosclerosis in different vascular beds and the risk of mortality. Arterioscler Thromb Vasc Biol 32: 140-146.
-
Agatston AS, Janowitz WR, Hildner FJ, Zusmer NR, Viamonte M Jr, et al. (1990) Quantification of coronary artery calcium using ultrafast computed tomography. J Am Coll Cardiol 15: 827-832.
-
Demer LL, Tintut Y (2008) Vascular calcification: pathobiology of a multifaceted disease. Circulation 117: 2938-2948.
-
Santos RD, Rumberger JA, Budoff MJ, Shaw LJ, Orakzai SH, et al. (2010) Thoracic aorta calcification detected by electron beam tomography predicts all-cause mortality. Atherosclerosis 209: 131-135.
-
Budoff MJ, Nasir K, Katz R, Takasu J, Carr JJ, et al. (2011) Thoracic aortic calcification and coronary heart disease events: the multi-ethnic study of atherosclerosis (MESA). Atherosclerosis 215: 196-202.
-
Jacobs PC, Gondrie MJ, Mali WP, Oen AL, Prokop M, et al. (2011) Unrequested information from routine diagnostic chest CT predicts future cardiovascular events. Eur Radiol 21: 1577-1585.
-
Sabour S, Rutten A, van der Schouw YT, Atsma F, Grobbee DE, et al. (2007) Inter-scan reproducibility of coronary calcium measurement using Multi Detector-Row Computed Tomography (MDCT). Eur J Epidemiol 22: 235-243.
-
Detrano RC, Anderson M, Nelson J, Wong ND, Carr JJ, et al. (2005) Coronary calcium measurements: effect of CT scanner type and calcium measure on rescan reproducibility-MESA study. Radiology 236: 477-484.
-
Wong ND, Gransar H, Shaw L, Polk D, Moon JH, et al. (2009) Thoracic aortic calcium versus coronary artery calcium for the prediction of coronary heart disease and cardiovascular disease events. J Am Coll Cardiovasc Img 2: 319-326.
-
Kälsch H, Lehmann N, Möhlenkamp S, Hammer C, Mahabadi AA, et al. (2013) Prevalence of thoracic aortic calcification and its relationship to cardiovascular risk factors and coronary calcification in an unselected population-based cohort: the Heinz Nixdorf Recall Study. Int J Cardiovasc Imaging 29: 207-216.
-
Roos CJ, Delgado V, de Koning EJ, Rabelink TJ, Jukema JW, et al. (2014) Associations of atherosclerosis in the descending thoracic aorta on CTA with arterial stiffness and chronic kidney disease in asymptomatic patients with diabetes mellitus. Int J Cardiovasc Imaging 30: 1151-1159.
-
Cecelja M, Hussain T, Greil G, Botnar R, Preston R, et al. (2013) Multimodality imaging of subclinical aortic atherosclerosis: relation of aortic stiffness to calcification and plaque in female twins. Hypertension 61: 609-614.
-
Sekikawa A, Shin C, Curb JD, Barinas-Mitchell E, Masaki K, et al. (2012) Aortic stiffness and calcification in men in a population-based international study. Atherosclerosis 222: 473-477.
-
Tsao CW, Pencina KM, Massaro JM, Benjamin EJ, Levy D, et al. (2014) Cross-sectional relations of arterial stiffness, pressure pulsatility, wave reflection, and arterial calcification. Arterioscler Thromb Vasc Biol 34: 2495-2500.





