International Journal of Surgery Research and Practice
Comparative Study of Laparoscopic Versus Open Resection of Colorectal Liver Metastasis
Christian Hunnicke Petersen*, Peter Nørgaard Larsen, Jan Storkholm, Nicolai Aagaard Schultz and Kenneth Højsgaard Jensen
Department of Gastrointestinal Surgery and Transplantation, Rigshospitalet, Copenhagen University Hospital, Denmark
*Corresponding author: Christian Hunnicke Petersen, Department of Gastrointestinal Surgery and Transplantation, Rigshospitalet, Copenhagen University Hospital, Denmark, E-mail: christian1990hp@gmail.com
Int J Surg Res Pract, IJSRP-3-040, (Volume 3, Issue 1), Original Article; ISSN: 2378-3397
Received: September 12, 2015 | Accepted: March 28, 2016 | Published: March 30, 2016
Citation: Petersen CH, Larsen PN, Storkholm J, Schultz NA, Jensen KH (2016) Comparative Study of Laparoscopic Versus Open Resection of Colorectal Liver Metastasis. Int J Surg Res Pract 3:040. 10.23937/2378-3397/1410040
Copyright: © 2016 Petersen CH, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Introduction: Laparoscopic liver resection is gaining increased application in the treatment of colorectal liver metastases (CRLM). The aim of this study is to compare safety and feasibility of minor laparoscopic liver resection vs. open liver resection in patients with CRLM.
Methods: One hundred and twenty-eight patients who underwent minor resection of CRLM were retrospectively reviewed. Sixty-four consecutive cases of laparoscopic liver resections (LLR) were compared to a matched control group of 64 open liver resections (OLR).
Results: No difference was observed in the rate of negative resection margins (LLR 92 % vs. OLR 91 %) . 1-, 3- and 5-year overall survival was 98 %, 79 %, 54 % vs. 95 %, 81 %, 65 % in LLR and OLR respectively, log rank P = 0.465. In LLR 1-, 3- and 5-year disease free survival was 66 %, 61 % and 61 % vs. 81 %, 58 % and 50 % in OLR, log rank P = 0.890. Length of stay (median, 2 days in LLR vs. 4 days in OLR; p < 0.001) and operative time (median, 92 min vs. 105 min for OLR; p = 0.042) were both significantly shorter in LLR. Blood loss was significantly lower in LLR (median, 100 ml vs. 500 ml in OLR; p < 0.001). Nineteen (30 %) LLR patients had complications vs. 22 (34 %) in OLR. The distribution of Clavien-Dindo grades between the two groups was comparable.
Conclusion: We found minor laparoscopic liver resections of ≤ 4 metastatic lesions to be safe and feasible with shorter length of stay and comparable short and long term oncologic outcomes. Laparoscopic resection may further achieve shorter operative time and lower blood loss.
Keywords
Laparoscopic Surgery, Liver Surgery, Liver Resection, Hepatectomy, Colorectal Cancer, Liver Metastasis, Colorectal Liver Metastasis
Introduction
Fifteen to twenty-five % of patients diagnosed with colorectal cancer have synchronous liver metastases at the time of diagnosis and an additional 13-25 % will develop metachronous liver metastases [1,2]. For patients with colorectal liver metastasis (CRLM), liver resection is considered the only potentially curative treatment with a 5-year survival of 25-40% [3-5]. The laparoscopic approach for liver resections is technically challenging, but reduces the surgical trauma and stress response and allows for quicker healing [6]. The use of laparoscopic liver resection (LLR) is rapidly increasing due to multiple studies finding LLR to be associated with lower blood loss and shorter length of stay while showing oncologic outcomes similar to open liver resection (OLR) [7]. Recommendations for laparoscopic liver resection, stated in the report from the second international consensus conference held in Morioka 2014, concluded that minor LLR (≤ 2 segments) is now considered standard practice while major LLRs (≥ 3 segments) are still innovative procedures in the exploration phase [8]. Prospective randomized controlled trials for comparison of LLR and OLR of colorectal liver metastases has yet to be published but study protocols for randomized controlled trials do exist [9,10]. The aim of this study is to analyze the safety and feasibility of minor laparoscopic resection of CRLM.
Methods
Patients
Laparoscopic and open liver resections performed for all indications at a single high volume hepato-pancreato-biliary centre during the time period of July 2009 to November 2014 were retrieved from the database. The search provided a total of 1467 consecutive patients including 64 minor LLRs performed for CRLM. Patients with hepatic lesions of non-colorectal origin were excluded. A control group of 64 minor OLRs for CRLM were then selected by head-to-head matching with the 64 laparoscopically resected patients on the following matching criteria based on the clinical risk score proposed by Fong [11]: positive node of primary, disease free interval < 1 year, preoperative CEA > 200 ng/ml, number of metastases, size of largest tumour and presence of extrahepatic disease. Patients with tumours located in the anterior or the left lateral segments were preferably selected for laparoscopic resection. Because oncologic outcomes had the higher priority, recurrence prognostic measures have been emphasized on behalf of more technical aspects such as specific tumour location. Patients were further matched by number of liver segments involved in the resections, Charlson comorbidity index (CCI) [12], prior liver resection, gender and age. Short term outcomes included resection margin positivity, blood loss, operative time, blood transfusions, length of stay and complications. Complications were graded according to the Clavien-Dindo classification for surgical complications [13] and operative time was defined as the time from the first incision in the skin to complete closing of the skin at the end of surgery. Long term outcomes were disease free (DFS) and overall survival (OS). In patients with recurrence, DFS was defined as time from surgery to first imaging finding of recurrence. The radiologic follow-up program for CRLM consisted of CT-scans of the thorax and abdomen as well as measurement of carcinoembryonic antigen (CEA) at postoperative month 3, 6, 9, 12, 18, 24, 36, 48 and 60.
Data were retrospectively collected from regional and national patient files. Following the principle of an intention-to-treat analysis, laparoscopic procedures converted to open procedures remained in the laparoscopic group.
Surgical technique
LLR was performed with the patient in the supine and 30° anti-Trendelenburg position with the surgeon standing between the patient’s legs and assistants on one or both sides. 4 to 6 port sites were inserted in the upper abdominal quadrant: the 12-mm ports were placed to allow insertion of a 30° optical device, the linear stapler and LigaSure; the 15-mm port for the surgical aspirator and ultrasound scans, the 5-mm port was used mainly to allow irrigation and aspiration during surgery. Carbon dioxide pressure for pneumoperitoneum was kept between 12-15 mmHg during hepatic parenchymal division. Pressure above 15 mmHg was primarily used to prevent venous bleeding. No pedicle clamping was performed. Operative assessment of tumours and surgical margins was undertaken by intra-operative ultrasound scans. Parenchymal division and coagulation was performed using the LigaSure or staplers. Finally, the specimen was extracted in a plastic bag through the 15-mm port incision. In open liver resections a transverse incision in the right upper quadrant extended upwards in the midline was used. If possible, an upper midline incision was used for resections confined to the left lateral segment. Parenchyma was transected using Waterjet or clamp crushing while the LigaSure was used for division of vessels. Pringle manoeuvre was selectively applied for intervals of up to 16 minutes.
Perioperative care principles
Patients in both groups followed similar fast-track perioperative care principles as described by Schulz et al. [14]. Clinical project nurses systematically informed patients about the optimized perioperative care. Catheters and drains were systematically removed early, and patients were mobilized and started eating and drinking from the day of surgery. All patients received a multimodal pain treatment for one week including 1 g paracetamol every 6 h, 200 mg celecoxib every 12 h, and 300 mg gabapentin in the morning and 600 mg in the evening. Patients in the open group had epidural analgesia for 0-48 h after surgery while patients in the laparoscopic group had no epidural analgesia. All patients in both groups were discharged when meeting all of the following criteria: pain sufficiently controlled by oral analgesics, no untreated surgical complications and the patient feeling comfortable with discharge. Assessment of pain was done at rest and activity at POD 1 and POD 3, using a 100-mm visual analogue scale (VAS) [15]. Discharge was decided by whichever surgeon doing bedside visits on the given day.
Statistical analysis
Statistical analysis was performed using SPSS (SPSS, version 23.0) and Excel (Microsoft version 12.0). Significance was accepted at p < 0.05. Categorical variables were compared between groups using the chi-squared test and continuous data were compared using Mann-Whitney test. Survival was evaluated using Kaplan-Meier curves and log-rank test. Follow-up time was evaluated using Kaplan-Meier estimate of potential follow-up [16].
Results
Short-term results
Patient and tumour characteristics of the study patients are summarized in table 1 and short term results are summarized in table 2. The LLRs were performed by a total of 12 different surgeons with three surgeons performing 50 / 64 (78 %) of LLRs included in this study. To test for the effect of increased experience through the learning curve the first and second half of the LLRs sorted by the date of surgery were compared in terms of median operative time and blood loss. No difference was found in this regard. Eight laparoscopic procedures were converted to open surgery (13%). Laparoscopic procedures were converted for oncologic reasons including unknown metastasis, tumour extend or difficult tumour location (n = 4), strong adhesions or fibrosis due to prior surgery (n = 2), lesion of the gallbladder (n = 1) and bleeding (n = 1). We found significantly shorter length of stay with a median (25:75 percentile) of 2 days (2:4) and 4 days (4:5) in the LLR and OLR group respectively, p < 0.001. Blood loss was significantly lower with a median (range) of 100 ml (0 -2500) in LLR and 500 ml (0-2800) in OLR, p < 0.001. The Pringle manoeuvre was applied in 15/64 (23 %) of cases in OLR and not applied in LLR. Operative time was also found to be shorter in the laparoscopic group with a median (range) of 92 minutes (37-273) vs. 105 minutes (54 - 240) in the open group, p = 0.042. No significant difference in the number of patients receiving blood transfusions was found. Complication rates of 19/64 (30 %) in LLR and 22/64 (34 %) in OLR, p 0.570 as well as the distribution of Clavien-Dindo grades were comparable between the two groups. The LLR group had three grade 3 complications including bleeding from the resected liver surface requiring laparotomy, abscess in the umbilical port wound and liver abscess, respectively treated with percutaneous drainage. In the OLR group four grade 3 complications were seen: liver abscess treated with percutaneous drainage, bile leakage treated by endoscopic retrograde cholangiography with stenting, rupture of fascia and rupture of incisional wound respectively treated with resuturing in universal anaesthesia. No grade four or five complications were seen. Sixty-day mortality was zero in both groups.
![]()
Table 1: Patient and tumour characteristics of the study patients.
View Table 1
![]()
Table 2: Short-term results.
View Table 2
Oncologic results
Microscopically negative resection margins (R0) were achieved in 92 % of LLRs and 91 % of OLRs. Both groups had similar follow-up times with a median (25:75 percentile) of 49 (23:54) months (range 11-74 months) in LLR and 47 (28:52) months (range 5-72 months) in OLR, p = 0.442. 1-, 3- and 5-year overall survival was 98 %, 79 %, 54 % vs. 95 %, 81 %, 65 % in LLR and OLR respectively, log rank P = 0.465. In LLR, 1-, 3- and 5-year disease free survival was 66 %, 61 % and 61 % vs. 81 %, 58 % and 50 % in OLR, log rank P = 0.890. The Kaplan-Meier survival curves are shown in figure 1 and figure 2.
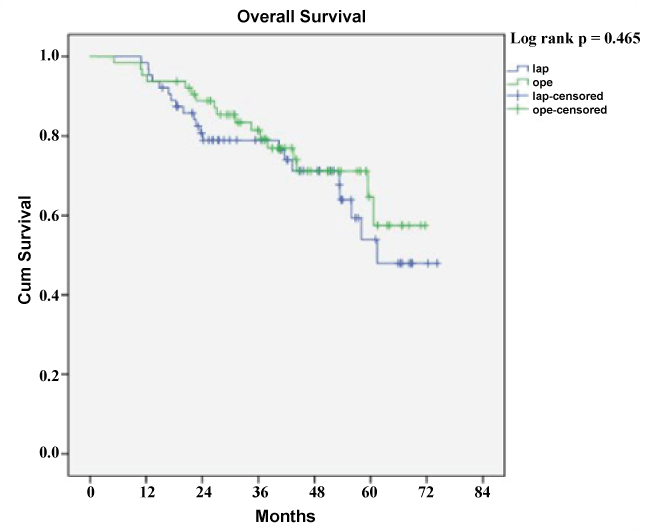
.
Figure 1: Overall survival for LLR and OLR.
Legends: Lap, laparoscopic resection; ope, Open resection; Blue line represents the laparoscopic resection group; Green line represents the open resection group; Cum Survival, cumulative survival.
View Figure 1
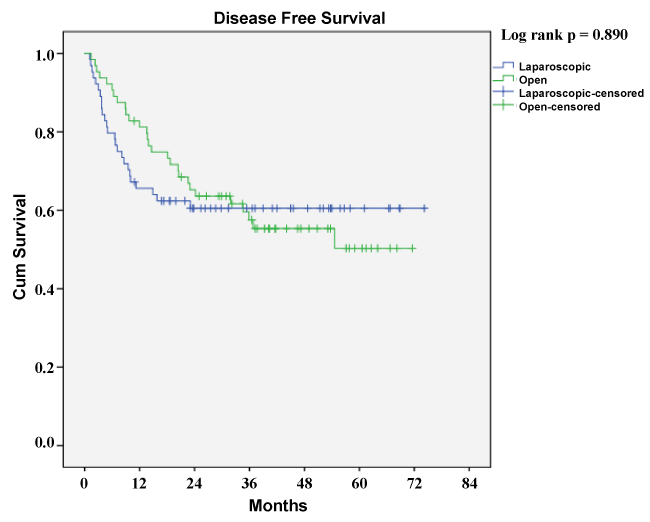
.
Figure 2: Disease free survival for LLR and OLR.
Blue line represents the laparoscopic resection group; Green line represents the open resection group; Cum Survival, cumulative survival.
View Figure 2
Discussion
The application of minor laparoscopic liver resection has become widespread in specialized centres due to multiple findings of shorter length of stay and lower blood loss and has so far been found to be safe when performed in selected patients. We found no difference in the rate of positive resection margins, overall survival or disease free survival. In agreement with our findings meta-analysis have shown no difference in overall or disease free survival between LLR and OLR [17,18].
We found significantly shorter length of stay in LLR. The groups followed similar fast-track principles for perioperative care and discharge criteria as described in methods. The finding of shorter length of stay in LLR is thought mainly to be an advantage of the reduced surgical trauma of LLR and is supported by the majority of comparative studies finding a significantly reduced length of stay for LLR [19-23]. The relatively short length of stay in both groups of our study is due to the fact that this is a study of exclusively minor resections and to the fast track principles for perioperative care aiming for fast recovery and short length of stay. The hepatic lesions were well matched in terms of number and size of metastases as well as prior liver surgery and the number of liver segments involved in the resection. Though matching of specific tumour location would be desirable, the higher priority of matching prognostic characteristics for oncological measures combined with preferred selection of LLR for tumours located in the anterolateral segments (segment 2, 3, 4b, 5 or 6) in LLR makes matching of the specific tumour location in our study extremely difficult. Hence the LLR group included more cases of patients with tumours located in the anterolateral segments (95 % in LLR vs. 70 % in OLR). A higher proportion of tumours located in posterosuperior segments (segment 4a, 7 or 8) in OLR, occasionally needing wide mobilization, as well as more tumours located near hepatic vessels might affect blood loss and operative time. However, the establishment of positive pressure pneumoperitoneum (12-15 mmHg) in LLR, which can be transiently elevated in cases of bleeding as well as the magnified view of the operating field, is thought to make a considerable contribution in reducing blood loss in LLR. The frequent use of staplers in the LLRs further reduces blood loss. The find of reduced blood loss in LLR is in agreement with multiple studies of LLR vs. OLR [19-23]. In LLR the time saved from the opening and closing of the skin and fascia allows for shorter operative time in minor resections of few metastases. The frequent use of staplers for parenchymal dissection in LLR also shortens operative time compared to OLR.
We found no significant difference in the overall rate of complications (30.6 % for LLR vs. 35.5 % for OLR). In the literature the complication rates vary between 15-27 % for LLR and 28 - 50 % for OLR and it remains questionable whether LLR significantly improves complication rates with some studies finding a significant improvement while other studies do not [19-22,24]. The complication rate for LLR in our study is relatively high and might be explained by the fact that our study seems to have a very high proportion of low grade complications (Clavien-Dindo grade 1 and 2) with 16/19 complications being low grade for LLR and 17/22 for OLR. This is most likely due to a very low threshold for inclusion of mild events as complications in our study. The conversion rate was 13 % which is comparable to similar studies ranging from 5 - 15.8 % [20-22,24].
Conclusion
We found minor laparoscopic liver resections of ≤ 4 metastatic lesions to be safe and feasible with shorter length of stay and comparable short and long term oncologic outcomes. Laparoscopic resection may further achieve shorter operative time and lower blood loss.
References
-
Scheele J, Stangl R, Altendorf-Hofmann A. (1990) Hepatic metastases from colorectal carcinoma: impact of surgical resection on the natural history. Br J Surg 77: 1241-1246.
-
Manfredi S, Lepage C, Hatem C, Coatmeur O, Faivre J, et al. (2006) Epidemiology and management of liver metastases from colorectal cancer. Ann Surg 244: 254-259.
-
Fong Y (1999) Surgical therapy of hepatic colorectal metastasis. CA Cancer J Clin 49: 231-255.
-
Nordlinger B, Guiguet M, Vaillant JC, Balladur P, Boudjema K, et al. (1996) Surgical resection of colorectal carcinoma metastases to the liver. A prognostic scoring system to improve case selection, based on 1568 patients. Association Francaise de Chirurgie. Cancer 77: 1254-1262.
-
Simmonds PC, Primrose JN, Colquitt JL, Garden OJ, Poston GJ, et al. (2006) Surgical resection of hepatic metastases from colorectal cancer: A systematic review of published studies. Br J Cancer 94: 982-999.
-
Kazuki Ueda, Patricia Turner, Michel Gagner (2004) Stress response to laparoscopic liver resection. HPB (Oxford) 6: 247-252.
-
Nguyen KT, Marsh JW, Tsung A, Steel JJ, Gamblin TC, et al. (2011) Comparative benefits of laparoscopic vs open hepatic resection: a critical appraisal. Arch Surg 146: 348-356.
-
Wakabayashi G, Cherqui D, Geller DA, Buell JF, Kaneko H, et al. (2015) Recommendations for laparoscopic liver resection: a report from the second international consensus conference held in Morioka. Ann Surg 261: 619-629.
-
van Dam RM, Wong-Lun-Hing EM, van Breukelen GJ, Stoot JH, van der Vorst JR, et al. (2012) Open versus laparoscopic left lateral hepatic sectionectomy within an enhanced recovery ERAS® programme (ORANGE II-trial): study protocol for a randomised controlled trial. Trials 13: 54.
-
Fretland ÅA, Kazaryan AM, Bjørnbeth BA, Flatmark K, Andersen MH, et al. (2015) Open versus laparoscopic liver resection for colorectal liver metastases (the Oslo-CoMet study): study protocol for a randomized controlled trial. Trials 16: 73.
-
Fong Y, Fortner J, Sun RL, Brennan MF, Blumgart LH (1999) Clinical score for predicting recurrence after hepatic resection for metastatic colorectal cancer: analysis of 1001 consecutive cases. Ann Surg 230: 309-318.
-
Charlson ME, Pompei P, Ales KL, MacKenzie CR (1987) A new way of classifying prognostic comorbidity in longitudinal studies: Development and validation. J Chron Dis 40: 373-383.
-
Dindo D, Demartines N, Clavien PA (2004) Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg 240: 205-213.
-
Schultz NA, Larsen PN, Klarskov B, Plum LM, Frederiksen HJ, et al. (2013) Evaluation of a fast-track programme for patients undergoing liver resection. Br J Surg 100: 138-143.
-
Hawker GA, Mian S, Kendzerska T, French M (2011) Measures of adult pain: Visual Analog Scale for Pain (VAS Pain), Numeric Rating Scale for Pain (NRS Pain), McGill Pain Questionnaire (MPQ), Short-Form McGill Pain Questionnaire (SF-MPQ), Chronic Pain Grade Scale (CPGS), Short Form-36 Bodily Pain Scale (SF-36 BPS), and Measure of Intermittent and Constant Osteoarthritis Pain (ICOAP). Arthritis Care Res (Hoboken) 60: 240-252.
-
Schemper M, Smith TL (1996) A note on quantifying follow-up in studies of failure time. Control Clin trials 17: 343-346.
-
Schiffman SC, Kim KH, Tsung A, Marsh JW, Geller DA (2015) Laparoscopic versus open liver resection for metastatic colorectal cancer: A metaanalysis of 610 patients. Surgery 157: 211-222.
-
Luo LX, Yu ZY, Bai YN (2014) Laparoscopic hepatectomy for liver metastases from colorectal cancer: A meta-analysis. J Laparoendosc Adv Surg Tech A 24: 213-222.
-
Cannon RM, Scoggins CR, Callender GG, Robert M. Cannon, Robert C.G. Martin II (2012) Laparoscopic versus open resection of hepatic colorectal metastases. Surgery 152: 567-574.
-
Qiu J, Chen S, Pankaj P, Wu H (2013) Laparoscopic Hepatectomy for Hepatic Colorectal Metastases - A Retrospective Comparative Cohort Analysis and Literature Review. PLoS One 8: e60153.
-
Alfredo D. Guerron, Shamil Aliyev, Orhan Agcaoglu, Erol Aksoy, Halit Eren Taskin, et al. (2013) Laparoscopic versus open resection of colorectal liver metastasis. Surg Endosc 27: 1138-1143.
-
R. Montalti, G. Berardi, S. Laurent, S. Sebastiani, L. Ferdinande, et al. (2014) Laparoscopic liver resection compared to open approach in patients with colorectal liver metastases improves further resectability: Oncological outcomes of a case-control matched-pair analysis. Eur J Surg Oncol 40: 536-544.
-
Cheung TT, Poon RT, Yuen WK, Chok KS, Tsang SH, et al. (2013) Outcome of laparoscopic versus open hepatectomy for colorectal liver metastases. ANZ J Surg 83: 847-852.
-
Castaing D, Vibert E, Ricca L, Azoulay D, Adam R, et al. (2009) Oncologic results of laparoscopic versus open hepatectomy for colorectal liver metastases in two specialized centers. Ann Surg 250: 849-855.





