International Journal of Surgery Research and Practice
Pain Management in Knee Arthroplasty: An Overview
Md Quamar Azam1*, Mir Sadat-Ali1 and Ahmed Bader2
1Department of Orthopedics, College of Medicine, University of Dammam, Saudi Arabia
2Department of Physiology, College of Medicine, University of Dammam, Saudi Arabia.
*Corresponding author: Dr. Md Quamar Azam, Department of Orthopedics, College of Medicine, University of Dammam, PO Box- 40071, Al-Khobar-31952, Saudi Arabia, Tel: +966-532750228, E-mail: qazam47@Gmail.com
Int J Surg Res Pract, IJSRP-2-035, (Volume 2, Issue 2), Review Article; ISSN: 2378-3397
Received: August 06, 2015 | Accepted: December 24, 2015 | Published: December 28, 2015
Citation: Azam MQ, Sadat-Ali M, Bader A (2015) Pain Management in Knee Arthroplasty: An Overview. Int J Surg Res Pract 2:035. 10.23937/2378-3397/1410035
Copyright: © 2015 Azam MQ, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Perioperative pain management after knee arthroplasty has undergone a conceptual revolution in the last decade. Along with other exciting innovations, including minimally invasive techniques, computer-assisted procedures and a significant stride in tribology, understanding pain modulation and drug action at molecular level is recognized as the game changer in arthroplasty surgeries.
While most patients usually recover and experience pain relief within 3 months after TKA, about 20% (10-34%) of the patients are left with an unfavorable long-term pain outcome. According to Andersen et al 52% of patients report moderate pain and 16% report severe pain at rest (PAR) 30 days after TKA, while pain at movement affects as much as 78% of the patients. Inability to adequately control post-op pain causes undue sufferings, inability to participate in fast-track rehabilitation programs, sleep disturbance (44% patients first 3 nights), delayed discharge and the development of persistent postsurgical pain (PPSP).
Goal of this review article is to give an overview of the fundamental concept of surgical pain, the molecular mechanism of action of different drugs, evolution of the concept of preventive analgesia and finally state of the art for current pain management. When combined and standardized, these factors allow arthroplasty surgeons to offer outpatient arthroplasty procedures.
Keywords
Current, Concept, Pain, Management, Arthroplasty
Introduction
Advances in the last decades has been characterized by exciting innovations in total hip and knee arthroplasty, including minimally invasive techniques, computer-assisted procedures, ingenious design significant stride in tribology and improved perioperative pain management. Understanding pain modulation and drug action at molecular level revolutionized postoperative pain management and rehabilitation protocol. Today, most surgeons [1,2] recognize this cultural shift as the game changer in arthroplasty surgeries
While most patients usually recover and experience pain relief within 3 months after TKA [3] about 20% (10-34 %) of the patients are left with an unfavorable long-term pain outcome according. According to Andersen et al. [4] 52 % of patients report moderate pain and 16% report severe pain at rest (PAR) 30 days after TKA, while pain at movement affects as much as 78% of the patients. Inability to adequately control post-op pain causes undue sufferings, inability to participate in fast-track rehabilitation programs, sleep disturbance (44% patients first 3 nights), delayed discharge and the development of persistent postsurgical pain (PPSP) [5-7]. The unexplained painful TKA without problems related to implants continue to be a challenge for the surgeon [5] (55 of 10) and remain a cause of revision surgery [8]. Goal of this article is to give an overview of the fundamental concept of surgical pain, the molecular mechanism of action of different drugs, risk factors for acute and chronic pain, evolution of the concept of preventive analgesia and finally state of the art for current pain management. When combined and standardized, these factors allow arthroplasty surgeons to offer outpatient arthroplasty procedures [9,10].
Patho-Physiology of Pain Pathway
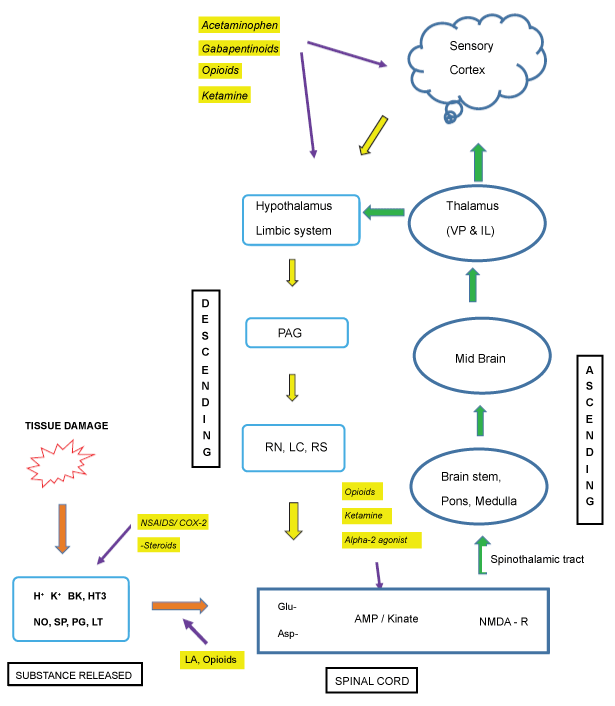
The complex pain-pathway can be briefly summarized as transduction, transmission, modulation and perception (Figure 1).
Transduction (noxious stimuli translated into electrical activities at the sensory nerve endings)
Surgical incision is the starting point which effects potassium, serotonin and histamine release from damaged cells, bradykinin (BK) from injured vessels and prostaglandin (PG) from nerve endings (nociceptors). These mediators induce further recruitment of other inflammatory agents like IL-1, IL-6, TNF-alpha, Substance-P, Acetylcholine (Ach) etc. Substance-P, a vasoactive neuropeptides is responsible for further release of BK, histamine from mast cells and serotonin from platelets all together then stimulates additional nociceptors. Stimulation of nociceptors results in depolarization of nerve endings which is carried to the spinal cord via A-delta and C fibers. Neural depolarization reduces excitatory threshold of nociceptors of both the injured area (primary hyperalgesia) and that of non-injured area (secondary hyperalgesia) and this phenomenon is known as peripheral sensitization [11-13].
A-delta is myelinated fibers responsible for quickly conducting pain perception and accurately localizes pain producing area. C-fibers are unmyelinated and slower in conducting pain information from a diffuse area [14]. These fibers mainly terminate at secondary afferent neurons of Rexed laminae 1 and 2 in the dorsal horn of spinal cord. Adjacent interneuron circuits, descending inputs from higher spinal cord, mid brain areas (periaqueductal areas, raphe nucleus, locus ceruli, reticulo-spinal) and cerebral centers significantly modulates information carried by afferent fibers.
Transmission (synaptic transfer of generated electrical activities from one neuron to another from periphery to higher centers through spinal tracts)
Neurotransmitters of majority of the interneurons are gamma-aminobutyric acid (GABA) and glycine both of which are inhibitory in action. Main excitatory amino acid is alpha amino-3-hydroxy-5-methyl-4-isozole propionic acid (AMPA/Kinate receptor) located at the post-synaptic to primary afferent fibers. . It is important to note that N-methyl-D-aspartate (NMDA) receptors are post-synaptic to interneurons and the AMPA/Kinate and substance-P must be activated prior to NMDA receptor activation [11,15]. The first event at molecular level in the dorsal horn is release of excitatory amino acid like glutamate and aspartate from primary afferent fiber nerve endings (Figure 2). They bind to AMP/ kinate receptors leading to opening of ion channels and depolarization of second order neurons. These voltage sensitive events remove magnesium plug responsible for keeping NMDA receptors in inactive state. Glycine binding also takes place to finally activate NMDA receptors.
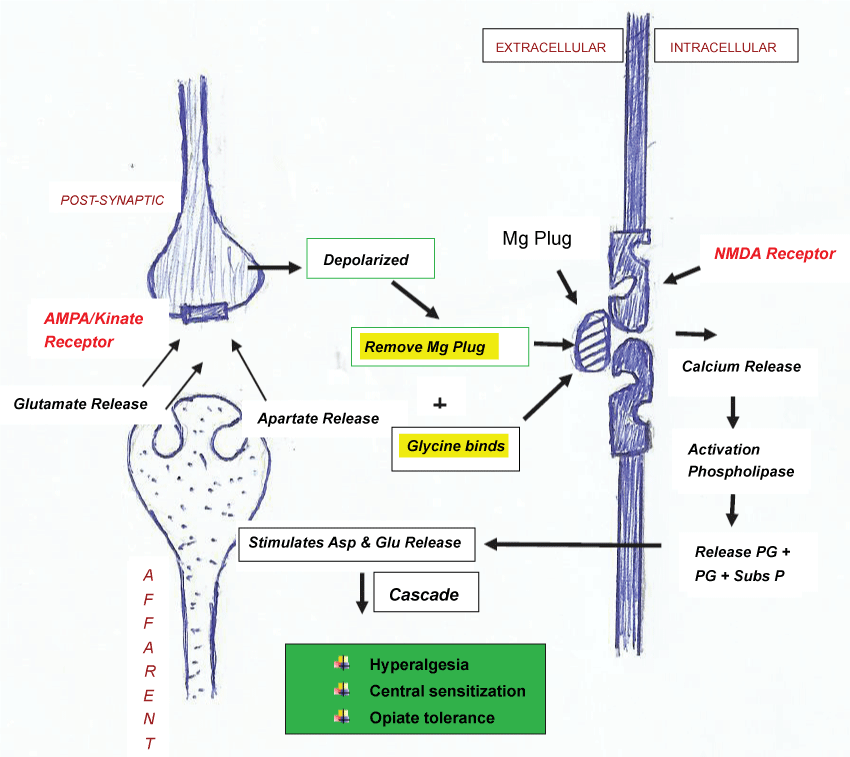
.
Figure 2: Role of NMDA receptor in hyperalgesia, central sensitization & opiate tolerance .
View Figure 2
Now a complex cascade of events occur which include marked release of intracellular calcium which then activates phospholipase-A2, enhances PG production and increases production of substance-P. NMDA activation also causes release of nitric oxide (NO). Both PG and NO diffuses extracellular to induce primary afferent neurons to release excitatory neurotransmitters. It is believed that once the cascade of events is initiated, blockade of peripheral nociceptor inputs fails to completely stop dorsal horn neurons from firing. This wind up phenomenon leads to clinical sequel of hyperalgesia, muscle spasm, allodynia, increase sympathetic tone and subsequent decrease blood flow [16-18]. Higher doses of analgesics such as opiates are required to suppress the pain and hence NMDA receptors are implicated in the development of opiate tolerance. This also explains why long standing pain syndrome fail to improve even after surgical intervention and correction of primary anatomic abnormality. This is responsible for chronic pain and pain syndrome even after successful TKR in some patients who had severe long term osteoarthritis pain. NMDA receptors are also held responsible for the complex phenomenon of central sensitization (ability of benign and low threshold stimuli to activate second order neurons) if afferent nerve stimulation is intense and of sufficient duration [4,11-13,15].
Modulation (release of chemical messengers from higher center and brain stem that modulates the painful stimuli)
After the extensive modulation at dorsal horn, the second degree afferent from the dorsal spinal cord ascends one-two level before crossing to other side and ascends as Crossed Spinothalamic tract. These tracts ends in the thalamus and third order neurons start from here to end in the cerebral cortex and here second degree of modulation takes place also known as central nociceptive processing [6,11,15,16]. On the way there are projections to periaqueductal gray matter (PAG) which is specialized for pain localization. The spinoreticular tract is another ascending tract which synapses at the brainstem reticular formation, before terminating in thalamus and hypothalamus. This tract is particularly responsible for emotional aspect of pain. Descending tracts through PAG and rostral ventromedial medulla inhibit pain transmission due to presence of high concentration of opioid receptors and endogenous opioids.
Perception (Complex interaction in thalamus, cortex, limbic system and reticular system leading to recognition and reaction to primary stimuli)
A large area of cerebral cortex known as "pain matrix" is activated during acute pain activation which includes somato-sensory area (S1 & S2), insular, anterior cingulate cortex as well as thalamus. Thalamus modulation is responsible for sensory discriminative processing whereas cerebral cortex produces emotional and affective responses. This explains why pain perception is affected by factors such as cognition (e.g. distraction or catastrophizing), mood, beliefs and genetics.
Traditional Pain Management
Conventional pain management is unimodal and additive in nature. In other words, it usually involves administration of opioids (injectable) with or without NSAIDS as required. This often requires higher doses of opioids with its potential side effects and the administration of drug being nursing staff dependent usually gets delayed. To overcome this patient control analgesia (PCA) gained worldwide acceptance by both patients and surgeons.
Patient controlled analgesia
PCA is an interesting, attractive, appealing and effective concept which involves patients in pain control management. The PCA uses a microprocessor controlled infusion device that can deliver a continuous baseline dose as well as it is programmed for additional, small and repetitive doses with a lock-out period (5-10 minutes) after each demand dose. The program also decides maximum total dose to be delivered per hour. Lock out time needs calibration in the beginning. Setting the lock out time too short allows the patient to self-administer additional medication (overdose) whereas a prolonged lockout interval fail to give adequate analgesia [4,19,20].
PCA remains a very popular and reasonably viable option however it has certain flaws. One, chances of overmedication with potential adverse effects such as nausea, vomiting, respiratory depression, ileus, urinary retention, pruritus, hypotension, bradycardia, hyperalgesia and cognitive changes. Second, despite the ease of administration and titratability, parenteral opioids typically do not provide adequate analgesia for total joint replacement patients, particularly during movement with ambulation. Movement evoked pain (MEP) is 95-226% more intense than pain at rest (PAR), awakens the patients from sleep, not reduced by opiates and is a potential risk of persistent postsurgical pain syndrome (PPSP) [4,21]. Finally, by activation of NMDA receptors in the central nervous system opioids induces a complex phenomenon of hyperalgesia by paradoxically lowering the pain threshold. This in turn rapidly escalates greater opioid requirements (overdose) in an attempt to reduce pain following surgical procedure with unacceptable side effects. This phenomenon may be minimized by limiting opioids and maximizing non-opioid drugs [22].
Transdermal PCA utilizes ionotophoresis technology to deliver drug (fentanyl) through the skin by use of an external electric field [23-25]. This includes needle free credit card sized system that is applied to the patient's upper arm or chest for 24 hours. The system utilizes on demand delivery dose of 40 microgram of fentanyl for 10 min upto six doses an hour with a maximum dose of 80 doses.
Definition and Constitution of Multimodal Pain (MMP) Control?
Multimodal analgesia is a multidisciplinary approach to pain management which takes advantage of synergistic effects of various analgesics with different mechanism of action (Figure 1) to achieve maximum control of pain with minimum side effects [26]. A multimodal pain protocol consisting of (i) Patient education, (ii) Pre-emptive oral pain medications pre-operatively, (iii) Preference to regional anesthesia, (iv) Peripheral nerve block (v) Intra-operative modalities like periarticular infiltration of cocktail of drugs and finally a standardized post-op rehabilitation program and few more references). Because many of the negative effects of analgesic therapy are related to parenteral opioids, limiting their use is a major principle of multimodal analgesia [2,19,27].
Education
Pain perception has two major components: the sensory discriminating component and affective-motivational component, which underlies the emotional effects of the pain and is responsible for learned avoidance and other behavioral responses. MMP control begins with patient education in the form of preoperative interactions with patients and relatives to draw realistic goal, to explain various steps the patient would undergo, the likely things to happen and the supportive care available. This decreases anxiety and the 'fear of the unknown' and hence improve co-operation and decrease in pain score and overall better patient satisfaction [28,29].
Preemptive analgesia
It involves the administration of analgesics prior to painful stimuli to prevent peripheral and central sensitization. This starts before surgery and covers period of surgery and the initial period of postoperative period. Ref 1 Preemptive analgesia is more effective only when the treatment used is adequate to break the vicious cycle of noxious stimuli recruiting more and more neural pathway. The interventions, therefore, must produce a dense blockade of appropriate duration in order to block the transmission of noxious afferent information from the peripheral nervous system to the spinal cord and the brain [30]. Scientific communities have attempted multiple modifications in the pursuit of uniformly acceptable multi-modal pain control regimen in the last few years. This include fixed dose vs. titrated dose of opioids in PCA, single dose vs. continuous epidural analgesia, single shot vs. continuous peripheral nerve blocks (femoral, sciatic or adductor nerve block) and periarticular soft tissue infiltration of drugs.
Drugs commonly used in MMP control
The various drugs employed are acetaminophen, COX inhibitors, opioids (and its derivatives), gabapentinoids, dexamethasone etc.
a. Acetaminophen: Acetaminophen is a non-opioid, non-NSAID analgesic. Its action is predominantly by inhibiting prostaglandin synthesis in the central nervous system, which possibly play a role in preventing central sensitization syndrome. These attributes make it an integral part of most multimodal postoperative pain regimen. Oral acetaminophen continues to be most commonly prescribed route however its intravenous administration is very effective and reduces post-op opioids consumption [31]. In an adult weighing > 50 kg the 1g of intravenous paracetamol can be administered four times a day. However, in those weighing < 50 kg the safe dose is 15 mg/kg with a maximum dose of 60 mg/kg a day.
b. Non-steroidal anti-inflammatory drugs and COX-2 inhibitors: NSAIDs operate by binding and inhibiting cyclo-oxygenase enzymes (COX) involved in the conversion of arachidonic acid to prostaglandins which truncates the inflammatory mediator cascade. By inhibiting reduction of PG synthesis both peripherally and centrally, NSAIDS not only prevents peripheral tissue sensitization and secondary hyperalgesia effect on the uninjured tissues but also minimizes central sensitization.
The COX-1 isoenzyme, ubiquitous in gastric mucosa, platelets, kidneys, and liver, and is involved in maintaining normal organ function, such as mucosal blood flow and barrier function in the stomach, and in mediating platelet aggregation. Hence, use of nonselective NSAIDS prior to surgery potentially increases bleeding (by decreasing platelet aggregation), gastritis and peptic ulcer disease, renal impairment, and poor wound healing. NSAIDs are therefore relatively contraindicated in patients who are anticoagulated with warfarin, unfractionated and low molecular weight heparin (LMWH), Factor Xa inhibitor (rivaroxaban), direct thrombin inhibitors (dabigatran and fondaparinaux).
The other COX-2 isoenzyme typically present at lower level in these organs is produced primarily in inflamed tissues. COX-2 selective inhibitors lack platelet inhibition and adverse gastrointestinal effects, however, its long term use has fallen out of favor due to its unfavorable risk of cardiovascular side effects. Published literature demonstrated that use of COX-2 inhibitors (celecoxib & rofecoxib) as pre-emptive analgesia significantly reduces pain scores as compared to placebo at 1 hr., 2 hr., and 24 hours after surgery [32]. It is further established use of COX-2 significantly reduces consumption of opioids (referred as opioid sparing), faster time to physical rehabilitation, reduced nausea and vomiting, better sleep patterns and greater patient satisfaction after surgery [33,34]. Alexander et al. [35] showed that a single dose of pre-operative diclofenac or ketorolac reduced morphine consumption by 29% compared to placebo with an additional decrease in post-operative nausea, vomiting and pruritus in patients undergoing joint arthroplasty.
c. Glucocorticoids: Glucocorticoids are known for their potent anti-inflammatory properties. It is established that a single preoperative dose of long acting glucocorticoids like dexamethasone and methylprednisolone are effective in preventing post-operative nausea and vomiting [36-38]. Additionally, dexamethasone has been shown to reduce post-operative dynamic pain [39]. Koh et al. [40] concluded that dexamethasone as part of a multimodal regimen reduced post-operative emesis and pain without an increased risk of wound complications.
d. Opioids: Opioid analgesics have long been, and continue to be, part of the surgeon's armamentarium for the treatment of post-operative pain. Opiates exert their effect by binding to three principal opioid receptors viz. mu, kappa and delta [13]. Opiates or its synthetic derivatives (meperidine or fentanyl) are metabolized in the liver and its onset of action largely depends on lipid solubility. Lipid soluble agents such as meperidine and fentanyl being has rapid action but of short duration as it crosses blood brain barrier easily. In presence of renal impairment morphine and meperidine is avoided while fentanyl, hydromorphone, and oxycodone (which have minimal renal excretion) is preferred.
Oral opioids are available in immediate-release and controlled-release formulations. The former are effective in relieving moderate to severe pain, but require administration every 4 hours. Interruption in timely administration offer suboptimal pain control particularly in the night. The Acute Pain Management Guidelines developed by the Agency for Healthcare Policy and Research [41] recommend a fixed dosing schedule for all patients requiring opioid medications for more than 48 hours postoperatively. The adverse effects of oral opioid administration are considerably less than those of intravenous administration and are mainly gastrointestinal in nature.
i. Morphine: Is metabolized to its active metabolite morphine-6-glucuronide (M6G) in the liver and its half-life of between two and five hour and finally this is excreted in the urine. In patients with renal impairment (estimate glomerular filtration rate < 60 ml/min/1.73m2) there is a risk of respiratory depression due to accumulation of M6G. Morphine may cause significant biliary and urinary tract spasm and few cardiovascular effects.
ii. Fentanyl: It is a synthetic opiates with half-life of 2 to 3 hours which demonstrate rapid onset of action due to lipid solubility but for shorter duration because of redistribution. It has excellent cardiovascular safety profile. It is available as transdermal patches (25, 50, 75 or 100 μg/h) which last for 72 hours, transoral formulations and also is often administered intrathecally and epidurally. It is useful for PCA analgesia, with typical 4-hour doses of 200 to 400 μg depending on weight of patient and tolerance.
iii. Oxycodone: Controlled-release formulation of oxycodone demonstrated excellent and sustained pain relief over an extended period of upto 72 hours and is associated with less sedation, vomiting, or sleep disturbances.
iv. Codeine: Codeine is probably the most widely prescribed oral opiate which differs from other opiates in having a ceiling effect at 60 mg. It has a relatively low incidence of euphoria and thus perhaps a lower abuse potential however nausea and vomiting are common side effects. Its semisynthetic derivative hydrocodone is more potent with lesser side effects and is commonly prepared in combination with ibuprofen.
v. Tramadol: It is another synthetic analog of codeine. Besides being a weak μ-receptor agonist it blocks serotonin and norepinephrine reuptake similar to tricyclic antidepressants. It has excellent oral bioavailability with a half-life of 4 to 6 hours. Tramadol has gained popularity because of the low incidence of adverse effects, specifically respiratory depression, constipation, and potential for abuse.
e. Gabapentinoids (pregabalin and gabapentin): Pregabalin and its predecessor gabapentin were first developed as an anticonvulsant medication. It has shown promising result in neuropathic pain by acting on the voltage gated calcium channels in the central nervous system through alpha2-delta (α2-δ) subunit thereby decreasing neurotransmitter release. Neuropathic pain is a complex phenomenon recognized in as high as 12.7% of patients following TKA. Use of pregabalin in the perioperative period reduces neuropathic pain minimizes opioid consumption and lessens sleep disturbance [42,43]. They also demonstrate synergistic effect with COX-2 inhibitors in clinical studies. The adverse side effects of gabapentinoids include dizziness and somnolence with long-term use.
f. Ketamine: Ketamine is a noncompetitive N-methyl-d-aspartate (NMDA) receptor antagonist that may play a critical role in the intensity of perceived postoperative pain and preventing central sensitization. Ketamine has opioid-sparing effects but no reduction in opioid-related adverse effects. In patients undergoing knee arthroplasty, a low-dose infusion of ketamine (3 μg/kg per minute intra-operatively and 1.5 μg/kg per minute for 48 hours postoperatively reduced morphine requirements and decreased the time to achieve 90° of active flexion.31 Intravenous ketamine can be used in conjunction with femoral nerve blocks or epidural analgesia after total knee arthroplasty. It can also be infiltrated into the wound.
Anesthesia option
Hypotensive regional anesthesia (spinal, epidural or combined) gained rapid popularity over general anesthesia as it avoids CNS depression and by modest reduction in arterial blood pressure it also contributes to reduced surgical blood loss and decreases risk of deep venous thrombosis (DVT) and thrombo-embolism. Furthermore it has less cardiac and respiratory depression. Besides neuraxial anesthesia allows post-operative pain management by appropriately titrating dose of opioids with local anesthetics.
Neuraxial/Regional Anaesthesia: Spinal and epidural opioids provide superior analgesia compared with systemic opioids. The onset and duration of neuraxial opioids are determined by the lipophilicity of the drug. For example, lipophilic opioids, such as fentanyl, provide a rapid onset of analgesia (10-15 min), effective for shorter duration (2-4 hours), limited spread within the cerebrospinal fluid (and hence less respiratory depression), and rapid clearance and resolution. Conversely, hydrophilic opioids, including morphine and hydromorphone, have a longer duration of action (18-24 hours) but are associated with a greater frequency of side effects, such as pruritus, nausea, and vomiting, as well as delayed respiratory depression [44]. Patients given a single dose of extended release epidural morphine [45] have demonstrated 48 hour period of analgesia.
Though epidural anesthesia (EA) is preferred by some clinicians the limitations are failed or dislodged catheters. Epidural catheter used for analgesia also leads to motor blockade (quadriceps weakness necessitating use of knee immobilizer), numbness in contralateral limb, urinary retention and chances of epidural hematoma, if clexane or fondaparinaux is concomitantly used for DVT prophylaxis. It is important to note that the typical side effects of opioids are much more common (and more prolonged) after neuraxial administration compared with all other routes. For example, in a large series, the frequency of pruritus, nausea and vomiting, and respiratory depression was 37%, 25%, and 3%, respectively, with an intrathecal morphine injection [46]. Therefore, patients who exhibit sensitivity to an opioid when administered systemically should not receive that agent neuraxially.
Peripheral nerve block (PNB)
Use of nerve stimulator and ultrasound have facilitated accurate localization of the neural structure and thereby significantly improved their safety, and success rate. Recently, single-dose and continuous peripheral nerve block techniques (femoral, sciatic and adductor canal block) have drawn attention in joint replacement surgeries because of its ability to provide quality of analgesia similar to those of continuous epidural analgesia, but with fewer side effects (reduced incidence of arterial hypotension or urinary retention) besides being compatible with anticoagulants [47-50].
After total knee arthroplasty, patients receiving epidural analgesia or continuous femoral block reported lower pain scores, better knee flexion, faster ambulation, and shorter hospital stays than did patients who received intravenous PCA morphine. However, continuous femoral block was the preferred analgesic technique in each study because fewer technical problems and fewer side effects were noted compared with the epidural and PCA approaches. Recent literature notes that 90% of patients undergoing minimally invasive primary hip or knee replacement were ready for discharge from the hospital within 48 hours if continuous peripheral nerve blocks is combined with multiple scheduled analgesics [2,51].
Neurologic dysfunction and intravascular injection are the primary concerns associated with peripheral blockade. However, in a large series involving more than 50,000 peripheral blocks, there were six seizures, and 12 patients (0.02%) reported postoperative nerve injury. Most neurologic complications were tran-sient [52].
Techniques
Femoral block
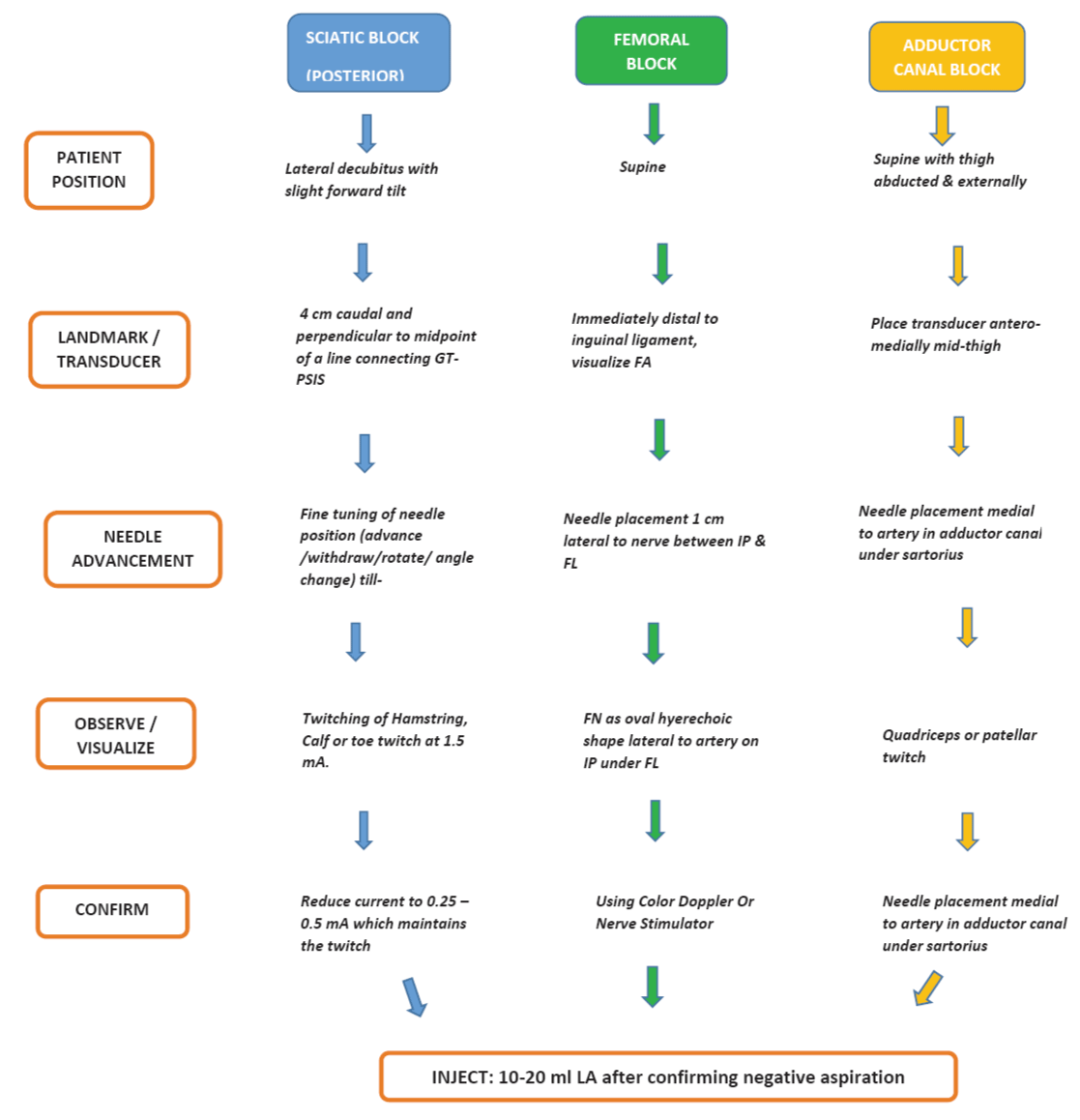
Lumbar plexus block can be achieved by psoas approach, femoral approach or fascia iliaca approach. Psoas approach blocks the femoral nerve along with lateral femoral cutaneous nerve and obturator nerve rendering it preferable in hip replacement surgeries. For knee replacement femoral approach and fascia iliaca approaches are sufficient.
Patient is positioned supine and a linear transducer (8-14 MHz) is placed just below inguinal ligament and moved slightly medial or lateral to identify femoral artery (Figure 3). If unable to visualize tilting transducer cranio-caudal helps artery visualization or color Doppler is used. Then femoral nerve is located lateral to the artery as hyperechoic triangular or oval shaped on the iliopsoas muscle under fascia lata. Needle is placed 1 cm lateral to the nerve, after negative aspiration 15-25 ml of local anesthetic drug is injected. If nerve stimulator is used, presence of presence of patellar or quadriceps twitch between 0.3-0.5 mA confirms appropriate position of needle.
Sciatic nerve block
A sciatic nerve block results in anesthesia of the skin of the posterior aspect of the thigh, hamstring, and biceps femoris muscles; part of the hip and knee joint; and the entire leg below the knee with the exception of the skin of the medial aspect of the lower leg.
Sciatic nerve block can be given by posterior approach or anterior approach. In the classic posterior approach, the patient is positioned in lateral decubitus with slight forward tilt. Needle insertion is 4-5 cm caudal and perpendicular to the midpoint of the line connecting greater trochanter and posterior superior iliac spine (Figure 3). A 10-cm (4-in) stimulating needle is advanced until either a tibial or peroneal motor response is elicited, then 20-30 mL of local anesthetic is incrementally injected. The nerve stimulator may be used to deliver an initial current intensity of 1.5 mA to visualize twitches of either tibial or peroneal motor response (hamstring, calf, foot, or toe twitches). The stimulating current is gradually decreased until twitches are still seen or felt at 0.2 to 0.5 mA which typically occurs at a depth of 5 to 8 cm. At this low current intensity, any observed motor response is from the stimulation of the sciatic nerve, rather than direct muscle stimulation (false twitch). After negative aspiration for blood, 15 to 20 mL of local anesthetic is injected slowly.
Adductor canal block (Hunter's block)
Adductor canal block targets the saphenous nerve (sensory only branch of the femoral nerve) in adductor canal. Saphenous nerve travels between the medial border of the vastus medialis muscle and the medial border of the adductor muscles, along the femoral artery and courses from lateral to medial beneath the Sartorius muscle. Being pure sensory block it relieves pain and also spares quadriceps muscle thereby facilitates physiotherapy, rehabilitation and early independent ambulation.
Patient lies supine with knee slightly flexed and leg externally rotated. A high frequency, linear array ultrasound probe is placed in the antero-medial thigh to first localize femoral artery and traces it distally where it gives off genicular artery (Figure 3). Doppler scanning may be used in difficult situation to trace the femoral artery caudally from the inguinal crease. Once the femoral artery is identified, the needle is inserted in-plane in a lateral-to-medial orientation, and advanced toward the femoral artery. Place the needle tip just medial to the artery in the adductor canal, underneath the Sartorius muscle. Saphenous nerve is identified along the genicular artery. If nerve stimulation (0.5 mA) is used, the passage of the needle through the Sartorius and/or adductor muscles and into the adductor canal is usually associated with the patient reporting a paresthesia in the saphenous nerve distribution. After confirming needle placement and negative aspiration, 15 ml of local anesthetic drug is injected.
Peri-articular soft tissue infiltration
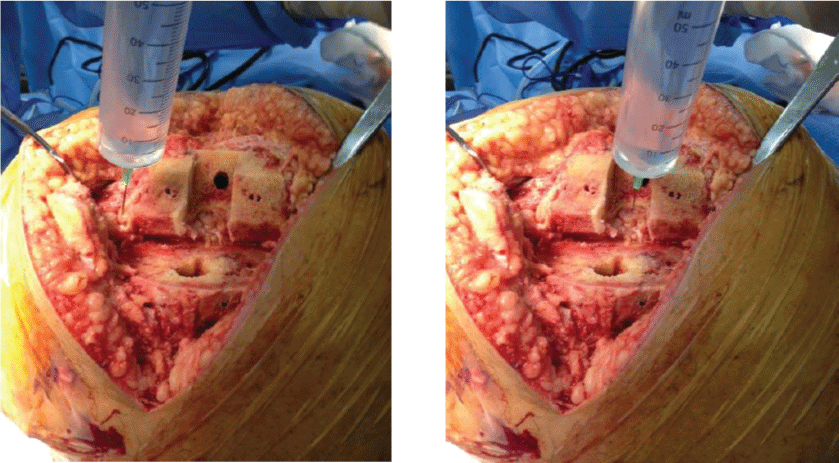
Periarticular infiltration in TKA is a technique where a cocktail of drug combination is injected in the peri-articular soft tissues such as posterior capsule, medial and lateral collateral ligaments, quadriceps mechanism and peripatellar tissue at the end of the surgery (Figure 3). Though there is no standardized protocol but usually it is a combination of long acting local anesthetic drug along with steroids, morphine and epinephrine. Steroids prevent local inflammation, morphine blocks the three opiate receptors and epinephrine prolongs the action of local anesthesia by decreasing its absorption via its alpha-2 adrenergic effect. Injection of the mixture is avoided in the posterolateral corner to prevent inadvertent injury to the peroneal nerve (Figure 4).
Recent studies [53-60] found that periarticular injection significantly improved pain relief from 24 hours to one week and straight leg raise in the early postoperative period. In addition, the meta-analysis of opioid consumption via PCA also corresponded well with the outcome of pain score, which showed that periarticular injection consumed significantly less PCA during the first 24 h postoperatively. The meta-analysis showed that more patients could do active straight leg raise in the injection group than that in noinjection group from postoperative 1 day to discharge day. Ropivacaine has the similar efficiency to bupivacaine but is associated with a longer acting and fewer complications in nervous and cardiovascular systems [53]. Some prefer leaving an indwelling catheter tube before joint closure for postop analgesia.
Author's Preferred Method
All patients received Pregabalin 150 mg in the night before and celecoxib 400 mg orally two hours before surgery with a sip of water. 8 mg IV dexamethasone (for anti-inflammatory and to minimize nausea and vomiting). A cocktail of drug mixture (Figure 5) is injected after component implantation, into the posterior aspect of the joint capsule, medial and lateral collateral ligaments, soft tissue around the quadriceps tendon and patellar tendon, fat pad, and synovium. Rationale of ropivacaine is its longer action and superior cardiovascular and neurotoxic profile. Morphine relieves pain by acting on morphine receptors present in the articular activity and addition of adrenaline slows release of ropivacaine into the vascular system.
A standard postoperative regimen included perfalgan, celecoxib, and pregabalin along with morphine/fentanyl or tramadol as rescue drugs (Figure 5). Onandesterone 4 mg is used to control vomiting in the post op period and pantoprazole 40 mg daily is given for gastrointestinal prophylaxis till discharge. Liberal ice packs application, limb elevation and pneumatic compression device both legs were other measures to reduce swelling. Standard DVT prophylaxis is followed; active straight leg raising and range of motion was is encouraged from the same day. Patient is discharged usually at 3rd post-op day after they gain 80-90 degree flexion and can walk independently with support for at least 50 feet.
Conclusion
Fundamental understanding of different modes of pain control techniques along with limitation of each technique is the first vital step towards effective control of post-surgical pain. A close observation to notice potential side effect of any drug is the next most important thing. We must move forward towards a more holistic, interdisciplinary, multimodal approach to pain medicine. Patient involvement by educating them about the realistic goal is the cornerstone of pain management.
References
-
Maheshwari AV, Blum YC, Shekhar L, Ranawat AS, Ranawat CS (2009) Multimodal pain management after total hip and knee arthroplasty at the Ranawat Orthopaedic Center. Clin Orthop Relat Res 467: 1418-1423.
-
Berend ME, Berend KR, Lombardi AV Jr (2014) Advances in pain management: game changers in knee arthroplasty. Bone Joint J 96: 7-9.
-
Vilardo L, Shah M (2011) Chronic pain after hip and knee replacement. Tech Reg Anesth Pain Manag 15: 110-115.
-
Grosu I, Lavand'homme P, Thienpont E (2014) Pain after knee arthroplasty: an unresolved issue. Knee Surg Sports Traumatol Arthrosc 22: 1744-1758.
-
Hofmann S, Seitlinger G, Djahani O, Pietsch M (2011) The painful knee after TKA: a diagnostic algorithm for failure analysis. Knee Surg Sports Traumatol Arthrosc 19: 1442-1452.
-
Kehlet H, Jensen TS, Woolf CJ (2006) Persistent postsurgical pain: risk factors and prevention. Lancet 367: 1618-1625.
-
Wylde V, Rooker J, Halliday L, Blom A (2011) Acute postoperative pain at rest after hip and knee arthroplasty: severity, sensory qualities and impact on sleep. Orthop Traumatol Surg Res 97: 139-144.
-
Mont MA, Serna FK, Krackow KA, Hungerford DS (1996) Exploration of radiographically normal total knee replacements for unexplained pain. Clin Orthop Relat Res: 216-220.
-
Chen D, Berger RA (2013) Outpatient minimally invasive total hip arthroplasty via a modified Watson-Jones approach: technique and results. Instr Course Lect 62: 229-236.
-
Cross MB, Berger R (2014) Feasibility and safety of performing outpatient unicompartmental knee arthroplasty. Int Orthop 38: 443-447.
-
Phillips WJ, Currier BL (2004) Analgesic pharmacology: I. Neurophysiology. J Am Acad Orthop Surg 12: 213-220.
-
Serpell M (2006) Anatomy, physiology and pharmacology of pain. Surgery 24: 350-353.
-
Basbaum AI, Bautista DM, Scherrer G, Julius D (2009) Cellular and molecular mechanisms of pain. Cell 139: 267-284.
-
Murinson BB, Griffin JW (2004) C-fiber structure varies with location in peripheral nerve. J Neuropathol Exp Neurol 63: 246-254.
-
Phillips WJ, Currier BL (2004) Analgesic pharmacology: II. Specific analgesics. J Am Acad Orthop Surg 12: 221-233.
-
Kawasaki Y, Zhang L, Cheng JK, Ji RR (2008) Cytokine mechanisms of central sensitization: distinct and overlapping role of interleukin-1beta, interleukin-6, and tumor necrosis factor-alpha in regulating synaptic and neuronal activity in the superficial spinal cord. J Neurosci 28: 5189-5194.
-
Woolf CJ, Chong MS (1993) Preemptive analgesia-treating postoperative pain by preventing the establishment of central sensitization. Anesth Analg 77: 362-379.
-
Chen L, Huang LY (1992) Protein kinase C reduces Mg2+ block of NMDA-receptor channels as a mechanism of modulation. Nature 356: 521-523.
-
Horlocker TT, Kopp SL, Pagnano MW, Hebl JR (2006) Analgesia for total hip and knee arthroplasty: a multimodal pathway featuring peripheral nerve block. J Am Acad Orthop Surg 14: 126-135.
-
Dalury DF, Lieberman JR, Macdonald SJ (2012) Current and innovative pain management techniques in total knee arthroplasty. Instr Course Lect 61: 383-388.
-
Srikandarajah S, Gilron I (2011) Systematic review of movement-evoked pain versus pain at rest in postsurgical clinical trials and meta-analyses: a fundamental distinction requiring standardized measurement. Pain 152: 1734-1739.
-
Parvizi J, Porat M, Gandhi K, Viscusi ER, Rothman RH (2009) Postoperative pain management techniques in hip and knee arthroplasty. Instr Course Lect 58: 769-779.
-
Ashburn MA, Streisand J, Zhang J, Love G, Rowin M, et al. (1995) The iontophoresis of fentanyl citrate in humans. Anesthesiology 82: 1146-1153.
-
Viscusi ER, Reynolds L, Taint S, Melson T, Atkinson LE (2006) An Iontophoretic Fentanyl Patient-Activated Analgesic Delivery System for Postoperative Pain: A Double-Blind, Placebo-Controlled Trial. Anesth Analg 102: 188-194.
-
Viscusi ER, Reynolds L, Chung F, Atkinson LE, Khanna S (2004) Patient-Controlled Transdermal Fentanyl Hydrochloride vs Intravenous Morphine Pump for Postoperative Pain: A Randomized Controlled Trial. JAMA 291: 1333-1341.
-
Kehlet H, Dahl JB (1993) The value of "multimodal" or "balanced analgesia" in postoperative pain treatment. Anesth Analg 77: 1048-1056.
-
Maheshwari AV, Boutary M, Yun AG, Sirianni LE and Dorr LD (2006) Multimodal Analgesia without Routine Parenteral Narcotics for Total Hip Arthroplasty. Clin Orthop Relat Res 453: 231-238.
-
Dorr LD (2006) The new process of total hip replacement. In: Hip arthroplasty. Minimally invasive techniques and computer navigation. Philadelphia, PA: Saunders Elsevier, pp 1-14.
-
Giraudet-Le Quintrec JS, Coste J, Vastel L, Pacault V, Jeanne L, et al. (2003) Positive effect of patient education for hip surgery: a randomized trial. Clin Orthop Relat Res: 112-120.
-
Kissin I (2000) Preemptive analgesia. Anesthesiology 93: 1138-1143.
-
Sinatra RS, Jahr JS, Reynolds LW, Viscusi ER, Groudine SB, et al. (2005) Efficacy and safety of single and repeated administration of 1 gram intravenous acetaminophen injection (paracetamol) for pain management after major orthopedic surgery. Anesthesiology 102: 822-831.
-
Reuben SS, Bhopatkar S, Maciolek H, Joshi W, Sklar J (2002) The preemptive analgesic effect of rofecoxib after ambulatory arthroscopic knee surgery. Anesth Analg 94: 55-59.
-
Buvanendran A, Kroin JS, Tuman KJ, Lubenow TR, Elmofty D, et al. (2003) Effects of perioperative administration of a selective cyclooxygenase 2 inhibitor on pain management and recovery of function after knee replacement: a randomized controlled trial. JAMA 290: 2411-2418.
-
Reuben SS, Connelly NR (2000) Postoperative analgesic effects of celecoxib or rofecoxib after spinal fusion surgery. Anesth Analg 91: 1221-1225.
-
Alexander R, El-Moalem HE, Gan TJ (2002) Comparison of the morphine-sparing effects of diclofenac sodium and ketorolac tromethamine after major orthopedic surgery. J Clin Anesth 14: 187-192.
-
Pulos N, Sheth N (2014) Perioperative Pain Management Following Total Joint Arthroplasty. Ann Orthop Rheumatol 2: 1029.
-
Miyagawa Y, Ejiri M, Kuzuya T, Osada T, Ishiguro N, et al. (2010) Methylprednisolone reduces postoperative nausea in total knee and hip arthroplasty. J Clin Pharm Ther 35: 679-684.
-
Backes JR, Bentley JC, Politi JR, Chambers BT (2013) Dexamethasone reduces length of hospitalization and improves postoperative pain and nausea after total joint arthroplasty: a prospective, randomized controlled trial. J Arthroplasty 28: 11-17.
-
Kardash KJ, Sarrazin F, Tessler MJ, Velly AM (2008) Single-dose dexamethasone reduces dynamic pain after total hip arthroplasty. Anesth Analg 106: 1253-1257.
-
Koh IJ, Chang CB, Lee JH, Jeon YT, Kim TK (2013) Preemptive low-dose dexamethasone reduces postoperative emesis and pain after TKA: a randomized controlled study. Clin Orthop Relat Res 471: 3010-3020.
-
(1992) Acute pain management: operative or medical procedures and trauma, Part 2. Agency for Health Care Policy and Research. Clin Pharm 11: 391-414.
-
Mathiesen O, Jacobsen LS, Holm HE, Randall S, Adamiec-Malmstroem L, et al. (2008) Pregabalin and dexamethasone for postoperative pain control: a randomized controlled study in hip arthroplasty. Br J Anaesth 101: 535-541.
-
Lee JK, Chung KS, Choi CH (2015) The effect of a single dose of preemptive pregabalin administered with COX-2 inhibitor: a trial in total knee arthroplasty. J Arthroplasty 30: 38-42.
-
Rathmell JP, Lair TR, Nauman B (2005) The role of intrathecal drugs in the treatment of acute pain. Anesth Analg 101: S30-43.
-
Viscusi ER, Martin G, Hartrick CT, Singla N, Manvelian G (102) Forty-eight hours of postoperative pain relief after total hip arthroplasty with a novel, extended-release epidural morphine formulation. Anesthesiology 102: 937-47.
-
Fu PL, Xiao J, Zhu YL, Wu HS, Li XH, et al. (2010) Efficacy of a multimodal analgesia protocol in total knee arthroplasty: a randomized, controlled trial. J Int Med Res 38: 1404-1412.
-
Chelly JE, Greger J, Gebhard R, Coupe K, Clyburn TA, et al. (2001) Continuous femoral blocks improve recovery and outcome of patients undergoing total knee arthroplasty. J Arthroplasty 16: 436-445.
-
Kaloul I, Guay J, Cote C, Fallaha M (2004) The posterior lumbar plexus (psoas compartment) block and the three-in-one femoral nerve block provide similar postoperative analgesia after total knee replacement. Can J Anesth 51: 45-51.
-
Ben-David B, Schmalenberger K, Chelly JE (2004) Analgesia after total knee arthroplasty: is continuous sciatic blockade needed in addition to continuous femoral blockade? Anesth Analg 98: 747-749.
-
Pham Dang C, Gautheron E, Guilley J, Fernandez M, Waast D, et al. (2005) The value of adding sciatic block to continuous femoral block for analgesia after total knee replacement. Reg Anesth Pain Med 30: 128-133.
-
Hebl JR, Kopp SL, Ali MH, Horlocker TT, Dilger JA, et al (2005) A comprehensive anesthesia protocol that emphasizes peripheral nerve blockade for total knee and total hip arthroplasty. J Bone Joint Surg Am 87: 63-70.
-
Auroy Y, Benhamou D, Bargues L, Ecoffey C, Falissard B, et al. (2002) Major complications of regional anesthesia in France: The SOS Regional Anesthesia Hotline Service. Anesthesiology 97: 1274-1280.
-
Teng Y, Jiang J, Chen S, Zhao L, Cui Z, et al. (2014) Periarticular multimodal drug injection in total knee arthroplasty. Knee Surg Sports Traumatol Arthrosc 22: 1949-1957.
-
Essving P, Axelsson K, Kjellberg J, Wallgren O, Gupta A, et al. (2010) Reduced morphine consumption and pain intensity with local infiltration analgesia (LIA) following total knee arthroplasty a randomized double-blind study involving 48 patients. Acta orthop 81: 354-360.
-
Fu P, Wu Y, Wu H, Li X, Qian Q, et al. (2009) Efficacy of intra-articular cocktail analgesic injection in total knee arthroplasty - a randomized controlled trial. Knee 16: 280-284.
-
Joo JH, Park JW, Kim JS, Kim YH (2011) Is intra-articular multimodal drug injection effective in pain management after total knee arthroplasty? A randomized, double-blinded prospective study. J Arthroplast 26: 1095-1099.
-
Koh IJ, Kang YG, Chang CB, Do SH, Seong SC, et al. (2012) Does periarticular injection have additional pain relieving effects during contemporary multimodal pain control protocols for TKA? A randomised, controlled study. Knee 19: 253-259.
-
Koh IJ, Kang YG, Chang CB, Kwon SK, Seo ES, et al. (2010) Additional pain relieving effect of intraoperative periarticular injections after simultaneous bilateral TKA: a randomized, controlled study. Knee Surg Sports Traumatol Arthrosc 18: 916-922.
-
Mullaji A, Kanna R, Shetty GM, Chavda V, Singh DP (2010) Efficacy of periarticular injection of bupivacaine, fentanyl, and methylprednisolone in total knee arthroplasty:a prospective, randomized trial. J Arthroplasty 25: 851-857.
-
Qian WW, Weng XS, Fei Q, Li Y, Zhai JL, et al. (2010) Application study of periarticular multimodal drug injection in total knee arthroplasty. Zhonghua Yi Xue Za Zhi 90: 2593-2596.