International Journal of Surgery Research and Practice
Vascular Complications after Adult to Adult Living Donor Liver Transplantation: Single Center Experience
Emad Hamdy Gad1*,Ahmed Elshawadfy Sherif1, Hosam Solyman1, Mohammed Elwarraky2, Khaled Abou Elela1, Tarek Ibraheem1 and Koichi Tanaka3
1Hepatobiliary and liver transplantation, National Liver Institute, Menoufiya University, Shebein Elkoum, Egypt
2Radiology, National Liver Institute, Menoufiya University, Shebein Elkoum, Egypt
3Liver transplantation, Kobe International Frontier Medical Center, Kobe City, Japan
*Corresponding author: Emad Hamdy Gad, Hepatobiliary and liver transplantation, National Liver Institute, Menoufiya University, Shebein Elkoum, Egypt Tel: 02-0100-3031128, Email: Emadgadsalemaa@yahoo.com
Int J Surg Res Pract, IJSRP-1-005, (Volume 1, Issue 1), Research Article; ISSN: 2378-3397
Received: August 24, 2014 | Accepted: September 23, 2014 | Published: September 25, 2014
Citation: Gad EH, Sherif AE, Solyman H, Elwarraky M, AbouElela K, et al. (2014) Vascular Complications after Adult to Adult Living Donor Liver Transplantation: Single Center Experience. Int J Surg Res Pract 1:005. 10.23937/2378-3397/1410005
Copyright: ©2014 Gad EH, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
List of Abbreviations
A-ALDLT: Adult to adult living donor liver transplantation
ALT: Alanine transaminase
AST: Aspartate transaminase
BCS: Budd chiari syndrome
BMI: Body Mass Index
CNIs: CalciNeurin Inhibitors
CNS: Central nervous system
CSA: CycloSporine
CTA: Computed tomography angiography
CUSA: Cavitron ultrasonic surgical aspirator
DM: Diabetes mellitus
ERCP: Endoscopic retrograde cholangio pancreatography
FK or FK-506 Tacrolimus
GDA: Gastroduodenal artery
GIT: Gastrointestinal tract
GRWR: Graft Recipient Weight Ratio
HAS: Hepatic artery stenosis
HAT: Hepatic artery thrombosis
HBV: Hepatitis B virus
HCC: HepatoCellular Carcinoma
HCV: Hepatitis C virus
HPB: HepatoPancreatoBiliary
HTK: Hydroxy tryptophan ketoglutarate
HTN: Hypertension
HVT: Hepatic vein thrombosis
IRB: Institutional review board
IRHV: Inferior right hepatic vein
IVC: Inferior vena cava
LDLT : Living donor liver transplantation
LFT: Liver function tests
LHV: Left hepatic vein
LLG: Left lobe graft
LRDT: Living related donor transplantation
LT : Liver Transplantation
MELD: Model for End stage Liver Disease
MHV: Middle hepatic vein
MMF: Mycophenolate MoFetil
MRA: Magnetic resonance angiography
MRCP: Magnetic resonance cholangio pancreatography
NLI: National Liver Institute
OLT: Orthotopic liver transplantation
PBC: Primary biliary cirrhosis
POD: Post operative day
PSC: Primary sclerosing cholangitis
PVS: Portal vein stenosis
RLG: Right liver graft
SFSG: Small for size graft
SFSS: Small for size syndrome
SRL: SiRoLomus
PVT: Portal vein thrombosis
VC: Vascular complications
Abstract
Objectives: Vascular complications (VC) after living donor liver transplantation (LDLT) are one of the most feared problems that result in graft and patient loss. The aim of this study was to assess the incidence, treatment, and outcome of vascular complications of patients after adult to adult LDLT in a single Egyptian center.
Methods: Between April 2003 and February 2013, we performed 167 adult to adult LDLT. The overall male/female ratio was 147/20. We retrospectively identified and analyzed vascular complications in recipients.
Results: The overall incidence of VC was 21.6% (n=36) and individually HA thrombosis 3.6% (n=6), HA stenosis 10.2% (n=17), HA injury 0.6% (n=1), PV thrombosis 4.8% (n =8), PV stenosis 0.6% (n=1), HV thrombosis 0.6% (n=1), HV stenosis 0.6% (n=1) and IVC tear 0.6% (n=1). 12/36 (33.3%) patients died as a direct result of the vascular complications. The overall 1-, 3-, 5- and 7-year survival rates in our series were 61.1%, 56.9%, 56.3% and 55.1% respectively. The 1-, 3-, 5- and 7-year survival rates in patients with and without VC were 41.7%, 36.1%, 36.1% and 36.1% and 66.4%, 62.6%, 61.8% and 60.3% respectively.
Conclusions: VC were associated with significant reduction in survival in our study, so prevention and proper treatment of them is required to achieve better survival among patients undergoing LDLT.
Keywords
Living Donor Liver Transplantation, Vascular complications, Graft survival
Introduction
Liver transplantation (LT) has become the treatment of choice for patients with end-stage liver disease [1]. However vascular problems such as thrombosis and stenosis of the hepatic artery (HA), portal vein (PV) and hepatic vein (HV) are among the most serious complications reported after LT and are more frequently seen among recipients of LDLT. These complications can lead to increased morbidity, graft loss, and patient death [2]. Reported rates can be as high as 25%, 16%, and 11% for HAT, PVT, and HAS, respectively [3]. Early diagnosis and appropriate management of VC result in longer survival. Close surveillance of all vascular anastomoses using Duplex ultrasonography facilitates early detection and treatment of these complications before irreversible graft failure. Treatment options usually include surgical revascularization, percutaneous thrombolysis, percutaneous angioplasty, retransplantation, or less commonly, a conservative approach [4].
Patients and Methods
After approval of Institutional Review Board (IRB) and obtaining written informed consents from both donors and recipients, we retrospectively analyzed VC related to the HA, PV and HV in liver transplanted adult patients in the department of HPB surgery, NLI, university of Menoufiya, Menoufiya, Egypt, in the period from April 2003 to November 2013 with mean follow up period of 24.7�25.5m, range: (0-120 m). During the period, about 167 adult patients underwent LDLT. The following data were studied:
A- Preoperative Variables
Donor's age, gender, body mass index (BMI), donor to recipient relation, recipient age, gender, blood group matching, primary disease, Child Pugh and MELD scores, co- morbidity (DM, HTN) and previous vascular thromboses (HA, PV and HV).
B- Intraoperative Variables
Duration of the operation per hours, actual graft weight, actual graft recipient weight ratio (GRWR), number and time of arterial, portal and hepatic venous reconstruction, cold and worm ischemia times per minute, blood and plasma transfusion per unit.
Surgical Techniques: HV anastomoses were performed using various surgical techniques to match graft hepatic veins to recipient inferior vena cava(IVC) optimally depending on the anatomic variation, furthermore, tying both anastomoseses were done end-to-end using continuous 5/0 prolene suture using 3 loupe magnification (I.e. The hepatic venous drainage pathways of a right liver graft (RLG) consist of the right hepatic vein (RHV),without or with middle hepatic vein (MHV) and in some cases with inferior RHV (IRHV) and in other cases with segment V and or segment VIII veins. On the other hand, the hepatic venous drainage pathways of a LT lobe graft(LLG) consist of the left hepatic vein (LHV) with middle hepatic vein (MHV) in a single stump or separately. Complete reconstruction of these venous outflow pathways is essential to prevent hepatic venous congestion for a fully functional RLG or LLG.
The PV anastomosis was then performed with the routine use of about 1 cm growth factor while tying both anastomoseses were done end-to-end using continuous 6/0 prolene suture using 3 loupe magnification. Moreover, in cases with preoperative PVT, thrombectomy was done with or without using vein graft for anastomosis.
The HA anastomosis performed using 6.5 loupe magnification with interrupted 8/0 monofilament Prolene with double needles, which facilitates secure sutures with good intima adaptation. Before performing arterial reconstruction, it is necessary to confirm adequate blood flow by releasing the clamp on the recipient hepatic artery. Both the arteries (graft side and recipient side) were fixed in a microsurgical double-clamp type A-II (Ikuta Microsurgery Instruments, Mizuho, Tokyo, Japan), which had 2 bulldog clamps fitted to a sliding bar. First, the angle sutures were placed at both the edges and tied with 8-0 monofilament (Prolene) sutures. The 8-0 Prolene suture with double needles and a short thread (5 cm) was specially devised for this technique (Bearen WT07F08N15-5; Bear Medical Corporation, Tokyo, Japan). Subsequently, the other sutures on the anterior side between the angle sutures were placed and tied. After completion of the anterior wall sutures, posterior wall sutures were performed in the same manner by turning the double clamps. Finally, the double clamps were removed and arterial reperfusion was performed.
C- Postoperative Variables
A. Hospital stay ( per days)
B. Based on our institutional policy
Immunosuppression protocol: the standard is combined 3 drugs calcineurin inhibitors (CNIs), steroids and mycophenolate mofetil (MMF). The initial methylprednisolone dose is 500 mg intraoperatively with a brief taper of prednisone from 240 to 40 mg/d over 6 days followed by 5�20 mg/d maintenance treatment, with complete withdrawal at the end of 3rd month post LDLT. Cyclosporine (CsA) was used when neurotoxicty or nephrotoxicity developed with Tacrolimus. When CNIs are contraindicated or their side effects halt their use, sirolimus (SRL) was given at an initial dose of 3 mg/m2 and adjusted over time to achieve blood trough levels of approximately 5�8 ng/mL. Biopsy-proven acute rejection episodes were treated with steroid pulses (IV methylprednisolone 200 to 500 mg/d for 3 days), which were tapered over several days to the baseline dose [5-8].
Anticoagulants: Heparin infusion up to 180-200units/kg/day adjusted with reference to the activated clotting time [target levels, 180�200 seconds] and/or the activated partial thromboplastin time [target levels, 50�70 seconds]). But when thrombocytopenia occurred, heparin was shifted to clexan 20mg/12h, then at POD8 dipyridamole was given at a dose of (4 mg/kg/d) for three months as protocol [9-11].
Doppler ultrasonography (For measuring HA resistive index, PV and HV velocities in the liver graft) was routinely performed just after anastomoses and after abdominal closure to ensure vascular patency and twice a day until POD7, and once per day until the patients were discharged from the hospital. Then follow up was done monthly during the 1st 6 months, then every 3 months till the end of 1st year, then every 6 months till the end of follow up. While LFTs (mainly AST, ALT, bilirubin) were done once daily till discharge and then monthly during the 1st 6 months, then every 3 months till the end of the 1st year, then every 6 months till the end of follow up. If abnormal serum liver function tests results were obtained, we performed doppler ultrasonography as soon as possible.
Diagnosis of VC was suspected when the liver function test results became abnormal or when doppler ultrasound revealed poor blood flow (change of resistive index of HA (N.B. The resistive index was determined as: (diastolic maximum flow velocity - systolic maximum flow velocity)/systolic maximum flow velocity) that increases with stenosis) and change of blood flow velocity of HV and PV ( the velocity increases after stenotic segments), or no blood flow within the hepatic vessels. The VC was confirmed on either computerized tomography angiography (CTA), magnetic resonance angiography (MRA), or formal conventional angiography as necessary. VC was defined as early VC when occurring within the first 30 days after LT and as late VC if diagnosed after 30 days after LT.
Treatment of VC: For vascular thrombosis, prompt surgical thrombectomy and reconstruction with end to end anastomosis were always our first choice in early cases with post-operative therapeutic anticoagulant therapy, while percutaneous angiographic catheter thrombectomy and thombolysis using intravascular urokinase with or without stenting were used in late cases. If endovascular treatment was not possible or efficacious, we converted the procedure to open surgery; however, medical treatment using therapeutic anticoagulant therapy was the choice in some cases, however, for vascular stenosis, percutaneous angiographic dilatation with or without stenting was the 1st choice in early and late cases. If endovascular treatment was not possible or efficacious, we converted the procedure to open surgery; however, medical treatment using therapeutic anticoagulant therapy was the choice in some cases [12-14,4].
Statistical analysis: All data were tabulated and processed with SPSS software (Statistical Product and Service Solutions, version 21, SSPS Inc, Chicago, IL, USA) and Windows XP (Microsoft Corporation, Redmond, Washington, USA). Qualitative data were expressed in frequency and percentage and analyzed with the chi-square test. Quantitative data were expressed as the mean and standard deviation or median and range and were compared with the t test. Comparison between patients with and without VC was done using univariate analysis The Kaplan�Meier method was applied for survival analysis and compared using log-rank tests. In all tests, a P value of < 0.05 was considered significant.
Results
Characters of patients and their donors: They were classified as 147 (88%) males, and 20 (12%) females. Their mean age was 46.3�8.2. Their donors were classified as 114 (68.3%) males and 53 (31.7%) females, their mean age was 26.9�6.5. Sixty one (36.5%) of patients had co morbidity, in the form of Hypertension, DM, cardiac diseases and morbid obesity. Concerning vascular anastomoses, single HV, PV, and HA anastomoses were found in 115 (68.9%), 155 (92.8%) and 160 (95.8%) of patients respectively, while multiple anastomoses of the HV, PV and HA were 52 (31.1%), 12(7.2%) and 7 (4.2%) respectively. On the other hand, pre operative PVT was found in 19 (11.4%) of patients. The mean cold, warm ischemia and arterial anastomosis times were 74.9�52.1(min), 52.1�16.05(min) and 67.8�34.8(min) respectively. The mean intraoperative blood and plasma transfusions were 7.03�7.4 and 8.3�8.9 units respectively (Table 1).
![]()
Table 1: Characters of patients and their donors
View Table 1
Single anastomosis means one anastomosis only regarding HA, PV, HV, while multiple anastomoses mean more than one anastomosis as in cases of trifurcated PV, double HA of the graft, anastomosis of the RT, middle HV, segment V, egment VIII veins, and RT inferior HV( 2 or more of them).
Indications of LT: The most frequent indications were HCV (54.5%) followed by HCC (34.1%). (table 2)
![]()
Table 2: Indications of LT
View Table 2
Vascular complications and their management: The overall incidence of vascular complications was 36(21.6%), while Early VC (before 1 month) and Late VC ( After 1 month) were 9.6% and 12% respectively. These complications were classified into HA, PV, HV problems and other vascular complications.
The incidence of HA problems was 24 (14.4%), in the form of HA stenosis 17 (10.2%) (including 3 early and 14 late), HAT 6 (3.6%) ( N.B one of them had aneurysm) (including 3 early and 3 late) and early HA injury 1(0.6%).
Twelve over seventeen of patients with HA stenosis were successfully managed, where 10 of them underwent late angiographic dilatation and stenting, one of them underwent late angiographic dilation and the last patients underwent late anticoagulant therapy. On the other hand 5/17 of HA stenosis cases were unsuccessfully managed where 3 of them underwent early anticoagulant therapy, one of them underwent early angiographic dilatation and coiling of GDA and the last one underwent late angiographic dilatation and stenting, however, 4 of those 5 patients died.
Two of six patients with HAT were managed successfully, one of them underwent early surgical reconstruction and the other had medical treatment. Conversly, the other 4 patients with HAT underwent unsuccesfull treatment where 2 of them underwent angiographic thrombolytic therapy and stenting ( N.B one of them had aneurysm and underwent coiling), one of them underwent early surgical reconstruction and the last one underwent early medical treatment, however, all those 4 patients died. The only patient with early HA injury underwent unsuccessful surgical reconstruction and died.
The incidence of PV problems was 9 (5.4%) that was divided into late PV stenosis 1(0.6%) and PVT 8(4.8%)( including 6 early and 2 late). The only case with PV stenosis underwent unsuccessful angiographic dilatation and stenting and died, however, 1/8 of patients with PVT underwent successful late anticoagulant therapy. On the other hand, 7/8 of them underwent unsuccessful management, 4 of them underwent early surgical thrombectomy, and 3 of them underwent anticoagulant therapy (2 early and one late). However all those 7 patients died.
The incidence of HV problems was 2(1.2%) and classified into early HV stenosis 1(0.6%) and early HVT 1 (0.6%). The only case with HV stenosis underwent unsuccessful early anticoagulant therapy and died. Conversely, the other patient with HVT underwent successful early anticoagulant therapy.
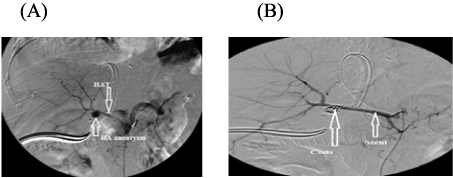 Figure 1: (A) A patient with HAT and aneurysm. (B) The patient underwent coiling of aneurysm and stenting of HAT.
View Figure 1
Figure 1: (A) A patient with HAT and aneurysm. (B) The patient underwent coiling of aneurysm and stenting of HAT.
View Figure 1
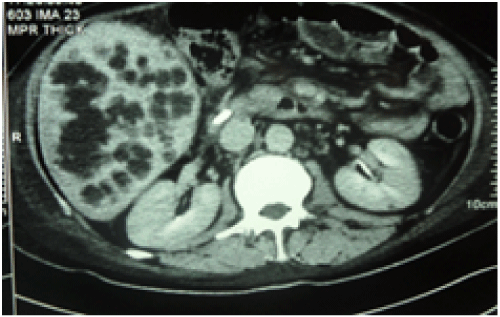 Figure 2: A patient with HAT and multiple hepatic abscesses managed with stenting of HAT and pigtail and antibiotics for abscesses
View Figure 2
Figure 2: A patient with HAT and multiple hepatic abscesses managed with stenting of HAT and pigtail and antibiotics for abscesses
View Figure 2
Lastly the patient with late IVC tear died intraoperative during operation for HCC recurrence (Figures 1, Figures 2, Figures 3, Table 3).
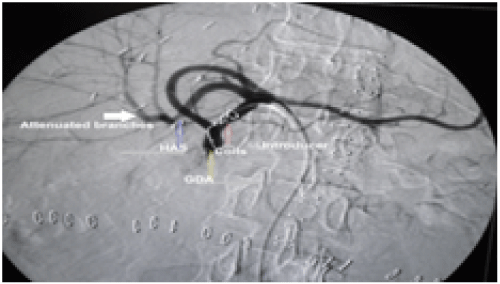 Figure 3: A patient with HAS underwent coiling of GDA
View Figure 3
Figure 3: A patient with HAS underwent coiling of GDA
View Figure 3
![]()
Table 3: Vascular complications and their management
View Table 3
Predictors of VC: Upon univariate analysis, multiple PV anastomosis was significant predictor of VC, while, compatible blood group matching between donor and recipient, multiple HV anastomosis and preoperative PVT affected VC with a trend towards significance. Conversely, Co-morbidity, Portal HTN, Bl. Group matching, Regimen of immunosuppresion including sirolomus, cold ischemia, worm ischemia, arterial anastomosis times, and amount of blood and plasma transfusion units had no effect on VC. (Table 4 and Table 5)Upon multivariate analysis by binary logistic regression test, there was no independent predictor of VC (Table 6).
![]()
Table 4: Univariate analysis of predictors of VC
View Table 4
![]()
Table 5: Univariate analysis of predictors of VC
View Table 5
5- Outcome of patients: The overall mortality in our patients was 75/167 (44.9%). However, mortalities in patients with VC and directly related to VC were 23/36 (63.8%) and 12/36 (33.3%) respectively (N.B. The five cases who died due to PVT did not have any other major complication and PVT lead to graft failure and death, furthermore, the three cases died due to HA stenosis had multiple hepatic abscesses and died from sepsis without any other major complications, similarly, the two cases with HAT died from sepsis without any other major complications, moreover, The patient died due to HA injury due to pigtail insertion to drain biloma, underwent unsuccessfull surgical reconstruction where recurrent bleeding occurred, then DIC, then death. Lastly, the patient with IVC tear died intraoperative due to massive bleeding from the tear, despite the exploration was due to HCC recurrence). While overall 1-, 3-, 5- and 7- year survival of our patients were 102 (61.1%), 95(56.9%), 94(56.3%) and 92(55.1%) respectively (Table 7).
![]()
Table 6: Multivariate analysis of predictors of VC ( Binary logistic regression test)
View Table 6
![]()
Table 7: Outcome of patients
View Table 7
6- VC and survival: Upon univariate analysis, VC was significant predictor of poor survival, while the effective management of them improved survival (Table 8, Figure 4).
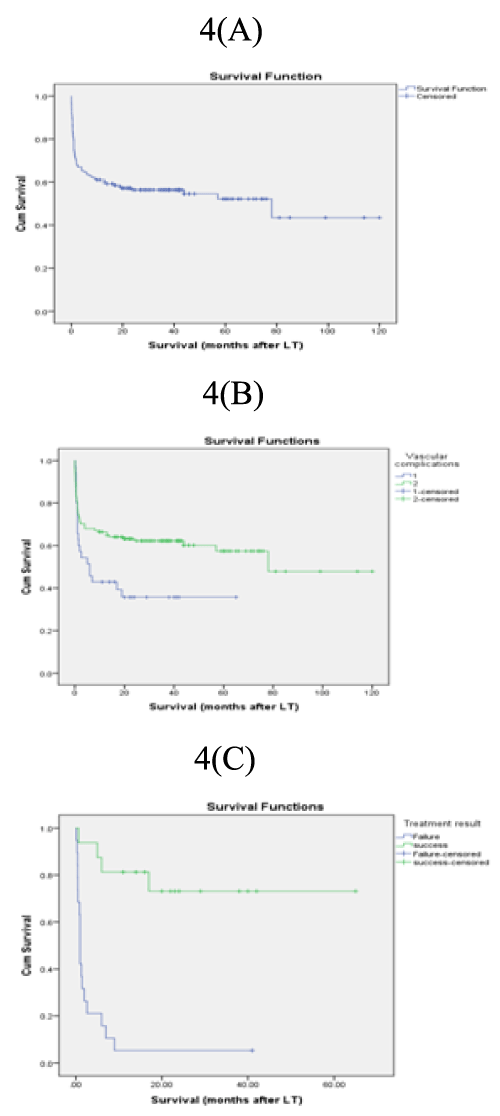
A: KM survival curve
B: VC and survival ( Log rank=0.01)
C: Effective treatment and survival (Log rank=0.00).
Figure 4: Kaplan-Meier survival curves
View Figure 4
![]()
Table 8: Univariate analysis of VC and survival
View Table 8
Discussion
Vascular complications remain a major cause of morbidity and mortality after orthotopic liver transplantation (OLT) (4). The risk of them is relatively high in LDLT when compared with whole graft transplantation (2). The overall incidence of vascular complications in our study was 36(21.6%) however it was 9/69(13%) and 3/66(4.5%) in [4] and [3] studies respectively.
Various HA complications, including thrombosis or stenosis, pseudoaneurysm, dissection, and arterial steal syndrome, may occur after LDLT [15], they result in increased graft loss, mortality and morbidity of the LDLT recipients [16,13]. The overall arterial complication rate was 21.5% and 24 (14.4%) in [16] and our studies respectively.
HAS was 11% and 17 (10.2%) in [17] and our studies respectively. It was successfully treated by balloon angioplasty in [3] study. Similarly, angiography was the main treatment option of our patients with HAS where 10/17 of them were successfully managed with angiographic dilatation and stenting.
HAT is a serious problem; It is associated with increased morbidity, graft loss, and mortality. Its incidence after LDLT varies from 4% to 25% [2,4,16-20]. However it was (3.6%) in our study.
The treatment options for HA thrombosis include urgent revascularization, either with the native HA following thrombectomy or with HA alternatives [12-14]. Other options include the use of intraarterial thrombolytics as urokinase [16,21], conservative treatment (In the absence of hepatic failure) [12] and lastly retransplantation [13]. On the other hand, two of our six patients with HAT underwent surgical reconstruction where one succeeded and the other failed, and another 2 of them underwent angiographic thrombolytic therapy but failed and the last 2 of them underwent medical conservative treatment where one succeeded but the other failed.
The postoperative portal venous complications (stenosis and thrombosis) following liver transplantation have been documented. They are more common with living related donor transplants (LRDT) [22]. They lead to graft failure [23]. So, Prompt detection and management are required to reduce the mortality and graft loss [24].
PV complications Reported rates can be as high as 16% after LDLT (17), It was 7.3% in [4] study. However it was 5.4% in our study. They can be managed through surgical treatments such as thrombectomy, surgical revision, and retransplantation or through angiographic interventions (Thrombolytics for PVT or balloon angioplasty for PVS) [4,23,25]. However, in our work, The only case with PV stenosis underwent unsuccessfull angiographic dilatation and stenting, however, 1/8 of patients with PVT underwent successful late anticoagulant therapy. On the other hand, 4/8 of them underwent unsuccessfull early surgical thrombectomy and 3/8 of them underwent unsuccessfull anticoagulant therapy.
Vascular outflow complications (Thrombosis or stenosis) occur in a proportion of patients after LDLT, often leading to serious graft dysfunction [26]. The overall incidences are around 1% [27] study. The incidence of HVS was 1.5% in [4] study and 0.6% in our study. On the other hand HVT rate was 1.5% in [3] study, and 0.6% in our work. Most outflow complications can be corrected by percutaneous angiographic measures [4,28]. On the other hand, the only our case with HV stenosis underwent unsuccessfull early anticoagulant therapy. Conversly, the other patient with HVT underwent successful early anticoagulant therapy.
The Long-term survival was statistically lower for recipients with vascular complications in [4] and [3] studies. Similarly, it was statistically lower for our recipients with vascular complications.
Conclusion
VC were associated with significant reduction in survival in our study, so prevention and proper treatment of them is required to achieve better survival among patients undergoing LDLT.
Declaration
This study was approved by our institution ethical committee. Forms of support received by each author for this study included good selection of cases, instructive supervision, continuous guidance, valuable suggestions and good instructions. No grant or other financial support was received for this study.
References
-
Karakayali H, Sevmis S, Boyvat F, Aktas S, Ozcay F, et al. (2011) Diagnosis and treatment of late-onset portal vein stenosis after pediatric living-donor liver transplantation. Transplant Proc 43: 601-604.
-
Duffy JP, Hong JC, Farmer DG, Ghobrial RM, Yersiz H, et al. (2009) Vascular complications of orthotopic liver transplantation: experience in more than 4,200 patients. J Am Coll Surg 208: 896-903.
-
Steinbr�ck K, Enne M, Fernandes R, Martinho JM, Balbi E, et al. (2011) Vascular complications after living donor liver transplantation: a Brazilian, single-center experience. Transplant Proc 43: 196-198.
-
Khalaf H (2010) Vascular complications after deceased and living donor liver transplantation: a single-center experience. Transplant Proc 42: 865-870.
-
Goldstein MJ, Salame E, Kapur S, Kinkhabwala M, LaPointe-Rudow D, et al. (2003) Analysis of failure in living donor liver transplantation: differential outcomes in children and adults. World J Surg 27: 356-364.
-
Li C, Wen TF, Yan LN, Li B, Yang JY, et al. (2011) Predictors of patient survival following living donor liver transplantation. Hepatobiliary Pancreat Dis Int 10: 248-253.
-
Ikegami T, Shirabe K, Yoshiya S, Yoshizumi T, Ninomiya M, et al. (2012) Bacterial sepsis after living donor liver transplantation: the impact of early enteral nutrition. J Am Coll Surg 214: 288-295.
-
Kim BS, Lee SG, Hwang S, Ahn CS, Kim KH, et al. (2012) Influence of Pretransplantation Bacterial and Fungal Culture Positivity on Outcome after Living Donor Liver Transplantation. Transplant Proc 41: 250-252.
-
Taniai N, Onda M, Tajiri T, Akimaru K, Yoshida H, et al. (2002) Anticoagulant therapy in living-related liver transplantation. Transplant Proc 34: 2788-2790.
-
Francoz C, Valla D, Durand F (2012) Portal vein thrombosis, cirrhosis, and liver transplantation. J Hepatol 57: 203-212.
-
Kaneko J, Sugawara Y, Tamura S, Togashi J, Matsui Y, et al. (2008) Heparin-induced thrombocytopenia after liver transplantation. Transplant Proc 40: 1518-1521.
-
Tian MG, Tso WK, Lo CM, Liu CL, Fan ST (2004) Treatment of hepatic artery thrombosis after orthotopic liver transplantation. Asian J Surg 27: 213-217.
-
Wang CC, Lin TS, Chen CL, Concejero AM, Iyer SG, et al. (2008) Arterial reconstruction in hepatic artery occlusions in adult living donor liver transplantation using gastric vessels. Surgery 143: 686-690.
-
Scarinci A, Sainz-Barriga M, Berrevoet F, van den Bossche B, Colle I, et al. (2010) Early Arterial Revascularization After Hepatic Artery Thrombosis May Avoid Graft Loss and Improve Outcomes in Adult Liver Transplantation. Transplantation Proc 42: 4403-4408.
-
Kim SY, Kim KW, Kim MJ, Shin YM, Lee MG, et al. (2007) Multidetector row CT of various hepatic artery complications after living donor liver transplantation. Abdom Imaging 32: 635-643.
-
Jeon GS, Won JH, Wang HJ, Kim BW, Lee BM (2008) Endovascular treatment of acute arterial complications after living-donor liver transplantation. Clin Radiol 63: 1099-1105.
-
Kim BS1, Kim TK, Jung DJ, Kim JH, Bae IY, et al. (2003) Vascular complications after living related liver transplantation: evaluation with gadolinium enhanced three-dimensional MR angiography. Am J Roentgenol 181: 467-470.
-
Yang Y, Yan LN, Zhao JC, Ma YK, Huang B, et al. (2010) Microsurgical reconstruction of hepatic artery in A-A LDLT: 124 consecutive cases without HAT. World J Gastroenterol 16: 2682-2688.
-
Nikeghbalian S, Kazemi K, Davari HR, Salahi H, Bahador A, et al. (2007) Early hepatic artery thrombosis after liver transplantation: diagnosis and treatment. Transplant Proc 39: 1195-1196.
-
Coelho GR, Leitao AS Jr, Cavalcante FP, Brasil IR, Cesar-Borges G, et al. (2008) Continuous versus interrupted suture for hepatic artery anastomosis in liver transplantation: Differences in the incidence of hepatic artery thrombosis. Transplant Proc 40: 3545-3550.
-
Kim BW, Won JH, Lee BM, Ko BH, Wang HJ, et al. (2006) Intraarterial thrombolytic treatment for hepatic artery thrombosis immediately after living donor liver transplantation. Transplant Proc 38: 3128-3131.
-
Buell JF, Funaki B, Cronin DC, Yoshida A, Perlman MK, et al. (2002) Long-term venous complications after full-size and segmental pediatric liver transplantation. Ann Surg 236: 658-666.
-
Woo DH, Laberge JM, Gordon RL, Wilson MW, Kerlan RK Jr (2007) Management of portal venous complications after liver transplantation. Tech Vasc Interv Radiol 10: 233-239.
-
Corno V, Torri E, Bertani A, Guizzetti M, Lucianetti A, et al. (2005) Early portal vein thrombosis after pediatric split liver transplantation with left lateral segment graft. Transplant Proc 37: 1141-1142.
-
Vignali C, Cioni R, Petruzzi P, Cicorelli A, Bargellini I, et al. (2004) Role of interventional radiology in the management of vascular complications after liver transplantation. Transplant Proc 36: 552-554.
-
Hwang S, Lee SG, Ahn CS, Moon DB, Kim KH, et al. (2010) Morphometric and simulation analyses of right hepatic vein reconstruction in adult living donor liver transplantation using right lobe grafts. Liver Transpl 16: 639-648.
-
Shirouzu Y, Kasahara M, Morioka D, Sakamoto S, Taira K, et al. (2006) Vascular reconstruction and complications in living donor liver transplantation in infants weighing less than 6 kilograms: the Kyoto experience. Liver Transpl 12: 1224-1232.
-
Ko GY, Sung KB, Yoon HK, Kim KR, Kim JH, et al. (2008) Early posttransplant hepatic venous outflow obstruction: Long-term efficacy of primary stent placement. Liver Transpl 14: 1505-1511.





