International Journal of Radiology and Imaging Technology
Low Dose CT of the Brain in the Follow-up of Intracranial Hemorrhage
Stella Blasel1*, Luisa Huck1, Juergen Konczalla2, Stephanie Lescher1, Hanns Ackermann3, Joachim Berkefeld1 and Marlies Wagner1
1Institute of Neuroradiology, Goethe-University Hospital Frankfurt, 60528 Frankfurt, Germany
2Department of Neurosurgery, Goethe-University Hospital Frankfurt, 60528 Frankfurt, Germany
3Institute of Biostatistics and Mathematical Modelling, Goethe University, 60590 Frankfurt, Germany
*Corresponding author:
Stella Blasel, Institute of Neuroradiology, University of Frankfurt, Schleusenweg 2-16, 60528 Frankfurt, Germany, Tel: + 49/69 63015462, Fax: + 49/69 63017176, E-mail: Stella.Blasel@kgu.de
Int J Radiol Imaging Technol, IJRIT-2-015, (Volume 2, Issue 2), Original Article
Received: April 09, 2016; Accepted: July 14, 2016; Published: July 16, 2016
Citation: Blasel S, Huck L, Konczalla J, Lescher S, Ackermann H, et al. (2016) Low Dose CT of the Brain in the Follow-up of Intracranial Hemorrhage. Int J Radiol Imaging Technol 2:015.
Copyright: © 2016 Blasel S, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Objectives:Adult patients with intracranial hemorrhage (ICH) are often young but receive serial cranial computer tomography (CCT). To lower their cumulative radiation dose risks, we evaluated applicability of low dose cranial CT (LD-CCT) in the follow-up of ICH patients compared to an initial standard dose cranial CT (SD-CCT).
Methods:53 ICH patients underwent initial SD-CCT (350 mAs/120 kV) and follow-up LD-CCT (either 220 or 240 mAs/120 kV). Iterative reconstruction used for both dose levels was increased by one factor for LD-CCT. Image quality parameters [visual impression (VI), gray matter (GM)/white matter (WM) differentiation, edema, ICH, cerebrospinal fluid spaces (CSF), postoperative changes, basal cisterns] were retrospectively assessed independently by an experienced neuroradiologist and neurosurgeon. Significance level was set at p < 0.05.
Results:Both raters accredited both LD-CCT protocols (220 and 240 mAs) to be equivalent to SD-CCT for display of ICH, CSF and basal cisterns. The SD-CCT was rated significantly better (p < 0.01) concerning VI, GM/WM differentiation and imaging of edema by the neuroradiologist only.
Conclusions:IR based LD-CCT with a dose reduction up to almost 40% are applicable for ICH follow-up concerning findings requiring immediate therapeutic intervention. However, LD-CCT quality deficits regarding GM/WM differentiation and hypodense lesions exclude stroke assessment.
Keywords
Head CT, Iterative reconstruction, Intracranial hemorrhage, Dose reduction
Abbreviations
ICH: Intracranial Hemorrhage; CCT: Cranial Computer Tomography; LD-CCT: Low Dose Cranial CT; SD-CCT: Standard Dose Cranial CT; IR: Iterative Reconstruction; GM: Gray Matter; WM: White Matter; CSF: Cerebrospinal Fluid Spaces
Introduction
Unenhanced cranial CT (CCT) is the standard imaging method to assess intracranial hemorrhage (ICH) including intracerebral, sub- and epidural and subarachnoid hemorrhage [1-4]. Patients with ICH require several follow-up CCTs during their hospitalization for monitoring of hemorrhage, edema, and localization of foreign materials like ventricle shunt systems and assessment of ventricle size. However, radiation exposure side effects increase with the number of CT examinations [5]. Therefore, especially young ICH patients could strongly benefit from head CT dose reduction techniques to lower their cumulative radiation exposure.
In 2009, iterative reconstructions (IR) techniques were established to improve image quality as prerequisite to reduce CT radiation exposure. Although IR techniques were already known for the first CT scanners in the 1970s [6], they only became feasible throughout the technical progress with more powerful processors [7]. Today, IR based CCTs are more commonly used with the increasing installation of latest generation CT scanners [8-10]. The first pilot studies with IR based low dose cranial CCT (LD-CCT) and 10-68% dose reductions delivered equivalent or even better image qualities than standard dose cranial CCT (SD-CCT) with traditional filtered back projection (FBP) reconstructions concerning correct assessment of the diverse structures of neuroanatomy [9,11-19]. Dose reduction is inversely correlated with noise increase in multi slice CT [20]. However, as compared to the traditional FBP technique, IR techniques modify CT raw- or image data to separate image information and image noise. Subsequently, iterative processing loops are applied to the data to reduce noise while preserving spatial resolution. Afterwards, missing image data is replaced based on a calculation model [19,21,22]. It is important to understand that IR algorithms do not primarily reduce absorbed radiation dose in CT scanning. IR techniques rather improve image quality through noise reduction that in turn can be used to realize good image quality at reduced dose levels [23,24]. However, with increasing IR factor, more image data has to be replaced by calculation. Therefore, if the IR factor is chosen too high, alienation of the CT images might be the consequence.
However, well balanced IR techniques allow performance of CCT with decreased levels of ionizing radiation and preserved spatial resolution [23,24]. The routine CCT in patients with new focal neurological deficits, especially in stroke patients, requires high image quality to depict even subtle density differences. Therefore, a small IR factor should be chosen in those patients to avoid image alienation. In contrast, in patients with ICH a certain decrease of image quality might be acceptable in follow-up CCTs to decrease radiation exposure, because follow-up CCTs are performed to assess structures with greater density differences including ICH, cerebrospinal fluid (CSF) spaces, and foreign material. While performance of head CTs with reduced radiation doses in non-emergency conditions such as follow-up of hydrocephalus, ICH or post-operative evaluation has already been well established in the pediatric world [25-30], literature on similar LD-CCT indications in adult patients is sparse [8,9].
The aim of our study was therefore to find out if further dose reduction compensated by an increased IR factor is feasible in follow-up CCTs of ICH patients. To account for different demands on image quality, all images were read independently by an experienced neuroradiologist and by an experienced neurosurgeon.
Materials and Methods
Study population
We retrospectively selected adult patients with ICH, who received an SD-CCT (SD as defined below) at hospital admission and at least one follow-up LD-CCT (LD as defined below) between 01/2014 and 06/2014. All CTs were based on clinical indication and retrospectively reviewed for this study. The follow-up CT of each patient was performed with LD-CCT protocol as described below, if one of the following indications was present: (1) Follow up of size of hemorrhage, (2) Follow up of ventricle size or herniation, (3) Postsurgical control of foreign material (ventricle shunt systems, pressure sensor, craniectomy/craniotomy). LD-CCT was not performed in patients with new focal neurological deficits or in patients after coiling or clipping of aneurysms as the first control.
We retrospectively analyzed 53 patients (27 male, 26 females, with a mean age of 61 ranging from 19-96 years). Hereof, 14 patients suffered from aneurysmal subarachnoid hemorrhage, 24 from atypically localized parenchymal hemorrhage, 8 from subdural hematoma, 4 from epidural hematoma, and 3 patients from basal ganglia hematoma.
Image acquisition and reconstruction parameters
All patients were scanned on a 64-multidetector CT scanner Ingenuity Core (Philips Healthcare, Cleveland, OH, US).
All patients received an initial SD-CCT in axial plane with a collimation of 16 × 0.625 mm, as can increment of 10 mm, a reconstruction slice thickness of 5 mm, a rotation time of 1 s, a tube current of 350 mAs, a tube voltage of 120 kV and an IR factor of 1 (algorithm "iDose" [31]).
For the follow-up CCT with reduced radiation dose (LD-CCT), we applied two different dose protocols: one group of patients (n = 31) received reduced dose protocols with 240 mAs and 120 kV, and the other group of patients (n = 22) 220 mAs and 120 kV. In both LD-CCT protocols, iDose level was 2. All reconstructions were performed with an UB reconstruction kernel in high resolution.
Image analysis
Image quality was assessed independently by an experienced neuroradiologist (M.W.; 11 years of experience) and an experienced neurosurgeon (J.K.; 8 years of experience). We established a simplified but thereby unambiguous 3-tier scoring system (1 = good, 2 = sufficient, 3 = insufficient) reviewing a pilot set of LD-CCT (n = 20) in advance to the actual image analysis [32]. For analysis, SD-CCT and LD-CCT images of all patients were randomly arranged and visually rated using a preset questionnaire with a 3-point scoring system for the following parameters: (1) subjective visual impression, (2) outer cerebrospinal fluid spaces, (3) gray matter and white matter differentiation, (4) size of the bleeding, (5) ventricle size, (6) size of edema, (7) postoperative changes (i.e. intracranial air, foreign material), and (8) basal cisterns.
Both raters were blinded to the dose protocol, patient information (name, sex and age) and acquisition date.
Signal-to-noise ratio (SNR) and contrast-to-noise ratio (CNR)
We further determined the signal-to-noise ratio (SNR) and the contrast-to-noise ratio (CNR).
For SD-CCT and LD-CCT signal analysis, regions of interest (ROI) were manually drawn in the gray matter (SNR GM) and in the white matter (SNR WM) of the superior frontal gyrus (L.H.) in each patient. The mean attenuation values of the gray matter and white matter ROIs were divided by their noise level (equal to their standard deviation) to calculate the SNR according to a previous report [8]. The CNR was defined according to the formula by Mullins et al. [33]: (mean GM-mean WM)/[(SD x GM)2+(SD x WM)2)0.5.
Dose estimates
For the estimation of radiation doses, we recorded the dose length product (DLP, in mGy x cm) and the weighted volume computed tomography dose index (CTDIvol in mGy). The effective dose (ED) was calculated as a product of DLP and the normalized value of effective dose per DLP for the head (0.0023 mSv, Gy-1 cm-1) [34].
Statistical analysis
Statistical analyses were performed using BIAS™ version 10.12 software (2014 Epsilon, Frankfurt, Germany) with a significance level of p < 0.05. Intraobserver differences of image quality ratings were assessed by the Mc Nemar test, and DLP and CTDIvol were tested with the Wilcoxon-matched pairs test.
Results
SD-CCT was performed at the day of admission in all patients and within the first three days after hemorrhage in 43 of the 53 patients. The mean time interval from the day of bleeding to the initial SD-CCT scan was 2.0 days with a range of 0-17 days. The follow-up low dose scan was performed with 220 mAs in 22 patients and with 240 mAs in 31 patients. LD-CCT followed SD-CCT after 0-19 days and 4.5 days later in average. The mean time interval of the day of the bleeding to the follow-up LD-CCT scan was 6.5 days ranging from 0-20 days.
Image quality
All images of both SD- and LD-CCT were rated good or sufficient for all parameters by both raters implying that all images were suitable for diagnostics.
The neuroradiologist rated three parameters to be significantly better in SD-CCT than in LD-CCT (consistently for both protocols) (Table 1), while the neurosurgeon did not find any significant differences between SD- and LD-CCT (Table 2). Beside the visual impression (220 mAs/240 mAs: p = 0.0005/p = 0.0005), the neuroradiologist rated gray matter and white matter differentiation (220 mAs/240 mAs: p = 0.0002/p = 0.001) and the size of the edema (220 mAs/240 mAs: p = 0.006/p = 0.004) to be significantly better assessable in SD-CCT than in LD-CCT. No differences were found for outer cerebrospinal fluid spaces, size of ICH, ventricle size, postoperative changes, and basal cisterns (Figure 1, Figure 2 and Table 1).
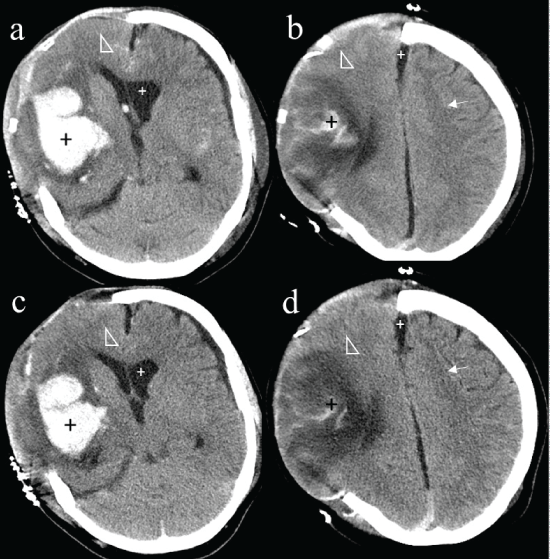
.
Figure 1: Upper row (a-b) initial head CT with the standard dose protocol (350 mAs), and follow-up CT (three days later) with the low dose protocol (220 mAs) in the lower row (c-d) of a patient with an intracerebral hemorrhage in the right-sided basal ganglia. The parenchymal and subarachnoid hyperdense blood (black plus signs, a vs. c) is equally well delineable in the low dose and standard dose CT respecting the normal course of blood resorption, whereas the surrounding hypodense edema (white arrowhead, a vs. c) is better visible in the initial CT in standard dose. Further, gray matter and white matter differentiation (white arrow, b vs. d) is better detectable in the standard dose CT compared with the low dose control. However, width of the inner and outer cerebrospinal fluid spaces are equally well depicted in the low dose and standard dose CT (small white plus sign, a-d).
View Figure 1
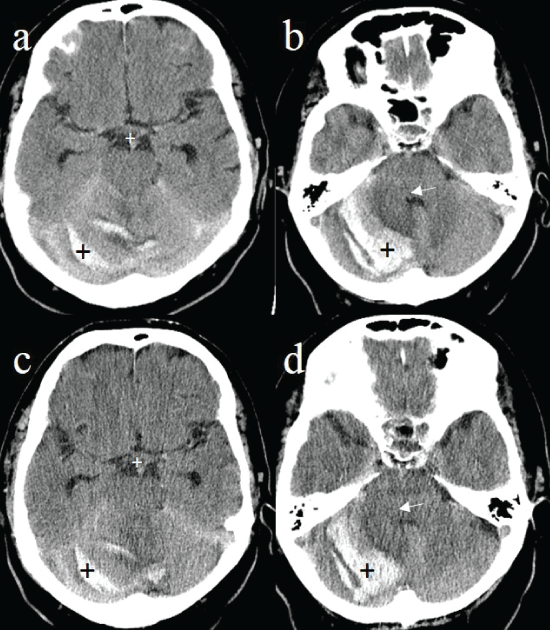
.
Figure 2: Upper row (a-b) initial head CT with the standard dose protocol (350 mAs) and follow-up scan (one day later) with the low dose CT protocol (240 mAs) in the lower row (c-d) of a patient with an atypical bleeding into the right cerebellum. The hyperdense intra- and extra-axial blood depositions in the cerebellar parenchyma and at the tentorium (black plus sign, a vs. c) are equally well visible in the low dose and in the standard dose CT respecting the normal time dependent course of blood resorption, whereas the surrounding hypodense edema (white arrow, b vs. d) is seen much better in the initial CT in standard dose. However, the growing mass effect of the edema with progressive compression of the fourth ventricle can be adequately seen in the follow-up low dose CT (white arrow, b vs. d) with preserved basal cisterns (white plus sign, a vs. c).
View Figure 2
SNR and CNR
SNR white matter, SNR gray matter and CNR were significantly lower in LD-CCT with 220 mAs compared to the SD-CCT with 350 mAs with p < 0.05, whereas the intrapatient comparison of the LD-CCT with 240 mAs to the SD-CCT with 350 mAs revealed no significant differences (Table 3).
![]()
Table 1: The neuroradiologist’s image quality parameters scores of low dose- and standard dose head CT protocols.
View Table 1
![]()
Table 2: The neurosurgeon’s image quality parameters scores of low dose- and standard dose head CT protocol.
View Table 2
Dose estimates
DLP, CDTIvol and ED were significantly lower in LD-CCT examinations (220 and 240 mAs) compared to the SD-CCT protocol (350 mAs) of each patient with p < 0.0001 (Table 4).
![]()
Table 3: Estimates of quantitative image quality parameters.
View Table 3
![]()
Table 4: Dose estimates for the low dose and standard dose CT protocols.
View Table 4
Discussion
Follow-up CCT in patients with ICH is indicated 4 to 8 hours after initial CCT to exclude rebleeding and reevaluate edema, in case of clinical deterioration (e.g. increasing intracranial pressure or rebleeding) or after surgical intervention (e.g. external ventricular drainage, decompressive craniectomy) [35,36]. Therefore clinical management of ICH requires repeated head CT-examinations controls in younger patients with increased risks for radiation injury.
Based on our results however, radiation exposure can be reduced up to almost 40% in LD-CCT controls of ICH patients using IR techniques. We could show that all findings that might have immediate therapeutic consequence are well assessable in follow-up of ICH patients using LD-CCT with the use of an increased IR factor ("iDose") compared to SD-CCT. LD-CCT was consistently rated to be an equivalent alternative to SD-CCT regarding the assessment of CSF spaces, size of the bleeding (independent of the type of ICH), and foreign material in the follow-up of ICH patients. As a consequence, LD-CCT might therefore be also applicable in other clinical indications in the future that also focus on the assessment of the CSF spaces or foreign material including patients prior to lumbar puncture, in the follow-up and monitoring of the ventricles width (e.g. in patients with intracranial neoplasms or suspected CSF leakage), or after implantation of ventricle shunt systems to control their localization and to exclude hemorrhage.
In order to increase the validity of our results, all images were rated by a neurosurgeon and a neuroradiologist independently, who might have different claims on image quality. It could be shown that the neuroradiologist rated the LD-CCT to be significantly inferior compared to SD-CCT regarding the gray matter and white matter differentiability, the assessment of edemas, and the visual impression. This finding however seems to exclude the standardized use of LD-CCT in every patient at the moment. In particular, patients with new focal neurological deficits, who might suffer from an acute stroke, encephalitis, small space occupying lesions, or other suspected lesions causing only subtle density differences should not be examined with LD-CCT as the primary imaging method [37-39]. Our findings are in line with the results of previous studies, where it could be shown that iterative reconstruction and reduced dose levels of 25% or more reduced the detection rate of low contrast cerebral lesions [40,41].
The reason for decreased visual impression, gray matter and white matter differentiability and assessment of edema in LD-CCT should lie primarily in its increased factor of iterative reconstruction to compensate decreased radiation dose. While our LD-CCT protocol is well balanced for high density differences, there is an imbalance of image quality and decreased radiation exposure for subtle density differences leading to impaired visualization. As described above in detail, the level of noise reductions in IR is inversely correlated with an altered "smoothened" or even "plastic" image appearance [11,23,24,42]. To account for this effect, the percentage of IR strength used must be adapted to the percentage of the reduced radiation dose in order to provide proper image quality [16].
Moreover, as each manufacturer uses different IR reconstruction algorithms, the noise properties of the resulting images are quite different [43]. As a consequence, image quality parameters such as sharpness and gray matter and white matter differentiation might also be dependent on the applied IR algorithm. Therefore, further large and multicenter investigations with different CT scanners and manufacturers are needed to learn more about the effects of iterative strength levels and algorithms on image quality parameters.
As expected and previously described [11,12,16,17], SNR and CNR were significantly lower in the LD-CCT protocol with 220 mAs compared to the SD-CCT protocol with 350 mAs, while only a trend to a decrease could be shown for the LD-CCT with 240 mAs.
To the best of our knowledge, this is the first study to evaluate diagnostic performance of IR based initial SD-CCT and IR based follow-up LD-CCT for the same adult patient with ICH. Recently, Corcuera-Solano et al. [9] compared objective image quality parameters such as SNR and CNR and subjective quality ratings of the granularity, gray matter and white matter differentiation, and the overall quality of LD- and SD-CCT for the same patient. However, they did not focus on the conspicuity of the hyperdense bleeding and its possibly space occupying side effects such as hypodense edemas or other shifting of the cerebrospinal fluid spaces, crucial for acute intervention. Further, we were first to compare image quality ratings from the neuroradiological and neurosurgical perspective with possibly different demands onto the CT examination.
As a limitation, time span from the bleeding to the SD-CCT and between SD-CCT and LD-CCT differed and reached up to 20 days. Therefore, density differences of bleeding and edema might have varied significantly. Further, the sample size of our retrospective study was quite low, and all scans were performed on the same scanner. It might be interesting to investigate the effects of different scanners and different manufactures in a multicenter study on a larger number of patients with uniform parameters of image acquisition/reconstruction and image quality assessment as a prerequisite for optimal comparability of the results.
In conclusion, low dose CCT scans with a dose reduction of up to almost 40% can be used in the routine follow-up of adult patients with intracranial hemorrhage. All relevant aspects including size of hemorrhage, mass effect, foreign material, and cerebrospinal fluid spaces can be assessed with good certainty using LD-CCT. As a consequence, LD-CCT might also be applicable in other indications focusing on those aspects and should be investigated in the future. However, image quality of LD-CCT is still not sufficient for the assessment of pathologies with subtle density differences. Therefore, it is not possible to use LD-CCT as the standard protocol for patients with focal neurological deficits, especially in stroke diagnostic, at the moment.
Acknowledgements
We would like to thank the native English speaking medical student Elizabeth Kanzow for language assistance in the preparation of the manuscript.
Ethical Statement
The local ethics committee approved this study. This is a retrospective evaluation of data acquired within the clinical framework, no extra CT scans were performed for study reasons. The work was part of a scientific cooperation between our institute and Philips Healthcare (Hamburg, Germany). The authors state that this work has not received any funding by the company.
References
-
Weisberg LA (1979) Computerized tomography in intracranial hemorrhage. Arch Neurol 36: 422-426.
-
Broderick JP, Adams HP, Barsan W, Feinberg W, Feldmann E, et al. (1999) Guidelines for the management of spontaneous intracerebral hemorrhage: A statement for healthcare professionals from a special writing group of the Stroke Council, American Heart Association. Stroke J Cereb Circ 30: 905-915.
-
Smith EE, Rosand J, Greenberg SM (2005) Hemorrhagic stroke. Neuroimaging Clin N Am 15: 259-272.
-
Mendelow AD, Teasdale G, Jennett B, Bryden J, Hessett C, et al. (1983) Risks of intracranial haematoma in head injured adults. Br Med J (Clin Res Ed) 287: 1173-1176.
-
Brenner DJ, Hall EJ (2007) Computed tomography-an increasing source of radiation exposure. N Engl J Med 357: 2277-2284.
-
Hounsfield GN (1973) Computerized transverse axial scanning (tomography). 1. Description of system. Br J Radiol 46: 1016-1022.
-
Kordolaimi SD, Argentos S, Pantos I, Kelekis NL, Efstathopoulos EP (2013) A New Era in Computed Tomographic Dose Optimization: The Impact of Iterative Reconstruction on Image Quality and Radiation Dose. J Comput Assist Tomogr 37: 924-931.
-
Bodelle B, Klein E, Naguib NNN, Bauer RW, Kerl JM, et al. (2014) Acute intracranial hemorrhage in CT: benefits of sinogram-affirmed iterative reconstruction techniques. AJNR Am J Neuroradiol 35: 445-449.
-
Corcuera-Solano I, Doshi AH, Noor A, Tanenbaum LN (2014) Repeated head CT in the neurosurgical intensive care unit: feasibility of sinogram-affirmed iterative reconstruction-based ultra-low-dose CT for surveillance. AJNR Am J Neuroradiol 35: 1281-1287.
-
Bodelle B, Wichmann JL, Scholtz J-E, Lehnert T, Vogl TJ, et al. (2015) Iterative Reconstruction Leads to Increased Subjective and Objective Image Quality in Cranial CT in Patients With Stroke. AJR Am J Roentgenol 205: 618-622.
-
Kilic K, Erbas G, Guryildirim M, Arac M, Ilgit E, et al. (2011) Lowering the Dose in Head CT Using Adaptive Statistical Iterative Reconstruction. AJNR Am J Neuroradiol 32: 1578-1582.
-
Korn A, Fenchel M, Bender B, Danz S, Hauser TK, et al. (2012) Iterative Reconstruction in Head CT: Image Quality of Routine and Low-Dose Protocols in Comparison with Standard Filtered Back-Projection. AJNR Am J Neuroradiol 33: 218-224.
-
Rapalino O, Kamalian S, Kamalian S, Payabvash S, Souza LCS, et al. (2012) Cranial CT with Adaptive Statistical Iterative Reconstruction: Improved Image Quality with Concomitant Radiation Dose Reduction. AJNR Am J Neuroradiol 33: 609-615.
-
Ren Q, Dewan SK, Li M, Li J, Mao D, et al. (2012) Comparison of adaptive statistical iterative and filtered back projection reconstruction techniques in brain CT. Eur J Radiol 81: 2597-2601.
-
Korn A, Bender B, Fenchel M, Spira D, Schabel C, et al. (2013) Sinogram affirmed iterative reconstruction in head CT: Improvement of objective and subjective image quality with concomitant radiation dose reduction. Eur J Radiol 82: 1431-1435.
-
Wu T-H, Hung S-C, Sun J-Y, Lin C-J, Lin C-H, et al. (2013) How far can the radiation dose be lowered in head CT with iterative reconstruction? Analysis of imaging quality and diagnostic accuracy. Eur Radiol 23: 2612-2621.
-
Ryska P, Kvasnicka T, Jandura J, Klzo L, Grepl J, et al. (2013) Reduction of effective dose and organ dose to the eye lens in head mdct using iterative image reconstruction and automatic tube current modulation. Biomed Pap. Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub 158: 265-272.
-
Komlosi P, Zhang Y, Leiva-Salinas C, Ornan D, Patrie JT, et al. (2014) Adaptive statistical iterative reconstruction reduces patient radiation dose in neuroradiology CT studies. Neuroradiology 56: 187-193.
-
Ozdoba C, Slotboom J, Schroth G, Ulzheimer S, Kottke R, et al. (2014) Dose Reduction in Standard Head CT: First Results from a New Scanner Using Iterative Reconstruction and a New Detector Type in Comparison with Two Previous Generations of Multi-slice CT. Clin Neuroradiol 24: 23-28.
-
Ohnesorge B, Flohr T, Schaller S, Klingenbeck-Regn K, Becker C, et al. (1999) The technical bases and uses of multi-slice CT. Radiologe 39: 923-931.
-
Becker HC, Augart D, Karpitschka M, Ulzheimer S, Bamberg F, et al. (2012) Radiation Exposure and Image Quality of Normal Computed Tomography Brain Images Acquired With Automated and Organ-Based Tube Current Modulation Multiband Filtering and Iterative Reconstruction: Invest Radiol 47: 202-207.
-
Beister M, Kolditz D, Kalender WA (2012) Iterative reconstruction methods in X-ray CT. Phys Medica PM Int J Devoted Appl Phys Med Biol Off J Ital Assoc Biomed Phys AIFB 28: 94-108.
-
Thibault J-B, Sauer KD, Bouman CA, Hsieh J (2007) A three-dimensional statistical approach to improved image quality for multislice helical CT. Med Phys 34: 4526-4544.
-
Nuyts J, De Man B, Dupont P, Defrise M, Suetens P, et al. (1998) Iterative reconstruction for helical CT: a simulation study. Phys Med Biol 43: 729-737.
-
Boone JM, Geraghty EM, Seibert JA, Wootton-Gorges SL (2003) Dose reduction in pediatric CT: a rational approach. Radiology 228: 352-360.
-
Shah R, Gupta AK, Rehani MM, Pandey AK, Mukhopadhyay S (2005) Effect of reduction in tube current on reader confidence in paediatric computed tomography. Clin Radiol 60: 224-231.
-
Ledenius K, Stålhammar F, Wiklund LM, Fredriksson C, Forsberg A, et al. (2010) Evaluation of image-enhanced paediatric computed tomography brain examinations. Radiat Prot Dosimetry 139: 287-292.
-
Wong ST, Yiu G, Poon Y-M, Yuen M-K, Fong D (2012) Reducing radiation exposure from computed tomography of the brain in children - report of a practical approach. Childs Nerv Syst 28: 681-689.
-
Vorona GA, Zuccoli G, Sutcavage T, Clayton BL, Ceschin RC, et al. (2013) The Use of Adaptive Statistical Iterative Reconstruction in Pediatric Head CT: A Feasibility Study. AJNR Am J Neuroradiol 34: 205-211.
-
Gabriel S, Eckel LJ, DeLone DR, Krecke KN, Luetmer PH, et al. (2014) Pilot study of radiation dose reduction for pediatric head CT in evaluation of ventricular size. AJNR Am J Neuroradiol 35: 2237-2242.
-
Noël PB, Fingerle AA, Renger B, Münzel D, Rummeny EJ, et al. (2011) Initial performance characterization of a clinical noise-suppressing reconstruction algorithm for MDCT. AJR Am J Roentgenol 197: 1404-1409.
-
Hu XH, Ding XF, Wu RZ, Zhang MM (2011) Radiation dose of non-enhanced chest CT can be reduced 40% by using iterative reconstruction in image space. Clin Radiol 66: 1023-1029.
-
Mullins ME, Lev MH, Bove P, O’Reilly CE, Saini S, et al. (2004) Comparison of image quality between conventional and low-dose nonenhanced head CT. AJNR Am J Neuroradiol 25: 533-538.
-
McNitt-Gray MF (2002) AAPM/RSNA Physics Tutorial for Residents: Topics in CT. Radiation dose in CT. Radiographics 22: 1541-1553.
-
Deutsche Gesellschaft für Neurochirurgie e.V. (DGNC) (2007) Schädel-Hirn-Trauma im Erwachsenenalter.
-
Steiger H-J, Reulen H-J, Steiger-Reulen (2006) Manual Neurochirurgie, 2, völlig überarb. und aktualisierte Aufl. ecomed Medizin, Landsberg.
-
Pexman JH, Barber PA, Hill MD, Sevick RJ, Demchuk AM, et al. (2001) Use of the Alberta Stroke Program Early CT Score (ASPECTS) for assessing CT scans in patients with acute stroke. AJNR Am J Neuroradiol 22: 1534-1542.
-
Islam MN, Kuddus R, Chowdhury NS, Akhter MD, Salahuddin G, et al. (2014) Radiologic evaluation of hyperacute brain infarction: a review. Mymensingh Med J MMJ 23: 621-635.
-
Malatt C, Zawaideh M, Chao C, Hesselink JR, Lee RR, et al. (2014) Head computed tomography in the emergency department: a collection of easily missed findings that are life-threatening or life-changing. J Emerg Med 47: 646-659.
-
McCollough CH, Yu L, Kofler JM, Leng S, Zhang Y, et al. (2015) Degradation of CT Low-Contrast Spatial Resolution Due to the Use of Iterative Reconstruction and Reduced Dose Levels. Radiology 276: 499-506.
-
Nishizawa M, Tanaka H, Watanabe Y, Kunitomi Y, Tsukabe A, et al. (2015) Model-based iterative reconstruction for detection of subtle hypoattenuation in early cerebral infarction: a phantom study. Jpn J Radiol 33: 26-32.
-
Leipsic J, Labounty TM, Heilbron B, Min JK, Mancini GBJ, et al. (2010) Adaptive statistical iterative reconstruction: assessment of image noise and image quality in coronary CT angiography. AJR Am J Roentgenol 195: 649-654.
-
Vaishnav JY, Jung WC, Popescu LM, Zeng R, Myers KJ (2014) Objective assessment of image quality and dose reduction in CT iterative reconstruction. Med Phys 41: 071904.





