International Journal of Pediatric Research
Short Lingual Frenulum and Obstructive Sleep Apnea in Pre-Pubertal Children
Yu-Shu Huang1, Stacey Quo2, J Andrew Berkowski3 and Christian Guilleminault3*
1Chang Gung Memorial Hospital and University Pediatric sleep laboratory, Taiwan
2UCSF Dental School, USA
3Stanford University Sleep Medicine Division, USA
*Corresponding author: Christian Guilleminault, Stanford University Sleep Medicine Division, 450 Broadway Pavilion C, 2nd floor, Redwood City, CA 94063, USA, E-mail: cguil@stanford.edu
Int J Pediatr Res, IJPR-1-003, (Volume 1, Issue 1), Research Article; ISSN: 2469-5769
Received: January 27, 2015 | Accepted: April 06, 2015 | Published: April 08, 2015
Citation: Huang YS, Quo S, Berkowski JA, Guilleminault C (2015) Short Lingual Frenulum and Obstructive Sleep Apnea in Children. Int J Pediatr Res 1:003. 10.23937/2469-5769/1510003
Copyright: © 2015 Yu-shu H, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Background: Abnormal short lingual frenulum may lead to impairment of orofacial growth in early childhood. This may reduce the width of the upper airway-a pliable tube-increasing its risk of collapse, particularly during sleep.
Study: A retrospective study of prepubertal children referred for suspicion of obstructive sleep apnea, found 27 subjects with non-syndromic short lingual frenulum. The children had findings associated with enlarged adenotonsils and/or orofacial growth changes.
Results: Children with untreated short frenulum developed abnormal tongue function early in life with secondary impact on orofacial growth and sleep disordered breathing (SDB).
After presence of SDB, analysis of treatment results revealed the following: The apnea-hypopnea index (AHI) of children with adenotonsillectomy (T&A) performed without frenectomy improved, but surgery did not resolve fully the abnormal breathing. Similar results were noted when frenectomy was performed simultaneously with T&A. Finally, frenectomy on children two years or older without enlarged adeno tonsils also did not lead to normalization of AHI. The changes in orofacial growth related to factors including short lingual frenulum lead to SDB and mouth-breathing very early in life. Recognition and treatment of short frenulum early in life-at birth, if possible-would improve normal orofacial growth. Otherwise, myofunctional therapy combined with education of nasal breathing is necessary to obtain normal breathing during sleep in many children.
Conclusion: Short lingual frenulum may lead to abnormal orofacial growth early in life, a risk factor for development of SDB. Careful surveillance for abnormal breathing during sleep should occur in the presence of short lingual frenulum.
Keyword
Obstructive sleep apnea, Prepubertal, Short lingual Frenulum, Ankyloglossia, Frenectomy, Myofunctional therapy
Introduction
Abnormally short lingual frenulum may be seen in association with well-described syndromes such as Beckwitz-Weideman, orofacial digital syndrome, cleft palate, Optiz syndrome and others. Many of these syndromes are associated with abnormal breathing during sleep and obstructive sleep apnea (OSA) [1]. However, they are most commonly isolated, involving more boys than girls with a ratio of 3:1, and with a reported incidence of 5% at birth [1]. Studies of the impact of short frenulum have shown that such anatomic presentation impairs suction [2-5], chewing, swallowing, and frequently leads to speech disorders [6]. Recently, protocols have been published for evaluation of lingual frenulum both in infants and in children [2,7-9]. It was emphasized that a short lingual frenulum modifies the position of the tongue, particularly early in life, and impairs orofacial development. There is secondary association with anterior and posterior crossbite, disproportionate growth of the mandible, and abnormal growth of the maxilla [10,11]. Treatments for short lingual frenulum have been proposed and involve lingual frenectomy [12-14].
The anatomical changes seen with short lingual frenulum are related to the interdependence between function and form as indicated by Melvin Moss [15]: The observed anatomical orofacial changes lead to development of abnormal anatomic support of the upper airway. The upper airway is a collapsible tube submitted to negative intrathoracic pressure during inspiration; during sleep there is an increased risk of collapsibility due to the change of muscle tone related to sleep stages and state along with the recumbent position associated with sleep [16].
We performed a retrospective study of children and teenagers referred for suspicion of sleep-disordered breathing (SDB) during the last five years, and identified from our de-identified database, individuals with short lingual frenulum. We considered the associated orofacial anatomical changes (including the presence of enlarged adenotonsils and other orofacial changes), the recommended treatments, and the outcome of these treatments. This retrospective study of anonymous data was approved by the IRB.
Subjects and Testing
The de-identified charts of children, age 2 to 17 years, referred for suspicion of SDB were selected. Obese and syndromic children were eliminated from the review. To be included in the analysis, charts contained information on initial complaints leading to consult with usage of Pediatric Sleep Questionnaire [17], report of clinical evaluation of orofacial findings indicating investigation of tonsils using the Friedman scale [18], investigation of subjective upper-airway opening using the Mallampati-Friedman scale [18], evaluation of inferior nasal turbinates, dental crowding, presence of over jet and/or overbite [18], and determination of "facial harmony" with frontal measurements [18]. If nasal allergies were suspected, a consult with an allergist for treatment was obtained, and if orthodontic problems were suspected, results of evaluation by a specialist were available. Indication of short lingual frenulum and its potential association with speech, swallow, or mastication problems were outlined. Such impairment may have occurred during early childhood, and treatment of the secondary consequence (such as speech therapy) may have been implemented without addressing the primary cause (short lingual frenulum).
All patients had in-laboratory diagnostic polysomnography (PSG) that included the following: EEG (4 leads), EOG, chin and leg EMG, ECG (one lead), and body position. The respiration was monitored using nasal pressure transducer; mouth-breathing with thermos-couple; chest and abdominal movements with inductive plethysmography bands, diaphragmatic-intercostal, and rectus-oblique muscle EMG; pulse oximetry (Massimo™) from which both oxygen saturation (SaO2) and finger-plethysmography were derived; and continuous video monitoring.
The conclusion of the testing was abnormal breathing during sleep with indication of the treatment plan.
Post treatment follow-up was available indicating:
a)Selected treatment
b)Changes compared to baseline, including questionnaire data
c)Post-treatment PSG findings
Based on the results obtained, a follow-up decision contained any further treatment recommendations and subsequent follow up information including PSG data.
Analysis
The data were collected and organized on spreadsheets. Sleep and respiratory scoring of PSGs followed the pediatric scoring guidelines, according to the American Academy of Sleep Medicine (AASM-2007) [19]. The presence of nasal flow limitation was determined using criteria published by Guilleminault et al. [20] and Palombini et al. [21]. The time spent mouth-breathing during sleep was also calculated as a percentage of total sleep time [22]. Data were analyzed using t-test for repeated measures.
Results
We identified 27 patients with association of short lingual frenulum and SDB who met entry criteria (Figure 1). The mean age was 11.4 years (range: 2 to 16 years). Children presented with symptoms of SDB (Table 1) such as snoring, poor sleep, and fatigue but also a history of symptoms associated with short lingual frenulum such as problems with speech, swallowing or suction, particularly early in life. Children with speech problems may have been sent to speech therapy, but none of the children had frenulum treatment. As can be seen (Table 1), 10 children had enlarged tonsils (Friedman scores of 3 and 4), five children had been recommended to consult an orthodontist due to evident crowding of dentition, 22 children presented with an abnormal Mallampati-Friedman score of 3 or 4 [18]. In all cases, there was presence of a high and narrow hard palate and distortion of the "harmonic face" [18] with either a longer lower third of the face or reduction of the middle third compared to the other thirds.
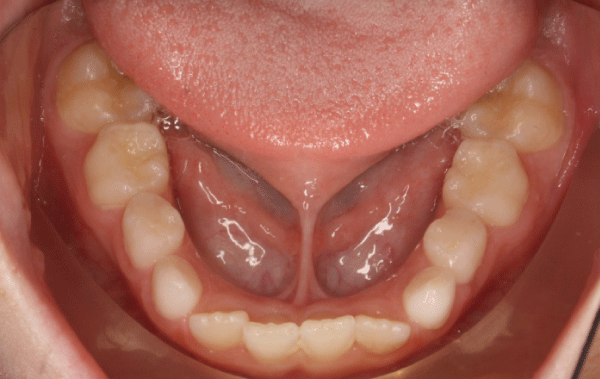
.
Figure 1: Example of a short lingual frenulum in a prepubertal child with obstructive sleep apnea
View Figure 1
![]()
Table 1: Disease characteristics at entry and after first treatment
View Table 1
All children presented with abnormal orofacial anatomy and reduced oral cavity size. None of the children or their parents was aware of the presence of an abnormal short lingual frenulum, despite history of speech therapy early in life, feeding and/or swallowing difficulties, or prior adenotonsillectomy (T&A) or orthodontic treatment.
All children had PSG s with an abnormal apnea-hypopnea-index (AHI) and abnormal nadir of oxygen saturation during sleep. Results are presented in table 1.
In sum: on initial presentation, children with a short lingual frenulum had associated anatomic problems linked to abnormal breathing during sleep, such as adenotonsillar hypertrophy, but all presented with orofacial features favoring collapse of the upper airway during sleep with presence of a high and narrow palatal vault [16,22].
Finally, detailed family history revealed that one parent had a short frenulum in 6 out of our 27 cases, and short frenulum was present in three siblings of the patients.
Following recognition of abnormal breathing during sleep, some children (n=10) had been referred to ear-nose-throat specialists (ENT) for T&A and frenectomy when large tonsils were present, and 8 children with normal sized tonsils (confirmed by naso-pharyngoscopy and/or lateral radiographs) were also referred to ENT for frenectomy. Nine children were referred to orthodontists for rapid maxillary expansion (RME) and need for frenectomy was also mentioned. Children were asked to have post-treatment follow-up by sleep medicine including investigation with PSG.
Follow-up post treatment#1
The sleep medicine follow-up occurred between three and four months post T&A with frenectomy or isolated frenectomy; and about one month after rapid maxillary expansion, with expander in place, for children sent to orthodontists. None of the children sent for orthodontia treatment had frenectomy and two children had T&A without frenectomy. In summary: 13 children had frenectomy either isolated (n=5) or in association with T&A (n=8), but frenectomy was not performed in 14 children despite recommendation to perform such treatment.
Table 1 presents the results obtained post treatment #1, independent of its type.
There was an overall improvement by clinical evaluation and PSG including children in which frenectomy had not been performed; parents reported, however, persistence of some symptoms particularly of "fatigue" and "poor sleep" in about one third of the cases. The AHI was significantly decreased, and oxygen saturation nadir was significantly improved (p=0.01 for each condition, paired t-test); flow limitation was also improved (p=0.05 paired t-test) but to a lesser degree.
There was persistence of mouth-breathing: only two children, treated by both T&A and frenectomy spent less than 10% of their sleep mouth breathing, a percentage considered as normal [28]. All others had abnormal mouth-breathing during sleep, including the five children that underwent isolated frenectomy.
The conclusion of this first follow-up was that children recognized with OSA were clearly improved after either T&A or orthodontics, performed with or without frenectomy. However, 92.5% of the treated children still had an abnormal amount of mouth-breathing during sleep.
After collection of data, children without frenectomy were referred back to ENT and recommended to undergo treatment (n=16). Considering the prior finding of persistence of mouth-breathing despite frenectomy, all children with residual AHI, flow limitation, and mouth-breathing were recommended to have myofunctional treatment with a specialist after surgery (n=25).
Follow-up post treatment #2
Eleven patients came back for further follow-up, six months after the first post treatment investigation. They had treatment with myofunctional therapy for 4 to 6 months [23-25].
These included those who underwent frenectomy with orthodontia (n=5), isolated frenectomy (n=4)), and children post T&A with new frenectomy subsequent to the first follow up visit (n= 2).
None of these 11 patients or their parents had clinical complaints. At follow-up PSG, these cases had a mean AHI of 0.8 ± 0.9, a mean oxygen saturation nadir of 97.2 ± 1.0%, a mean flow limitation during sleep of 7.5 ± 6%, and time spent mouth breathing during sleep of 4 ± 4.1%.
Discussion
Diagnosing frenulum abnormalities can be difficult because the examiner has to be aware of the anatomy of the tongue, including different aspects of the frenulum and adjacent regions. In addition, the examiner must know what functions may be affected by the alterations of the lingual frenulum. Moreover, the cut-off point between normal and abnormal frenulum may be a challenge particularly early in life. In this study, all patients were non-syndromic and had a clearly recognizable short lingual frenulum at clinical evaluation. None of the children referred for suspicion of SDB had been recognized with such anatomical abnormality before referral. Short lingual frenulum is a known factor in altering orofacial growth particularly impacting development of the maxilla due to the low placement of the tongue. It leads to the abnormal development of a high and narrow hard palate, and secondarily, mouth-breathing during sleep. These changes occur early in life, as the orofacial growth is particularly fast during the first two years of life. The observed secondary anatomical changes occurring in the oral cavity are known to increase the degree of collapsibility of the upper airway during sleep, and recognition of a short lingual frenulum in a toddler and older children should lead to investigation of SDB.
It has also been hypothesized that mouth-breathing may be a factor in tonsillar enlargement: normally a child is an an obligate nose-breather and the nose humidifies, heats, and regulates airflow before it reaches the distal portions of the upper airway. Mouth-breathing not only increases upper airway resistance [26] but also causes micro trauma to the back of the throat that may induce local inflammatory reactions in the tonsils, leading to their enlargement. Unrecognized short lingual frenulum at birth or very early in life may lead to suction, swallowing, masticatory, and speech acquisition problems. It negatively impacts orofacial growth leading to abnormal breathing during sleep, including development of mouth-breathing that will in turn worsen the abnormal orofacial growth. This can produce "nasal disuse," particularly during sleep, an often-missed condition [22] that will not spontaneously improve even after surgical elimination of the abnormal anatomic presentation; daytime reeducation and re-training of nasal usage will be needed.
Our study involves a relatively small number of children, and our longest follow-up investigation is with fewer children than at initial presentation due to loss to follow up, and such factors are limitations in the interpretation of our findings.
We have no indication why frenectomy was not performed earlier or even, in many cases, at time of T&A. Frenectomy does not significantly increase the surgical risks associated with T&A. Considering the negative impact of short lingual frenulum on orofacial development of children early in life, and the fact that the above children were already presenting with OSA, we suggest that lingual frenulum be systematically examined by otolaryngologists and pediatricians in the presence of early in life difficulties with function of the oral cavity and in the presence of SDB. Additionally, frenectomy should be integrated in surgical planning, independently of its timing [27].
Evaluation of breathing during sleep should be performed as early as possible in subjects recognized with short lingual frenulum. One fact is clear: frenectomy for short lingual frenulum in isolation or following T&A helps but is commonly insufficient to resolve all abnormal breathing patterns during sleep when SDB is present. Myofunctional therapy, which has been previously demonstrated to allow return to normal nasal breathing [22-25] may be needed post surgery.
Finally, the existence of familial cases and the association with genetic syndrome suggest that presence of a short lingual frenulum may be part of a specific genetic predisposition, but no genetic study has been performed on familial cases of short lingual frenulum.
In conclusion: children with SDB should be evaluated for a short lingual frenulum, and conversely, children with an abnormally short frenulum should be investigated for the presence of SDB. Frenectomy should be performed as early as possible but it may not be sufficient to restore normal nasal breathing function during sleep [28], particularly if the frenulum-related problem has lingered over years, and nasal breathing reeducation may be needed in these cases.
References
-
Klockars T (2007) Familial ankyloglossia (tongue-tie). Int J Pediatr Otorhinolaryngol 71: 1321-1324.
-
Kotlow LA (2004) Oral diagnosis of oral frenulum attachments in neonates and infants: evaluation and treatment of the maxillary and lingual frenulum using the Erbium-YAG laser. J Pediatr Dent Care 10: 11-13.
-
Dollberg S, Botzer E, Grunis E, Mimouni FB (2006) Immediate nipple pain relief after frenotomy in breast-fed infants with ankyloglossia: a randomized, prospective study. J Pediatr Surg 41: 1598-1600.
-
Srinivasan A, Dobrich C, Mitnick H, Feldman P (2006) Ankyloglossia in breastfeeding infants: the effect of frenotomy on maternal nipple pain and latch. Breastfeed Med 1: 216-224.
-
Wallace H, Clarke S (2006) Tongue tie division in infants with breast feeding difficulties. Int J Pediatr Otorhinolaryngol 70: 1257-1261.
-
Queiroz Marchesan I (2004) Lingual frenulum: classification and speech interference. Int J Orofacial Myology 30: 31-38.
-
Kotlow LA (1999) Ankyloglossia (tongue-tie): a diagnostic and treatment quandary. Quintessence Int 30: 259-262.
-
Martinelli RL, Marchesan IQ, Berretin-Felix G (2012) Lingual frenulum protocol with scores for infants. Int J Orofacial Myology 38: 104-112.
-
Martinelli RLC, Marchesan IQ, Berretin-Felix G (2014) Longitudinal study of the anatomical characteristics of the lingual frenulum and comparison to the literature. Rev. CEFAC 16: 1202-1207.
-
Defabianis P (2000) Ankyloglossia and its influence on maxillary and mandibular development. (A seven year follow-up case report). Funct Orthod 17: 25-33.
-
García Pola MJ, González García M, García Martín JM, Gallas M, Seoane Lestón J (2002) A study of pathology associated with short lingual frenum. ASDC J Dent Child 69: 59-62, 12.
-
Fiorotti RC, Bertolini MM, Nicola JH, Nicola EM (2004) Early lingual frenectomy assisted by CO2 laser helps prevention and treatment of functional alterations caused by ankyloglossia. Int J Orofacial Myology 30: 64-71.
-
Haytac MC, Ozcelik O (2006) Evaluation of patient perceptions after frenectomy operations: a comparison of carbon dioxide laser and scalpel techniques. J Periodontol 77: 1815-1819.
-
Olivi G, Signore A, Olivi M, Genovese MD (2012) Lingual frenectomy: functional evaluation and new therapeutical approach. Eur J Paediatr Dent 13: 101-106.
-
Moss-Salentijn L (1997) Melvin L. Moss and the functional matrix. J Dent Res 76: 1814-1817.
-
Guilleminault C , Akhtar F (2015) Pediatric sleep-disordered-breathing: new evidence on its development Sleep Med. Rev. (in press).
-
Chervin RD, Hedger K, Dillon JE, Pituch KJ (2000) Pediatric sleep questionnaire (PSQ): validity and reliability of scales for sleep-disordered breathing, snoring, sleepiness, and behavioral problems. Sleep Med 1: 21-32.
-
Kim JH, Guilleminault C (2011) The nasomaxillary complex, the mandible, and sleep-disordered breathing. Sleep Breath 15: 185-193.
-
Iber C, Ancoli-Israel S, Chesson AL, Jr., Quan SF. for the American Academy of Sleep Medicine (addended Berry RB, Brooks R, Gamaldo CE) (2012) The AASM manual for the scoring of sleep and associated events: rules, terminology and technical specifications.
-
Guilleminault C, Li KK, Khramtsov A, Pelayo R, Martinez S (2004) Sleep disordered breathing: surgical outcomes in prepubertal children. Laryngoscope 114: 132-137.
-
Palombini LO, Tufik S, Rapoport DM, Ayappa IA, Guilleminault C, et al. (2013) Inspiratory flow limitation in a normal population of adults in São Paulo, Brazil. Sleep 36: 1663-1668.
-
Lee SY , Guilleminault C, Chiu HY, Sullivan SS (2015) Mouth breathing, nasal 'dis-use and sleep- disordered-breathing Sleep-Breath. (in press)
-
Chauvois A, Fournier, M. Girardin, F (1991) Reeducation des fonctions dans la therapeutique orthodontiques. Paris: S.I.D.
-
Guimarães KC, Drager LF, Genta PR, Marcondes BF, Lorenzi-Filho G (2009) Effects of oropharyngeal exercises on patients with moderate obstructive sleep apnea syndrome. Am J Respir Crit Care Med 179: 962-966.
-
Guilleminault C, Huang YS, Monteyrol PJ, Sato R, Quo S, et al. (2013) Critical role of myofascial reeducation in pediatric sleep-disordered breathing. Sleep Med 14: 518-525.
-
Fitzpatrick MF, McLean H, Urton AM, Tan A, O'Donnell D, et al. (2003) Effect of nasal or oral breathing route on upper airway resistance during sleep. Eur Respir J 22: 827-832.
-
Marchesan IQ, Martinelli RL, Gusmão RJ (2012) Lingual frenulum: changes after frenectomy. J Soc Bras Fonoaudiol 24: 409-412.
-
Guilleminault C, Sullivan SS (2014) Towards restoration of continuous nasal breathing as the ultimate treatment goal in pediatric obstructive sleep apnea. Enliven: Pediatr Neonatol Biol 1: 001.





