International Journal of Ophthalmology and Clinical Research
Successful Results of Severe Corneal Perforation Using Long Term Glycerin Corneal Donor Cryopreserved At -80 °C: Case Report
Villalba R1*, Giménez-Almenara J2, Gallardo JM2 and Oyonarte S1
1Blood Transfusion Center and Tissue Bank of Córdoba, Spain
2Ophthalmology Service, Reina Sofía Hospital, Córdoba, Spain
*Corresponding author: Villalba R, Blood Transfusion Center and Tissue Bank of Córdoba, Avda, San Alberto magno s/n, 14004 Córdoba, Spain, Tel: +34-57-011100, Fax: +34-51-011111, E-mail: rafael.villalba.sspa@juntadeandalucia.es
Int J Ophthalmol Clin Res, IJOCR-4-068, (Volume 4, Issue 1), Case Report; ISSN: 2378-346X
Received: October 10, 2016 | Accepted: March 22, 2017 | Published: March 24, 2017
Citation: Villalba R, Giménez-Almenara J, Gallardo JM, Oyonarte S (2017) Successful Results of Severe Corneal Perforation Using Long Term Glycerin Corneal Donor Cryopreserved At -80 °C: Case Report. Int J Ophthalmol Clin Res 4:068. 10.23937/2378-346X/1410068
Copyright: © 2017 Villalba R, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Introduction
There has been long interest in developing alternatives to donor corneas preservation for providing tissue during prolonged period of time. In recent years, accumulative evidence suggest that glycerin preserved cornea at -80 °C as alternative method for corneal preservation maintaining sterility, thickness, optical transparency and mechanical strength of the tissue [1].
In the last 20 years we have observed a major change in the indications transplant corneas using lamellar techniques [2]. Among these techniques, the use of endothelial tissue for Descemet's membrane endothelial keratoplasty (DMEK) is a method with excellent clinical results [3].
With the preparation procedure of the grafts under strict controlled conditions (Clean-room) we obtained, together with the endothelium, another tissue that correspond to the anterior lamella. This structure basically is compoused by stroma and epithelium.
Accumulative evidence suggest that cellular components of the fresh corneal tissue including the epithelium, keratocytes and the bone marrow derived cells are sources of major histocompatibility complex (MHC) antigens and minor H antigens are responsible for tissue rejection [4].
Previous paper [4,5] have shown that corneal tissues stores in glycerol at -80 °C were least antigenic and also retains optical transparency, thickness and strength compared to tissue stored at 4 °C.
In this case report we have used an anterior lamella glycerol preserved at -80 °C in Tissue Banking after tissue preparation in a pre-cut cornea for DMEK procedure for the treatment of reconstructive surgery of an eye severely injured by trauma with an excellent clinical results.
Case Report
A 68-year-old man was injured in the left eyelid after accidental trauma for which he was treated with reconstructive surgery of upper and lower eyelid, and then with a deep anterior lamellar keratoplasty (DALK) for corneal leucoma produced by corneal laceration that he suffered in the same accident.
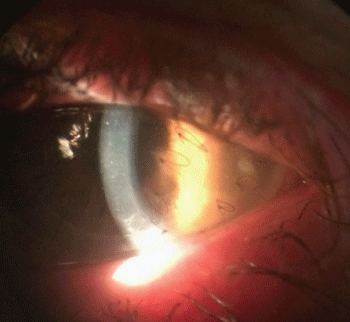
After a good postoperative course, 3 months later the patient present eye discomfort. The eye has melting at the recipient part of the cornea, without involvement of donor cornea. It is located at the inferior temporal margin and coinciding with suture that have been extruded from inferior eyelid.
The melting was following by corneal perforation with athalamia.
The emergency treatment consisted in the use of tissue adhesive (Histoacryl®) and placed a therapeutic lens. This treatment was followed by programmed surgery applying a partial graft in the melting area.
We clean the damaged area in an almond shape and prepare the allograft with the same shape of the receiving area, we reduce the thickness by removing the posterior stroma, descemet membrane, endothelium and epithelium, to adapt the graft perfectly.
The allograft we used was distributed from our Tissue Bank previously frozen at -80 °C in 100% glycerol by 30 days.
Allograft was sutured with nylon 10/0 stitches and covered with allograft of amniotic membrane to promote epithelialization of the area. That procedure solves the damage (Figure 1).
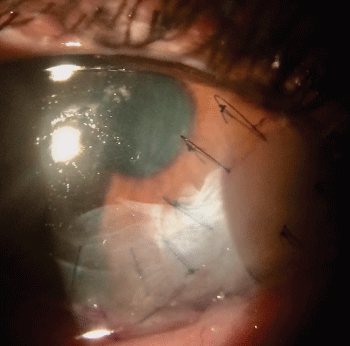
.
Figure 1: Two weeks postoperatively. The eye is quiet. The amniotic membrane remains.
View Figure 1
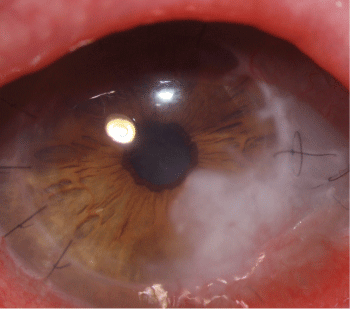
A positive outcome was observed postoperatively and after topical treatment based on corticosteroids, antibiotics and 20% autologous serum, maintaining a therapeutic lens during the first month. The graft epithelialization was complete at the end of the amniotic membrane resorption after 20 days (Figure 2).
To date, 6 months of follow-up the graft remains stable and has been vascularized. In addition to this has not presented rejection without immunosuppresive treatment. The patient show a correct apposition of edges, adequate transparency and graft thickness and satisfactory epithelialization without vascularitation or rejection signs (Figure 3).
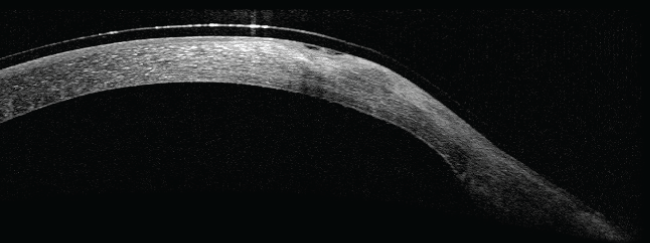
Optical coherence tomography (OCT) revealed a good tissue integration in host donor stroma was very similar to recipient (Figure 4).
Visual acuity is 1/3 with correction of + 3 - 7 × 60. We consider that this high astigmatism is caused by the stitches and we hope a decrease after its withdrawal at 6 months after surgery.
Discussion
For the treatment of this patient we could choose some procedures [6] that in our opinion, have a worst outcome.
-Perforation closure with amniotic membrane only [7]. The DALK have only three months, the melting decreases the tension of the suture, which can not be resolved properly only by membrane suturing because of the loss of substance in the area.
-Closing drilling with autologous [8], lyophilized or preserved [9] scleral graft could be an option but it has limitations due to its transparency, and a worse handling and adaptation to the area.
-Closing drilling by a fresh donor cornea [10]. In our opinion graft antigenicity in addition to a peripheral drilling, increases the vascularization risk promoting the rejection of the graft.
So we decided to carry out this technique with the use of lamellar anterior allograft frozen with glycerol 100%. Accumulative evidence suggest that cryopreservation reduce antigenicity that reduce the rejection likelihood [1,11].
Furthermore the graft is transparent, maintaining optical purpose of this area and additionally is identical to the altered structure thereby maintaining the anatomy of the area.
This is a unique case followed for 6 months, but thanks to the ease of handling of the graft, good evolution that has taken so far and transparency, stability, absence of rejection and vascularization makes us hope that this type graft can be used for this and more types of eye diseases.
The positive outcome observed in this case encourages us to follow the investigation. Prospective clinical studies would help validate these findings a possible prove beneficial patients care.
References
-
Chen W, Lin Y, Zhang X, Wang L, Liu M, et al. (2010) Comparison of fresh corneal tissue versus glycerin cryopreserved corneal tissue in deep anterior lamellar keratoplasty. Invest Ophthalmol Vis Sci 51: 775-781.
-
Lee WB, Meinecke E, Varnum B (2013) The evolution of eye banking and corneal transplantation: a symbiotic relationship. Int Ophthalmol Clin 53: 115-129.
-
Ruzza A, Parekh M, Salvalaio G, Ferrari S, Ponzin D (2016) Development of a new superficial punch for Descemet's membrane endothelial keratoplasty donor tissue preparation. Br J Ophthalmol 100: 443-445.
-
Tripathi H, Mehdi MU, Gupta D, Sen S, Kashyap S, et al. (2016) Long-term preservation of donor corneas in glycerol for keratoplasty: exploring new protocols. Br J Ophthalmol 100: 284-290.
-
Javadi MA, Feizi S, Javadi F, Kanavi MR, Ghasemi H, et al. (2014) Deep anterior lamellar keratoplasty using fresh versus cryopreserved corneas. Ophtalmology 121: 610-611.
-
Duchesne B, Tahi H, Galand A (2001) Use of human fibrin glue and amniotic membrane transplant in corneal perforation. Cornea 20: 230-232.
-
Vora GK, Haddadin R, Chodosh J (2013) Management of corneal lacerations and perforations. Int Ophthalmol Clin 53: 1-10.
-
Prasher P (2014) Use of an autologous lamellar scleral graft to repair a corneal perforation. Int Ophtalmol 34: 957-960.
-
Oh JH, Kim JC (2003) Repair of scleromalacia using preserved scleral graft with amniotic membrane transplantation. Cornea 22: 288-293.
-
Hanada K, Igarashi S, Muramatsu O, Yoshida A (2008) Therapeutic keratoplasty for corneal Perforation: Clinical Results and Complications. Cornea 27: 156-160.
-
Lin HC, Ong SJ, Chao AN (2012) Eye preservation tectonic graft using glycerol-preserved donor cornea. Eye (Lond) 26: 1446-1450.