International Journal of Ophthalmology and Clinical Research
Predictors of Visual Outcome in Eyes with Neovascular Age-Related Macular Degeneration: A New Stratified Approach Using Spectral-Domain Optical Coherence Tomography
Antonio Polito1*, Emilia Maggio1, Antonio Peroglio Deiro1, Massimo Guerriero2 and Grazia Pertile1
1Department of Ophthalmology, Ospedale S. Cuore - Don Calabria, Verona, Italy
2Department of Computer Science, Statistical Section, University of Verona, Verona, Italy
*Corresponding author: Antonio Polito, MD, Department of Ophthalmology, Ospedale S. Cuore - Don Calabria Via Don Sempreboni, 37024 Negrar, Italy, Tel: 0039-045-6013857, Fax: 0039-045-6013026, E-mail: antonio.polito@sacrocuore.it
Int J Ophthalmol Clin Res, IJOCR-3-063, (Volume 3, Issue 4), Research Article; ISSN: 2378-346X
Received: January 30, 2016 | Accepted: November 05, 2016 | Published: November 08, 2016
Citation: Polito A, Maggio E, Deiro AP, Guerriero M, Pertile G (2016) Predictors of Visual Outcome in Eyes with Neovascular Age-Related Macular Degeneration: A New Stratifed Approach Using Spectral-Domain Optical Coherence Tomography. Int J Ophthalmol Clin Res 3:063. 10.23937/2378-346X/1410063
Copyright: © 2016 Polito A, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Purpose: To report a new stratified approach to predict visual acuity outcome after anti-vascular endothelial growth factor therapy for neovascular age-related macular degeneration (AMD) using Spectral-Domain Optical Coherence Tomography.
Methods: We reviewed a consecutive series of 70 patients after three monthly intravitreal ranibizumab treatments for primary neovascular AMD. Age, sex, baseline vision, choroidal neovascularization type as well as OCT characteristics were correlated to final vision. On OCT, baseline integrity of the fovea was studied using foveal thickness (FTH), external limiting membrane (ELM) and ellipsoid zone as hallmarks. Masked readers graded images. Multivariate analyses and stepwise regressions were performed to correlate baseline characteristics with vision at 3 months.
Results: Baseline vision (p-value = 0.0001) and ELM (p-value = 0.033) were the only significant independent predictors of final vision. However, a regression model stratifying baseline vision according to the status of pretreatment ELM turned out to best predict the variance in vision at month 3 (R2 = 0.87, 0.75, 0.68 for absent, disturbed and present ELM, respectively).
Conclusion: Improved prognostic information can be obtained by using the pretreatment ELM as a stratifying biomarker of baseline vision. A reliable prognosis could be of great value to guide the therapeutic approach especially in patients that are unlikely to benefit from the treatment and are not supported by a health care insurance.
Introduction
Age-related macular degeneration (AMD) is the leading cause of severe vision loss in elderly patients in industrialized countries [1]. The introduction of anti-vascular endothelial growth factor agents has revolutionized the treatment of choroidal neovascularization (CNV) by providing favourable visual outcomes for the first time. However, despite complete resolution of exudative changes and reduction of retinal thickening, some patients show less improvement than others [2,3].
Over the past few years, a series of studies reported a good correlation between the integrity of the outer retinal layers at baseline as imaged by spectral domain optical coherence tomography (SD-OCT) and visual acuity after treatment for neovascular AMD [4-6].
In particular, the status of the inner segment ellipsoid zone (EZ), previously defined as the junction of the inner and outer segment of the photoreceptor, had been initially used to evaluate the correlation between retinal morphology and function in patients with AMD [7,8].
However, the detection of this line varies widely based on the activity and severity of the disease, so that recently more attention has been focused on a layer which is presumably less sensitive than the EZ in the earlier stages of disease: the external limiting membrane (ELM). This is a thin line above the EZ delimiting the border between the outer nuclear layer and inner segment myoid portion. ELM status has been used to assess the correlation between retinal damage and function in a series of maculopathies [9-11]. Moreover, recent studies have indicated this band as the best candidate to predict visual improvement in neovascular AMD patients treated with anti-VEGF therapy [4,5,12-14]. Nevertheless, in these reports, retinal morphology, including the ELM, only accounts for a small degree of post-treatment variation in visual acuity. Therefore, despite the advancement in imaging technology, the single best predictor of post-treatment visual acuity consistently identified across randomized clinical trials remains baseline visual acuity [15-19]. Nevertheless, BCVA alone is not sensible enough to be used as a predictor factor. There are some patients with low visual acuity at baseline that can significantly benefit from the treatment. Being able to predict the outcome of anti-VEGF therapy could be particularly important for counselling, in particular for patients with a poor functional prognosis that don' t have free access to this treatment.
In an effort to obtain the best prognostic information with the new SD-OCT technology, in our study we evaluated whether using retinal morphology to grade the integrity of the outer retinal layers, and in particular the ELM, would yield improved prediction of post-treatment visual acuity.
Patients and Methods
Institutional review board approval was obtained for this study. The clinical records of 70 eyes of 70 consecutive patients after three intravitreal ranibizumab injections for primary subfoveal or juxtafoveal neovascular AMD were retrospectively reviewed. Patients with lesion size greater than 12 disc areas, subfoveal fibrosis, center involving geographic atrophy and vision below 20/250 were excluded. The research adhered to the tenets set forth in the Declaration of Helsinki.
Data collection included best-corrected visual acuity (BCVA), fluorescein angiography, indocyanine green angiography and SD-OCT (Spectralis HRA+OCT, Heidelberg, Germany) examination at the initial visit and at 3 months. BCVA was recorded using Snellen visual acuity charts. Two single, horizontal and vertical 9 mm B-scans centred on the fovea and a 3-D cube scan consisting of 19 20° B-scans were recorded for each eye. The eye-tracking system was used to assure correct positioning and the location of the fovea was manually detected during the exam. Nineteen scans were averaged for each recording. The analysis was performed in the central 1 mm centred on the fovea. All eyes included in the study presented high quality images with good signal strength (> 16 dB). All OCT images were assessed independently by two graders (E.M. and A.P.D.). They were masked to the BCVA recordings. Discrepancies between the two graders were adjudicated between the graders and the senior author (A.P.).
The status of the inner segment EZ and ELM were graded as "absent" when the line could not be detected, "disturbed", when the line was only partially detected and appeared discontinuous, and "present" when the line was clearly detected and completely visible (Figure 1). Foveal thickness (FTH) was defined as the distance between the vitreoretinal border and the edge of the outer retina; it was measured manually at the precise centre of the fovea with a calliper measurement tool provided in the system. Eyes were also classified according to the combined angiographic and tomographic pattern of the choroidal neovascularization (CNV) [20]. Type 1 CNV membranes showed an occult angiography pattern and a major OCT component beneath the retinal pigment epithelium. Type 2 CNV membranes showed a predominantly classic angiographic pattern and a major component of CNV above the RPE, in the subretinal space. Type 3 lesions showed angiographic signs of retinal angiomatous proliferations corresponding to intraretinal tomographic abnormalities suggestive of an intraretinal CNV component.
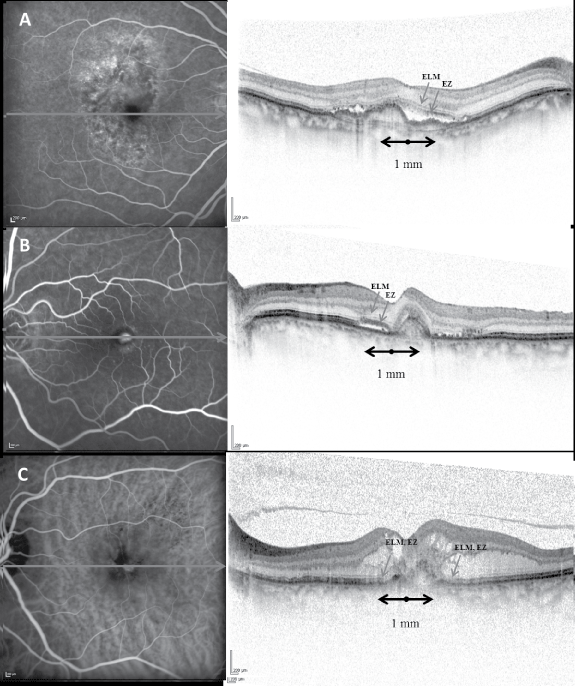
.
Figure 1: Classification of choroidal neovascularization (CNV) type, external limiting membrane (ELM) and ellipsoid zone (EZ) status in the central 1 mm centred on the fovea at baseline.
A) Simultaneous fluorescein angiography (FA) and SD-OCT demonstrates a type 1 (sub-RPE) CNV with subretinal fluid, present ELM and EZ. The line on FA image represents the B-scan location;
B) Simultaneous FA and SD-OCT show a type 2 (above the RPE) CNV with reflective material and fluid in the subretinal space. Note that the ELM and the EZ can be detected only in the nasal half of the macula (classified as "disturbed");
C) Indocyanine green angiography and corresponding SD-OCT scan demonstrates a type 3 (intraretinal neovascularization or retinal angiomatous proliferation) CNV with cystoid spaces and intraretinal reflective material. Both the ELM and EZ are absent and cannot be detected in the central 1 mm.
View Figure 1
All BCVAs were converted to logMAR equivalents, and visual acuity was expressed as logMAR units. Inter-grader reliability for the division of each group was assessed with k statistics and turned out to be excellent, (0.78 for the status of the EZ, 0.79 for the status of the ELM and 0.76 for the CNV type). Intraclass correlation coefficient of FTH was 0.88 and this parameter was included in the study by averaging the measurements of the two graders.
Statistical analysis
Results are expressed as mean and standard deviation if variables are continuous, and as absolute number or a percentage if variables are categorical. The Shapiro-wilk test was used to test the normality of the continuous variable. A Backward Stepwise Regression model was used to choose the best subset predictor variables of BCVA at month 3 with significance level for removal from the model equal to 0.01 (pr parameter) and significance level for addition to the model equal to 0.05 (pe parameter).
Interclass correlation and Cohen's K statistics were used to measure absolute agreement and consistency of agreement. A p-value < 0.05 was considered statistically significant. Analyses were performed using STATA version 12.0 (StataCorp, College Station, TX, USA).
Results
Seventy patients (27 men, 43 women) ranging in age from 56 to 95 years (mean and standard deviation, 78 and 8 years) were included in the study. Baseline characteristics and BCVA outcomes are shown in table 1. The CNV subtypes included 28 cases of type 1 CNV (40%), 17 cases of type 2 CNV (24%) and 25 cases of type 3 CNV (37%). The mean BCVA (SD) at baseline was 0.41 (0.2) and at 3 months was 0.32 (0.25). In total, 39 (56%) patients showed a clinically relevant increase of BCVA (>= 0.1 logMAR) 4 weeks after the third injection.
![]()
Table 1: Baseline characteristics and visual acuity outcomes.
View Table 1
In 28 (40%) and 16 (23%) eyes the ELM and EZ were present at baseline, respectively (Table 2). 81% of eyes with a present EZ also had a present ELM, whereas only 46% of eyes with present ELM had a present EZ. In all but one eye with absent ELM a disintegration of the EZ was noted. Four weeks after the third injection, in eyes with baseline present or disturbed ELM, the EZ became visible in more than half of the cases where it had been absent (10/16 eyes), whereas it remained undetectable in most eyes with pre-treatment absent ELM (10/14 eyes) (Table 3).
![]()
Table 2: Relationship between the status of the ELM and the EZ at baseline.
View Table 2
![]()
Table 3: Relationship between the change in the status of EZ after treatment and the integrity of ELM at baseline.
View Table 3
A stepwise regression analysis revealed that pretreatment BCVA and ELM present were the only significant predictors (Table 4). Age (P = 0.9106), sex (P = 0.2183), CNV type (P = 0.2375), EZ (P = 0.6023) and FTH (0.7450) showed no association with the final BCVA and were removed from the model. However, since the best predictor of final BCVA was pre-treatment BCVA, a new model of regression analysis stratified by the status of pretreatment ELM (present, disturbed and absent) was performed. The predictive value of baseline BCVA based on the integrity of the ELM may be assessed using the following equations:
![]()
Table 4: Stepwise regression, final model.
View Table 4
- ELM absent: predicted BCVA = 0.94 X pretreatment BCVA
(CI 95% = [0.73-1.15]; p-value < 0.001; adj R2 = 0.87)
- ELM disturbed: predicted BCVA = 0.74 X pretreatment BCVA
(CI 95% = [0.58-0.90]; p-value < 0.001; adj R2 = 0.75)
- ELM present: predicted BCVA = 0.61 X pretreatment BCVA
(CI 95% = [0.46-0.77]; p-value < 0.001; adj R2 = 0.68)
With this optimized model, the level of ELM integrity pre-treatment strongly affects the predictive value of pre-treatment BCVA; the change in BCVA post-treatment can vary from no vision gain if the ELM is absent to substantial gain if the ELM is present. In other words, if baseline BCVA is 20/50, after three intravitreal ranibizumab injections, vision may remain unchanged if the ELM is absent at baseline; improve to 20/40 if the ELM is disturbed or reach 20/32 if it is present. Three cases exemplifying the three different ELM categories are presented to show the influence of baseline ELM integrity on the predictive value of baseline vision (Figure 2). Moreover, these regression equations state that 87%, 75% and 68% of the variation in BCVA at month 3 is accounted for by the variation of baseline BCVA in the three different levels of pre-treatment ELM integrity. The effect of pre-treatment BCVA on visual response to the treatment in these three different prognostic subsets is shown in figure 3. None of these results could be obtained for the levels of pretreatment EZ (data not shown).
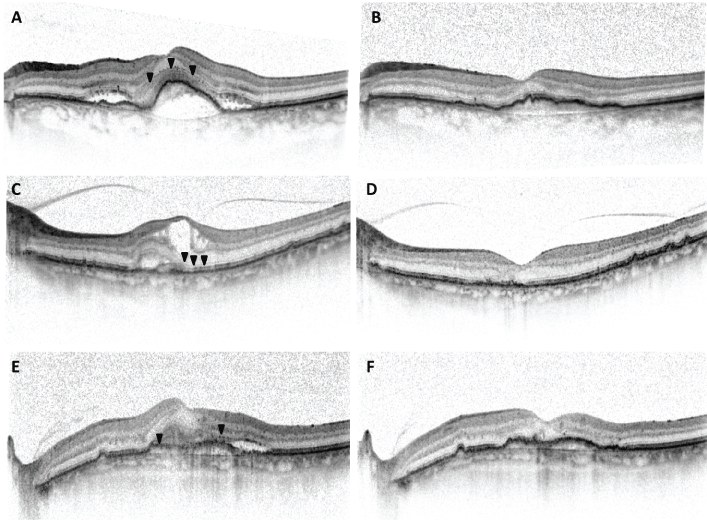
.
Figure 2: SD-OCT sections from three cases with similar baseline BCVA and different levels of external limiting membrane (ELM) integrity at baseline.
A) 82-year-old female with baseline BCVA of 20/50 and present ELM (arrowheads) in the central 1000 microns;
B) BCVA improved to 20/32 following 3 monthly intravitreal Ranibizumab injections;
C) 81-year-old male with baseline BCVA of 20/40 and a SD-OCT section revealing an ELM disturbed (arrowheads) and visible only in the nasal half of the central 1 mm;
D) BCVA improved to 20/32 (1 line only) following a Ranibizumab loading phase;
E) 73-year-old male with baseline BCVA of 20/40 presenting with completely absent ELM centrally (arrowheads);
F) After 3 intravitreal Ranibizumab, BCVA decreased to 20/50 despite complete resolution of subretinal material and fluid.
View Figure 2
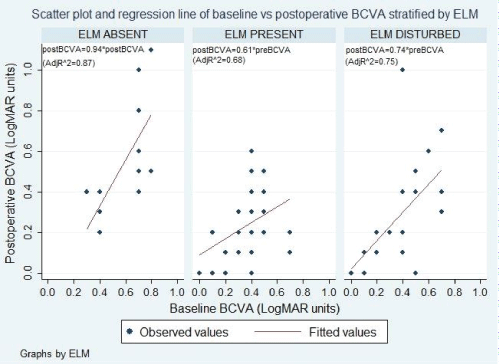
.
Figure 3: Graphs showing the effect of baseline BCVA on BCVA at month 3 for the three levels of pretreatment external limiting membrane (ELM) integrity.
View Figure 3
Discussion
Functional response to anti-VEGF therapy is highly variable in eyes with CNV secondary to wet AMD, even in the presence of complete resolution of the exudative change. The present study showed that correlations between different aspects of retinal anatomy (ELM, EZ, FTH, etc.) and visual acuity, even with advanced imaging modalities such as SD-OCT are weak if considered alone. The best baseline predictor of post-treatment vision is still baseline vision itself. However, the level of initial integrity of a particular tomographic layer, the ELM, appears to have great potential to stratify baseline vision into having a high or low chance of improvement.
In our study, the ELM was present in nearly half of the patients with active neovascular AMD, while the EZ was disturbed or absent in three out of four cases, and it reappeared in half of the patients after successful treatment. Therefore the ELM seems less sensitive to CNV activity, whereas the EZ may be temporarily disrupted by subretinal material (exudates, fibrin, and blood) in active disease. Moreover, the EZ recovers to a greater extent in the presence of a present ELM at baseline, suggesting that the integrity of this layer may be a better indicator of the reversibility of photoreceptor damage and the possibility of a full function recovery than the EZ. Oishi, et al. have recently described similar findings in a paper reporting the improvement of the IS/OS status – the former definition of the EZ - in response to anti-VEGF therapy [14].
Previous studies have investigated the relationship between morphology and function. Researchers noted a positive correlation between "post-treatment" ELM status and vision after PDT, and likewise between baseline ELM status and post-treatment IS-OS [12,13]. These observations prompted the researchers to further investigate the role of baseline ELM as a predictor of visual improvement after anti VEGF treatment. This aspect has been recently confirmed in two reports [5,6]. However, it should be noted that these studies found the degree of variation in vision associated with this morphologic feature to be modest, with an R2 value of 37% and 42%, respectively. Moreover, none of the studied features could predict any change in visual acuity from baseline. In addition, another study could find no correlation between baseline OCT parameters and post-treatment visual acuity [21].
Consequently, we propose a new approach that stratifies patients with similar baseline vision into subgroups based on the ELM status. In doing so, the amount of variation in post-treatment vision, predicted by the model, rises between 68% and 87%. Given the large proportion of patients with severe visual impairment at presentation, prior knowledge of predictive factors of visual improvement would prove very useful for patient counselling.
In this study, neither FTH nor the CNV type was correlated with final visual acuity. It seems that in neovascular AMD, the presence of substantial amounts of intraretinal fluid is not necessarily associated with final lower vision in eyes where the outer retina is preserved, while a thinner neurosensory retina at baseline may be a predictor of poor final vision due to atrophic changes. Similarly, a weak correlation between neurosensory retinal thickness and visual acuity was reported in earlier studies with the Stratus OCT - cross-sectional and longitudinal - following bevacizumab injection [2,22]. Even with the use of computer-assisted grading software to measure retinal morphology more precisely and reproducibly, the correlation was modest [23]. The authors of this study concluded that the complex pathophysiology of neovascular AMD might account for this finding. In apparent discordance, in 2013, a study evaluating baseline predictors for visual outcomes at one year in the Comparison of Age-related Macular Degeneration Treatments Trials (CATT), reported that thicker total foveal thickness, including subretinal fluid and subretinal tissue, was a significant predictor of worse vision at one year [18]. However, a further study on the same patients, analysing neuroretinal thickness independent from the subretinal fluid and subretinal tissue component, found not only thickened retinas but also thinned retinas to have worse visual acuity [3]. Furthermore, eyes with thinner than normal retinas tended to have worse vision than their thicker counterparts, suggesting a greater influence of neural tissue loss over intraretinal fluid on visual function.
The type of CNV, based on a combined tomographic and angiographic classification, also did not correlate with final vision. Earlier studies found that "occult" CNV had both increased vision and a better natural history compared to the "classic" [24]. More recent studies on the CATT participants have confirmed that the presence of a classic component is associated with worse vision at 1 year [18]. Thus, we would have expected type 2 CNV to be associated with poorer visual outcomes, due to a more extensive subretinal space involvement and direct photoreceptor damage. However, the lack of correlation could be due to the small number of eyes in each subgroup and to the inclusion of juxtafoveal type 2 CNV with central photoreceptor sparing by the subretinal tissue in addition to the short follow-up.
Our study has a number of limitations, as it is both retrospective and examines a small sample. Furthermore, OCT image was based on a personal judgement, rather than using more objective methods. The detection of the ELM and EZ may be have been compromised by overlying pathology, thus interfering with image interpretation. However, there was a high degree of inter-reader agreement in the grading of these two lines (P < 0.0001), suggesting a high reliability of this SD-OCT based classification. Lastly, visual acuity data were collected using Snellen charts in a clinical setting instead of Early Treatment Diabetic Retinopathy Study charts with protocol refraction. However, it has been recently reported that the discrepancy between these two measurements is more marked for acuities worse than 20/200, while our study included only two cases with vision below this range [25]. Thus we believe that our findings are not significantly influenced by Snellen results.
There are no other published studies evaluating the ELM as a stratifying biomarker of baseline visual acuity. Our results highlight the importance and clinical relevance of grading the integrity of this tomographic feature in order to better predict visual improvement for any given baseline vision. This study has shown that eyes with active neovascular AMD, similar pretreatment BCVA and various levels of ELM integrity, are likely to exhibit different functional treatment response to anti-VEGF therapy and confirm the importance of combining both imaging biomarkers and visual acuity in order to stratify patients into prognostic subsets. Moreover, this new approach provides more reliable prognostic information than that previously reported once based on BCVA or single OCT parameters and, as such, can be used for clinical purposes. It's worthy of attention that the group with absent ELM has the highest prognostic value (87%). This is the group in which the ability to predict the functional outcome can have the most practical implications.
References
-
Bressler NM (2004) Age-related macular degeneration is the leading cause of blindness. JAMA 291: 1900-1901.
-
Moutray T, Alarbi M, Mahon G, Stevenson M, Chakravarthy U (2008) Relationships between clinical measures of visual function, fluorescein angiographic and optical coherence tomography features in patients with subfoveal choroidal neovascularisation. Br J Ophthalmol 92: 361-364.
-
Jaffe GJ, Martin DF, Toth CA, Daniel E, Maguire MG, et al. (2013) Macular Morphology and Visual Acuity in the Comparison of Age-related Macular Degeneration Treatments Trials. Ophthalmology 120: 1860-1870.
-
Shin HJ, Chung H, Kim HC (2013) Correlation of foveal microstructural changes with vision after anti-vascular endothelial growth factor therapy in age-related macular degeneration. Retina 33: 964-970.
-
Mathew R, Richardson M, Sivaprasad S (2013) Predictive value of spectral-domain optical coherence tomography features in assessment of visual prognosis in eyes with neovascular age-related macular degeneration treated with ranibizumab. Am J Ophthalmol 155: 720-726.
-
Chhablani J, Kim JS, Freeman WR, Kozak I, Wang HY, et al. (2013) Predictors of visual outcome in eyes with choroidal neovascularization secondary to age related macular degeneration treated with intravitreal bevacizumab monotherapy. Int J Ophthalmol 6: 62-66.
-
Hayashi H, Yamashiro K, Tsujikawa A, Ota M, Otani A, et al. (2009) Association between foveal photoreceptor integrity and visual outcome in neovasculare age-related macular degeneration. Am J Ophhtalmol 148: 83-89.
-
Sayanagi K, Sharma S, Kaiser PK (2009) Photoreceptor status after anti-vascular endothelial growth factor therapy in exudative age-related macular degeneration. Br J Ophhtalmol 93: 622-626.
-
Yamaike N, Tsujikawa A, Ota M, Sakamoto A, Kotera Y, et al. (2008) Three-dimensional imaging of cystoid macular edema in retinal vein occlusion. Ophthalmology 115: e2.
-
Bottoni F, De Angelis S, Luccarelli S, Cigada M, Staurenghi G (2011) The dynamic healing process of idiopathic macular holes after surgical repair: a spectral-domain optical coherence tomography study. Invest Ophthalmol Vis Sci 52: 4439-4446.
-
Yalcinbayir O, Gelisken O, Akova-Budak B, Ozkaya G, Gorkem Cevik S, et al. (2014) Correlation of Spectral Domain Optical Coherence Tomography Findings and Visual Acuity in Central Serous Chorioretinopathy Retina 34: 705-712.
-
Oishi A, Hata M, Shimozono M, Mandai M, Nishida A, et al. (2010) The significance of external limiting membrane status for visual acuity in age-related macular degeneration. Am J Ophthalmol 150: 27-32.
-
Shin HJ, Chung H, Kim HC (2011) Association between foveal microstructure and and visual outcome in age-related macular degeneration. Retina 31: 1627-1636.
-
Oishi A, Shimozono M, Mandai M, Hata M, Nishida A, et al. (2013) Recovery of photoreceptor outer segment after anti-VEGF therapy for age-related macular degeneration. Graefes Arch Clin Exp Ophthalmol 251: 435-440.
-
Boyer DS, Antoszyk AN, Awh CC, Bhisitkul RB, Shapiro H, et al. (2007) MARINA Study Group. Subgroup analysis of the MARINA study of ranibi- zumab in neovascular age-related macular degeneration. Oph- thalmology 114: 246-252.
-
Kaiser PK, Brown DM, Zhang K, Hudson HL, Holz FG, et al. (2007) Ranibizumab for pre- dominantly classic neovascular age-related macular degeneration: subgroup analysis of first-year ANCHOR results. Am J Ophthalmol 144: 850-857.
-
Rosenfeld PJ, Shapiro H, Tuomi L, Webster M, Elledge J, et al. (2011) ANCHOR Study Group. Characteristics of patients losing vision after 2 years of monthly dosing in the phase III ranibizumab clinical trials. Ophthalmology 118: 523-530.
-
Ying GS, Huang J, Maguire MG, Jaffe GJ, Grunwald JE, et al. (2013) Baseline predictors for one-year visual outcomes with ranibizumab or bevacizumab for neovascular age-related macular degeneration. Ophthalmology 120: 122-129.
-
Bloch SB, la Cour M, Sander B, Hansen LK, Fuchs J, et al. (2013) Predictors of 1-year visual outcome in neovascular age-related macular degeneration following intravitreal ranibizumab treatment. Acta Ophthalmol 91: 42-47.
-
Freund KB, Zweifel SA, Engelbert M (2010) Do we need a new classification for choroidal neovascularization in age-related macular degeneration? Retina 30: 1333-1349.
-
Kolb S, Menghini M, Barthelmes D, Sutter F, Kurz-Levin M (2012) The predictive value of OCT characteristics for the visual outcome in neovascular AMD. Klinische Monatsblatter Fur Augenheilkunde 229: 343e7.
-
Unver YB, Yavuz GA, Bekiroğlu N, Presti P, Li W, et al. (2009) Relationship between clinical measures of visual function and anatomic changes associated with bevacizumab treatment for choroidal neovascularization in age-related macular degeneration. Eye (Lond) 23: 453-460.
-
Keane PA, Liakopoulos S, Chang KT, Wang M, Dustin L, et al. (2008) Relationship between optical coherence tomography retinal parameters and visual acuity in neovascular age-related macular degeneration. Ophthalmology 115: 2206-2214.
-
Bressler NM, Frost LA, Bressler SB, Murphy RP, Fine SL (1988) Natural course of poorly defined choroidal neovascularization associated with macular degeneration. Arch Ophthalmol 106: 1537-1542.
-
Falkenstein IA, Cochran DE, Azen SP, Dustin L, Tammewar AM, et al. (2008) Comparison of visual acuity in macular degeneration patients measured with Snellen and Early Treatment Diabetic Retinopathy Study charts. Ophthalmology 115: 319-323.





