International Journal of Ophthalmology and Clinical Research
Introduction of a Toric Intraocular Lens to a Non-Refractive Cataract Practice: Challenges and Outcomes
Clare Kirwan1,2,3*, John M Nolan3, Jim Stack3, Ian Dooley4, Johnny Moore1, Tara CB Moore1 and Stephen Beatty2,3
1Biomedical Science Research Institute, University of Ulster, Coleraine, Northern Ireland
2Institute of Eye Surgery, and Institute of Vision Research, Whitfield Clinic, Cork Road, Waterford, Ireland
3Macular Pigment Research Group, Waterford Institute of Technology, Waterford, Ireland
4University College Hospital Limerick, Ireland
*Corresponding author: Clare Kirwan, Macular Pigment Research Group, Carriganore House, Waterford Institute of Technology, Waterford, Ireland, Tel: 00353879197351, E-mail: ckirwanabroad@yahoo.com
Int J Ophthalmol Clin Res, IJOCR-3-056, (Volume 3, Issue 2), Research Article; ISSN: 2378-346X
Received: November 19, 2015 | Accepted: May 23, 2016 | Published: May 25, 2016
Citation: Kirwan C, Nolan JM, Stack J, Dooley I, Moore J, et al. (2016) Introduction of a Toric Intraocular Lens to a Non-Refractive Cataract Practice: Challenges and Outcomes. Int J Ophthalmol Clin Res 3:056. 10.23937/2378-346X/1410056
Copyright: © 2016 Kirwan C, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Aim: To identify challenges inherent in introducing a toric intraocular lens (IOL) to a non-refractive cataract practice, and evaluate residual astigmatism achieved and its impact on patient satisfaction.
Methods: Following introduction of a toric IOL to a cataract practice with all procedures undertaken by a single, non-refractive, surgeon (SB), pre-operative, intra-operative and post-operative data was analysed. Attenuation of anticipated post-operative astigmatism was examined, and subjectively perceived visual functioning was assessed using validated questionnaires.
Results: Median difference vector (DV, the induced astigmatic change [by magnitude and axis] that would enable the initial surgery to achieve intended target) was 0.93D; median anticipated DV with a non-toric IOL was 2.38D. One eye exhibited 0.75D residual astigmatism, compared to 3.8D anticipated residual astigmatism with a non-toric IOL. 100% of respondents reported satisfaction of ≥ 6/10, with 37.84% of respondents entirely satisfied (10/10). 17 patients (38.63%) reported no symptoms of dysphotopsia (dysphoptosia score 0/10), only 3 respondents (6.8%) reported a clinically meaningful level of dysphotopsia (≥ 4/10). Mean post-operative NEI VF-11 score was 0.54 (+/-0.83; scale 0 - 4).
Conclusion: Use of a toric IOL to manage astigmatism during cataract surgery results in less post-operative astigmatism than a non-toric IOL, resulting in avoidance of unacceptable post-operative astigmatism.
Keywords
Cataract surgery, Visual outcomes, Refractive outcomes, Surgical outcomes, Satisfaction, Dysphotopsia, Tecnis® toric intraocular lens, Rotational stability, Astigmatism reduction, Non-refractive surgeon
Introduction
Uncorrected astigmatism can be visually debilitating and, as thresholds for cataract surgery fall [1], the implications of this procedure for astigmatism cannot be ignored. The European Eye Epidemiology Consortium [2] found astigmatism in 27.3% of eyes, rising to 51.1% of eyes in those aged 80-84. Ferrer-Blasco et al. [3] reported astigmatism ≤ 1.25D, and ≥ 1.50D, in 64.4%, and 22.2%, of subjects undergoing cataract surgery, respectively.
Astigmatism can be managed intra-operatively by tailored incision axis, clear corneal incisions [CCIs], limbal relaxing incisions [LRIs] or implantation of a toric intra-ocular lens (IOL), or post-operatively (LRIs/refractive laser). Non-toric IOL methods are limited in the degree of treatable astigmatism and may be unpredictable due to age, healing properties and surgeon skill [3].
Approximately 70% of U.S. cataract surgeons do not perform refractive procedures. Patient profile, priorities and outcome measures for non-refractive cataract surgeons differ greatly from their refractive colleagues, with non-refractive surgeons primarily concerned with the avoidance of unacceptable post-operative astigmatism, while refractive surgeons target elimination of astigmatism.
For non-refractive cataract surgeons, toric IOLs are the most appropriate surgical means of reducing astigmatism during cataract surgery: tailoring incision axis is avoided, eliminating human error and precluding awkward surgical position; refractive skills are unnecessary; medical indemnity for refractive procedures and expensive refractive surgical equipment are not required.
We examine difficulties encountered when introducing a toric IOL to a non-refractive cataract practice, reporting visual, surgical, refractive and self-reported outcomes, in order to identify challenges and to evaluate residual astigmatism achieved and its impact on patient satisfaction.
Materials and Methods
We adhered to the Declaration of Helsinki, and the local ethics committee (Research Ethics Committee, Health Service Executive, South Eastern Area, Ireland) gave approval. The Tecnis® Toric ZCT 1-piece toric intraocular lens (ZCT) [Advanced Medical Optics Inc, Santa Ana, CA, USA] was introduced to the single surgeon (SB) practice (Institute of Eye Surgery [IoES], Whitfield Clinic) in June 2011. Patients implanted with the ZCT until May 2014 were identified from the electronic medical records (EMR; Medisoft Ophthalmology Version 5.1.2;), and included in this study.
Patients were deemed suitable for, and offered, a toric IOL under the following conditions:
1) visual potential ≥ 20/40;
2) anticipated post-operative corneal cylinder ≥ 1.5D;
3) with a pseudophakic fellow eye, use of a toric IOL will not result in astigmatic anisometropia [4].
Satisfaction of these criteria was determined by inputting keratometry readings and anticipated surgically induced astigmatism [5] into the Alcon website (www.acrysoftoriccalculator.com), which outputs anticipated post-operative corneal astigmatism were a non-toric IOL to be used. The Tecnis® toric calculator (www.tecnistoriccalc.com) was used to determine the appropriate ZCT to minimise post-operative corneal astigmatism while avoiding axis flip.
The 180° meridian was marked (patient upright) using a Bakewell Bubble Level marker (Mastel, Inc.). Using this pre-marked reference 0-180° axis, the alignment axis of the toric IOL was marked (patient supine) before making the corneal incision.
Procedures were performed using standard technique, [5] with review two weeks following an uneventful procedure [6]. Post-operative data included: post-operative complications; refractive status (auto-refraction, and best corrected subjective refraction performed by the patient's optometrist [four weeks post-operatively, and at least two weeks following removal of any corneal suture]).
Visually consequential ocular co-morbidity is associated with reduced self-assessed visual function post-cataract surgery [7]. These eyes were, therefore, excluded in analysis of subjectively perceived outcomes. Remaining patients were invited to complete two validated questionnaires, [8] one designed to assess impact of surgery on subjectively perceived visual functioning, including satisfaction, the second to identify dysphotopsia symptoms. Separate questionnaires were answered for each individual operated eye.
Statistical analysis
Snellen notation was converted to visual acuity rating (VAR) [9]. 20/20 was assigned a score of 100 with each correct letter given a nominal value of 1, giving 20/30 a score of 90, 20/40 a score of 85, etc. Our EMR cannot record additional letters on the next line or missed letters on an almost complete line (e.g. 20/20 +/-1), so the best complete line was recorded.
As this study was retrospective, acuity measurements were not consistent across all eyes, visual acuity was recorded (pre and post-operatively) under at least one of the three following conditions: unaided (UA); best corrected (BC); pinhole (PH). In cases of sub-optimal UAVA, BCVA and/or PHVA were tested. Where the same VA measurement (UA, PH or BC) was taken pre- and post-operatively, paired measurements were analysed, but the term Optimum VA (OptVA) was adopted to define best recorded VA (UA, PH or BC) to compare pre and post-operative acuity.
Refraction is expressed as sphere, cylinder and axis, the astigmatic component denoted by magnitude (dioptres [D]) and direction (degrees). Examining purely astigmatic magnitude, reduction in astigmatism resulting from implantation of a toric IOL can be easily quantified by comparing pre- and post-operative refraction for a particular eye.
To study large numbers of procedures, however, standard arithmetic analysis of cylindrical magnitude does not quantify the direction component of the astigmatism. Vector analysis treats cylinder as a vector with magnitude and direction, expressing refractive error as sphere/cylinder X-axis, allowing comparison of multiple vectors [5].
In order to compare pre-operative astigmatism with actual post-operative astigmatism, and with anticipated post-operative astigmatism should a non-toric IOL be used, we employed Vector Analysis, using the Alpins method [10,11].
Alpins Terminology:
1. Target induced astigmatism vector (TIA): intended astigmatic change (magnitude and axis);
2. Surgically induced astigmatism vector (SIA): actual astigmatic change (magnitude and axis);
3. Correction index (CI): ratio of SIA to TIA - preferably 1.0 (> 1.0 with over-correction, < 1.0 with under-correction);
4. Difference Vector (DV): astigmatic change (by magnitude and axis) that would enable the initial surgery to achieve its intended target - an absolute measure of success, preferably zero;
5. Index of success (IOS): calculated by dividing DV by TIA - a relative measure of success, preferably zero; [11]
Spectacle plane refraction was converted to corneal plane using:
Fc = lens power (D), corneal plane; Fs = lens power (D), spectacle plane;
d = vertex distance (mm) [12].
As correlation between fellow eyes in relation to refractive outcome is weak in terms of prediction error (PE; actual post-operative spherical equivalent [SE] minus target post-operative spherical equivalent [target SE]) and visual outcome, [13] where bilateral surgery was performed, each eye was analysed independently for these variables (SPSS [Version 20; IBM Corp Somers, NY]). For analysis of post-operative satisfaction, dysphotopsia, and function related to vision, however, one cannot assume right and left eyes of the same patient are truly independent. We, therefore, performed linear mixed model analysis (Random intercept model from the class of linear mixed models, package NLME; statistical programming language R [14]).
As analysis of satisfaction scores was limited by the small study size (42 eyes of 35 subjects), data was analysed one variable at a time. Furthermore, we simplified spectacle dependence by combining adjacent categories, to ensure sufficient data for meaningful statistical analysis - see Results.
Although Kinard et al. [8] Rasch-analysed questionnaire scores, we treated questionnaire items as equally important, summing and averaging with equal weight. We believe all dysphotopsia symptoms, and all aspects of visual function, are equally important and cannot assign greater weight to one over another.
Results
154 procedures with implantation of a ZCT IOL were performed during the study period.
Procedures were grouped as follows:
Group 1 (n = 72 [46.7%]: eyes exhibiting pre-operative visually consequential ocular co-morbidity.
Group 2 (n = 82 [53.9%]): eyes without pre-operative visually consequential ocular co-morbidity, these received the questionnaires.
Group 3 (n = 14 [9.1%]): eyes which experienced a post-operative complication, most commonly pseudophakic cystoid macular oedema (CMO; 7 procedures [4.5%]). No intra-operative complications occurred.
Refractive and visual results
Refractive results were available for 136 cases (88.3%). PE ranged from -1.2D to 1.77D; mean absolute PE was 0.27D (+/-0.36D), with 97.8% and 72.1% of eyes exhibiting absolute PE of ≤ 1D and ≤ 0.5D, respectively.
![]()
Table 1A: Measures of post-operative visual acuity, and change with respect to respective pre-operative measures of visual acuity.
View Table 1A
Visual results for the three groups, and statistical significance of observed changes from pre-operative VA, are given in table 1 (a) (p-values from paired sample t-tests). All measures of visual acuity improved for each group, except PHVA in Group 1 (5 eyes). Not all observed changes reached statistical significance, likely due to small numbers.
Self-reported post-operative results
123 procedures completed before December 1st, 2013 were examined in relation to self-reported post-operative results. 57 (45.5%) exhibited visually consequential ocular co-morbidity pre-operatively, and the remainder (66 [53.65%]) were invited to complete two questionnaires, and a satisfaction question, for each operated eye. 42 completed questionnaire sets (78.8%) were returned, representing 35 subjects. Table 1 (b) shows acuity changes for this cohort.
![]()
Table 1B: Measures of post-operative visual acuity, and change with respect to respective pre-operative measures of visual acuity for eyes with no pre-operative ocular co-morbidity, and who completed the questionnaires.
View Table 1B
Self-reported questionnaire scores
Three self-reported scores were analysed relative to other variables:
Satisfaction score: 35 respondents rated post-operative satisfaction 0 to 10, 10 representing complete satisfaction. Minimum satisfaction score was 6 (3 respondents [8.1%]). Fourteen respondents (37.84%) were entirely satisfied (10/10), with 10 (27.03%), 8 (21.6%) and 2 (5.4%) respondents reporting scores of 9/10, 8/10 and 7/10, respectively.
Dysphotopsia score: Pseudophakic dysphotopsia questionnaire (PDQ) Likert scale scores were averaged, higher scores representing poorer visual outcomes. Mean PDQ score was 1.5 (+/-2.2; maximum 10). 17 patients (38.63%) reported no symptoms of dysphotopsia (PDQ score 0); only 3 respondents (6.8%) reported clinically meaningful dysphotopsia (PDQ score ≥ 4/10).
Functionality score: Functionality questionnaire (NEI VF-11) Likert scale scores were averaged, higher scores indicating greater visual difficulty. Mean NEI VF-11 score was 0.54 (+/-0.83; maximum 4). Eleven respondents (26.2%) reported no dysfunction related to vision (NEI VF-11 score 0), 26 respondents (61.9%) had a score ≤ 1, and 3 respondents (7.1%) reported a score of ≥ 3.
Satisfaction score and other study variables
Age and satisfaction score: Age was the only statistically significant variable related to satisfaction score (p = 0.048, linear mixed model); younger subjects reported higher mean satisfaction score, and satisfaction score declined with increasing age. In the 37-66 age group, mean satisfaction score was 9.3 (+/-1.3), compared to 8.6 (+/-1.3) and 8.5 (+/-1) in the 67-72 and 73+ age groups, respectively.
Spectacle dependence and satisfaction score: Two patients reported complete spectacle independence (following bilateral surgery aimed at mini-monovision). One did not answer the individual satisfaction question, whereas the other reported total satisfaction (10/10). The first two categories were combined (18 subjects; “low” dependence, requiring spectacles for ≤ 1 viewing distance), as were the remaining two categories (17 subjects; “high” dependence, requiring spectacles for ≥ 2 distances). Satisfaction score was not significantly different (p = 0.21) for these categories.
Dysphotopsia and satisfaction score: To grade incidence and severity of pseudophakic dysphotopsia, we categorised ranges of scores (Table 2), showing some (statistically insignificant; p = 0.54) reduction in satisfaction with increasing PDQ score.
![]()
Table 2: Categories of dysphotopsia scores, and mean satisfaction score within each category.
View Table 2
Functionality and satisfaction score: Excepting lower satisfaction score in the “severe” category, no clear relationship is evident; statistically, visual functionality score was not significantly related to satisfaction score (p = 0.11).
Change in visual acuity and satisfaction score: Due to insufficient data for different measures of visual acuity, this part of the analysis was restricted to surgically-induced change in optimal visual acuity (OptVA). Statistically, this change was not significantly related to satisfaction score (p = 0.58).
Complications and satisfaction score: 7 of the 14 eyes in which a complication occurred had no visually consequential ocular co-morbidity, and were invited to complete the questionnaires; only three sets of questionnaires were received in relation to these eyes (of 2 patients). While no statistically significant correlation between complication and satisfaction score can be inferred, the first patient rated satisfaction at 8, while the second, in whom a complication occurred in each eye (corneal oedema, which resolved) reported satisfaction of 10/10 for each eye.
Satisfaction score and absolute prediction error (PE): The relationship between satisfaction score and absolute PE was not statistically significant (p = 0.22).
Vector analysis and reduction in astigmatism
Mean (± SD) absolute pre-operative astigmatism was 2.16 ± 1.25 D, mean absolute post-operative astigmatism was 0.97 ± 0.58 D and mean absolute target residual astigmatism was 0.26 ± 0.14 D.
Table 3 shows the vector which describes the difference between the actual post-operative result (SIA) and predicted residual astigmatism without a toric lens (DVNT; difference vector, no toric), calculated by subtracting the cylinder due to the toric IOL from the post-operative cylinder. The CI ratio shows that 1.17 of the TIA was achieved (ideal = 1.0), a slight overcorrection. The DVNT indicates a median of 2.38 D of astigmatism at 110 degrees would be required to undo the astigmatism due to the toric IOL.
![]()
Table 3: Summary of vector analysis post toric intraocular lens implantation.
View Table 3
Figure 1 shows vector plots of (a) pre-operative astigmatism, (b) post-operative astigmatism (c) predicted post-operative astigmatism, (d) surgically induced astigmatism (SIA), (e) target induced astigmatism (TIA), (f) difference vector (DV) and (g) difference vector no toric (DVNT). The origin (0.0) represents an astigmatism-free eye.
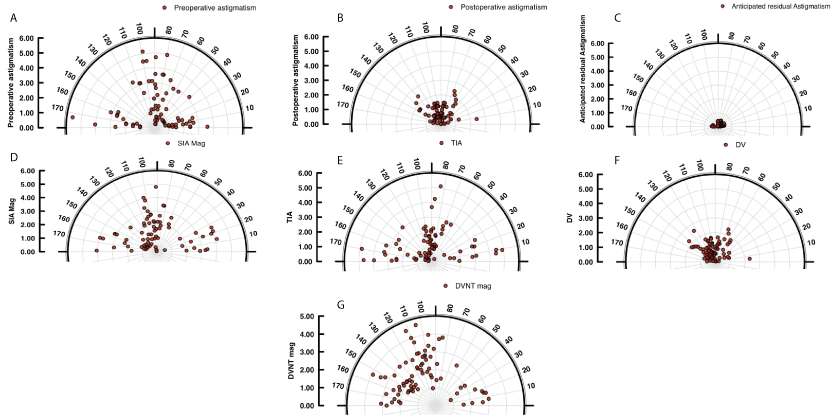
.
Figure 1: Shows vector plots of (a) pre-operative astigmatism, (b) post-operative astigmatism (c) predicted post-operative astigmatism, (d) surgically induced
astigmatism (SIA), (e) target induced astigmatism (TIA), (f) difference vector (DV) and (g) difference vector no toric (DVNT) for 77 eyes in this series.
View Figure 1
Discussion
As patient expectations rise, the refractive element of cataract surgery is increasingly important and toric IOL use becomes an integral component of standard practice.
Waltz [15] and Sheppard [16] report post-operative UAVA ≥ 85 (20/40) in 97.1% of 172 eyes (without pre-existing visually consequential ocular co-morbidity), and 87.7% of 67 eyes (including non-visually consequential ocular co-morbidity/subtle amblyopia) implanted with the ZCT, respectively. Excluding eyes with visually consequential ocular co-morbidity, our figure is 91.4%.
BCVA ≥ 85 (20/40) was seen in 95.4% of eyes in the Sheppard series, [16] comparable with OptVA ≥ 85 (20/40) in 98.8% of eyes without visually consequential ocular co-morbidity in this series.
In this study, 97.8% of operated eyes exhibited a PE ≤ 1D, comparing favourably with published findings [16-18].
Waltz [15] and Sheppard [16] report mean (± SD) pre-operative astigmatism of 1.94D (± 1.01D; range not reported) and -2.21D (± 0.91D; range -0.78D to -5.55D), respectively, and mean (± SD) cylinder reduction of 75.24% (± 59.29%) and 1.24D (± 1.2D), respectively. We report a comparable mean (± SD) absolute pre-operative astigmatism of 2.16D (± 1.25D; range 0.25D to 6.0D) and mean (± SD) cylindrical reduction of 2.19D (± 1.13D). Observed mean absolute post-operative astigmatism of 0.97D (± 0.58D; range 0D to 2.5D) in this study compares to the findings of Waltz [15] and Sheppard [16] of 0.67D (± 0.47D; range not reported) and -0.67D (± 0.54D; range 0D to -2.25D), respectively.
In order to measure mis-alignment of a toric intra-ocular lens (attributable to poor operative alignment and/or post-operative rotation of the implanted IOL), alignment must be assessed on at least two occasions post-operatively. In compliance with published protocol of this busy non-refractive cataract practice, [6] patients were reviewed 2 weeks post-operatively, without assessment of IOL alignment. Accordingly, we cannot comment on ZCT alignment in this series. The ZCT has been shown to surpass the stability requirements of the American National Standards Institute (≤ 5° axis rotation between two consecutive visits, at least three months apart, in at least 90% of toric IOLs), reflected in the findings of Waltz [15] Sheppard [16] Mazzini [19] and Hirnschall [20] (mean misalignment 2.7° - 3.6°), and comparable to results reported for the Acrysof Toric™ IOL [20]. Nevertheless, non-assessment of alignment of the implanted toric IOL represents a weakness of this study.
In a non-refractive cataract practice, the aim is avoidance of unacceptable post-operative astigmatism; the current series demonstrates that it is possible to achieve a substantial reduction in post-operative astigmatism compared with the use of a non-toric IOL, reflected in the mean (± SD) reduction of astigmatism of 2.19D (± 1.13). Careful patient selection resulted in satisfied patients, reflected in a minimum satisfaction score of 6/10, with 37.8% of respondents entirely happy (10/10), and 91.9% rating satisfaction as ≥ 7/10, indicating a subjectively perceived benefit following implantation of this toric IOL and consistent with recently published and favourable findings following implantation of the monofocal (non-toric) version of the ZCT [7].
Waltz [15] reports 88.8% of patients were satisfied/very satisfied following implantation of the ZCT, comparable to our findings of 37.8% and 48.6% reporting satisfaction of 10/10 and 8/10 or 9/10, respectively. Sheppard [16] reported satisfaction only in relation to post-operative UAVA following ZCT implantation, with 37.9% and 55.2% of patients reporting a satisfaction score of 5/5 and 4/5, respectively.
Only 8% of our respondents reported symptoms consistent with clinically meaningful dysphotopsia; average satisfaction score of this group was 8.67. Further, despite completion of two questionnaires, we report no intolerance to post-operative refraction. This finding is consistent with recently published findings following implantation of the monofocal (non-toric) version of the ZCT in a single-surgeon series of > 2,500 eyes, where clinically meaningful dysphotopsia was less prevalent than alternative models of IOL [7].
DV is an absolute measure of success, preferably zero; indicating the induced astigmatic change (by magnitude and axis) which would enable the initial surgery to achieve its intended target; median DV was 0.93D. DVNT (the anticipated difference vector resulting from a non-toric IOL) was 2.38D, indicating a large reduction in astigmatism achieved versus using a non-toric IOL.
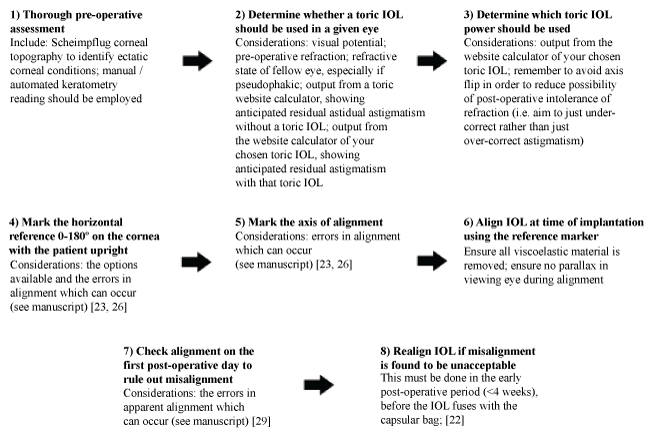
Introducing toric IOLs is relatively straightforward, even for a surgeon unskilled in refractive procedures; nevertheless, there are some pitfalls. Figure 2 shows factors to consider before embarking on this path.

.
Figure 2: Points to consider before introducing a toric IOL to a non-refractive cataract practice.
View Figure 2
Mild to moderate irregular astigmatism, satisfactorily correctable with spectacles and unlikely to progress, may be reduced using a toric IOL [21]. Scheimpflug imaging is advisable to exclude ectatic corneal disorders resulting in irregular astigmatism not correctable with a toric IOL [21].
Thresholds for toric IOL implantation should be raised where corneal pathology could result in corneal decompensation, e.g. Fuch's corneal dystrophy, and in eyes with zonular weakness (e.g. trauma, pseudoexfoliation), because of risk of rotation and/or decentration [21]
Previous studies typically report a lower limit of pre-operative corneal astigmatism (0.75D - 1D) as the sole criterion in determining eligibility for implantation of a toric IOL [15-17,19]. In non-refractive practice, the decision to implant a toric IOL should centre on acceptability of anticipated post-operative astigmatism, with analysis of pre-operative, anticipated post-operative, and to-be-surgically-induced astigmatism. Should a non-toric IOL result in 1.6D post-operative corneal cylinder compared to 2.3D pre-operative corneal cylinder, this represents a substantial improvement in refractive state, without a toric IOL. Similarly, should an eye with little pre-operative corneal astigmatism develop corneal astigmatism as a result of surgery, a toric IOL might indeed be offered.
The American National Standards Institute advises a method of guiding toric IOL axis alignment which corrects for head tilt and/or cyclotorsion, making reference to fixed anatomical features. Three steps are required: marking the horizontal 0-180° reference (patient upright); marking the alignment axis (patient supine); and alignment of the toric IOL axis with reference markings.
Popp [22] evaluated 4 common marking methods, concluding that the bubble marker method was easiest to master. Several sources of error can contribute to deviation from intended axis of alignment. Cyclotorsion of a given eye can vary; Visser [21] reports mean (± SD) test-retest variability of 1.5° (±1.2°), while Viestenz [23] reports mean (± SD) test-retest variability of 2.3° (±1.7°). Visser [24] reports errors in the limbal reference markings using a bubble level marker can contribute to mean (± SD) misalignment of 2° (± 1.8°), and that errors in the marking of the alignment axis relative to the reference marks can contribute to mean (± SD) misalignment of 3.3° (± 2°). Error in toric IOL positioning in relation to the marked alignment axis can contribute to mean (± SD) misalignment of 2.6° (± 2.6°) [24].
With recent availability of higher spherical and cylindrical IOLs, the importance of alignment accuracy increases. Longer eyes (generally requiring non-standard IOL powers) tend to demonstrate greater IOL rotation, especially in the early post-operative period. Miyake et al. [25] found rotation ≥ 20° in 6 eyes with axial length > 25 mm, due, they believe, to large capsular bags. The higher the cylindrical power, the greater the adverse impact caused by misalignment [24].
Despite perfect alignment of toric IOLs, unexpected residual astigmatism of nearly 0.4D [26] can result from a variety of sources, including pre-operative keratometry and post-operative refraction, and to errors in estimation of the effective cylindrical power of the IOL at the corneal plane.
In a given practice, the accuracy of axis alignment must be monitored over time to identify systematic errors in pre-operative assessment (keratometry readings, axial length etc.), toric IOL calculation, reference marking or alignment. It is equally important to ensure post-operative alignment is assessed accurately, to eliminate false-positives (correctly aligned IOL appears rotated) and false-negatives (rotated IOL appears correctly aligned) [27]. Slit lamp estimation of the axis of alignment by rotating a slit beam and aligning with a graticule is a simple and time-efficient technique, but is prone to error. Alignment can be analysed using vector analysis of post-operative refraction and keratometry readings, anterior segment optical coherence tomography, wavefront aberrometry or various digital overlays [28].
Figure 3 outlines some key points in optimal toric IOL implantation.
Conclusion
Toric IOLs are an excellent means for a non-refractive surgeon to avoid unacceptable post-operative astigmatism, resulting in substantial reduction in post-operative astigmatism relative to that which would occur were a toric IOL not implanted. Toric IOLs can be introduced safely to a non-refractive practice with minimal effort, provided certain pitfalls are avoided.
Acknowledgements
This study was funded by Abbott Medical Optics, Germany.
Conflict of Interest
All authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest (such as honoraria; educational grants; participation in speakers' bureaus; membership, employment, consultancies, stock ownership, or other equity interest; and expert testimony or patent-licensing arrangements), or non-financial interest (such as personal or professional relationships, affiliations, knowledge or beliefs) in the subject matter or materials discussed in this manuscript.
Ethical Statement
This study adhered to the tenets of the Declaration of Helsinki, and the local ethics committee (Research Ethics Committee, Health Service Executive, South Eastern Area, Ireland) gave approval as it represents clinical audit and, therefore, best clinical practice.
References
-
Dell S (2007) Screening and evaluating presbyopic patients. Cataract Refract Surg Today 81-82.
-
Williams KM, Verhoeven VJ, Cumberland P, Bertelsen G, Wolfram C, et al. (2015) Prevalence of refractive error in Europe: the European Eye Epidemiology (E(3)) Consortium. Eur J Epidemiol 30: 305-315.
-
Ferrer-Blasco T, Montés-Micó R, Peixoto-de-Matos SC, González-Méijome JM, Cerviño A (2009) Prevalence of corneal astigmatism before cataract surgery. J Cataract Refract Surg 35: 70-75.
-
Soo Han Lee, Ji Woong Chang (2014) The Relationship between Higher-order Aberrations and Amblyopia Treatment in Hyperopic Anisometropic Amblyopia. Korean J Ophthalmol 28: 66-75.
-
Dooley I, Charalampidou S, Malik A, Ormonde G, Loughman J, et al. (2010) Surgically induced astigmatism after phacoemulsification with and without correction for posture-related ocular cyclotorsion: randomized controlled study. J Cataract Refract Surg 36: 413-417.
-
Saeed A, Guerin M, Khan I, Keane P, Stack J, et al. (2007) Deferral of first review after uneventful phacoemulsification cataract surgery until 2 weeks: randomized controlled study. J Cataract Refract Surg 33: 1591-1596.
-
Kirwan C, Nolan JM, Stack J, Moore TC, Beatty S (2015) Determinants of patient satisfaction and function related to vision following cataract surgery in eyes with no visually consequential ocular co-morbidity. Graefes Arch Clin Exp Ophthalmol 253: 1735-1744
-
Kinard K, Jarstad A, Olson RJ (2013) Correlation of visual quality with satisfaction and function in a normal cohort of pseudophakic patients. J Cataract Refract Surg 39: 590-597.
-
Bailey IL, Lovie-Kitchin JE (2013) Visual acuity testing. From the laboratory to the clinic. Vision Res 90: 2-9.
-
Alpins N (2001) Astigmatism analysis by the Alpins method. J Cataract Refract Surg 27: 31-49.
-
Visser N, Berendschot TT, Bauer NJ, Nuijts RM (2012) Vector analysis of corneal and refractive astigmatism changes following toric pseudophakic and toric phakic IOL implantation. Invest Ophthalmol Vis Sci 53:1865-1873.
-
Novis C (2000) Astigmatism and toric intraocular lenses. Curr Opin Ophthalmol 11: 47-50.
-
Charalampidou S, Dooley I, Molloy L, Beatty S (2010) Value of dual biometry in the detection and investigation of error in the preoperative prediction of refractive status following cataract surgery. Clin Experiment Ophthalmol 38: 255-265.
-
Dean CB, Nielsen JD (2007) Generalized linear mixed models: a review and some extensions. Lifetime Data Anal 13: 497-512.
-
Waltz KL, Featherstone K, Tsai L, Trentacost D (2014) Clinical Outcomes of TECNIS Toric Intraocular Lens Implantation after Cataract Removal in Patients with Corneal Astigmatism. Ophthalmology122: 39-47.
-
Sheppard AL, Wolffsohn JS, Bhatt U, Hoffmann PC, Scheider A, et al. (2013) Clinical outcomes after implantation of a new hydrophobic acrylic toric IOL during routine cataract surgery. J Cataract Refract Surg 39: 41-47.
-
Mendicute J, Irigoyen C, Aramberri J, Ondarra A, Montés-Micó R (2008) Foldable toric intraocular lens for astigmatism correction in cataract patients. J Cataract Refract Surg 34: 601-607.
-
Gale RP, Saldana M, Johnston RL, Zuberbuhler B, McKibbin M (2009) Benchmark standards for refractive outcomes after NHS cataract surgery. Eye (Lond) 23: 149-152.
-
Mazzini C (2013) Visual and refractive outcomes after cataract surgery with implantation of a new toric intraocular lens. Case Rep Ophthalmol 4: 48-56.
-
Hirnschall N, Maedel S, Weber M, Findl O (2014) Rotational stability of a single-piece toric acrylic intraocular lens: a pilot study. Am J Ophthalmol 157: 405-411.
-
Popp N, Hirnschall N, Maedel S, Findl O (2012) Evaluation of 4 corneal astigmatic marking methods. J Cataract Refract Surg 38: 2094-2099.
-
Visser N, Berendschot TT, Bauer NJ, Jurich J, Kersting O, et al. (2011) Accuracy of toric intraocular lens implantation in cataract and refractive surgery. J Cataract Refract Surg 37: 1394-1402.
-
Viestenz A, Seitz B, Langenbucher A (2005) Evaluating the eye's rotational stability during standard photography: effect on determining the axial orientation of toric intraocular lenses. J Cataract Refract Surg 31: 557-561.
-
Visser N, Bauer NJ, Nuijts RM (2013) Toric intraocular lenses: historical overview, patient selection, IOL calculation, surgical techniques, clinical outcomes, and complications. J Cataract Refract Surg 39: 624-637.
-
Miyake T, Kamiya K, Amano R, Iida Y, Tsunehiro S, et al. (2014) Long-term clinical outcomes of toric intraocular lens implantation in cataract cases with preexisting astigmatism. J Cataract Refract Surg 40: 1654-1660.
-
Goggin M, Moore S, Esterman A (2011) Toric intraocular lens outcome using the manufacturer's prediction of corneal plane equivalent intraocular lens cylinder power. Arch Ophthalmol 129: 1004-1008.
-
Sanders DR, Sarver EJ, Cooke DL (2013) Accuracy and precision of a new system for measuring toric intraocular lens axis rotation. J Cataract Refract Surg 39: 1190-1195.
-
Teichman JC, Baig K, Ahmed II (2014) Simple technique to measure toric intraocular lens alignment and stability using a smartphone. J Cataract Refract Surg 40: 1949-1952.
-
Patel CK, Ormonde S, Rosen PH, Bron AJ (1999) Postoperative intra-ocular lens rotation: a randomized comparison of plate and loop haptic implants. Ophthalmology 106: 2190-2195