International Journal of Ophthalmology and Clinical Research
The Surgical Outcome of two Different Tear Drainage Tube in the Proximal Obstruction of Lacrimal System
Titap Yazicioglu1* and Isil Kutluturk2
1Kartal Research and Training Hospital, Eye Clinic, Istanbul, Turkey
2Umraniye Research and Training Hospital, Eye Clinic, Istanbul, Turkey
*Corresponding author: Titap Yazicioglu, MD, Kartal Research and Training Hospital, Eye Clinic, Konaklar mh. Sebboy sok. Petekler sit. G/B D:18, Yenilevent, Istanbul, Turkey, Tel: +905322947119, E-mail: t_yazicioglu@hotmail.com
Int J Ophthalmol Clin Res, IJOCR-3-051, (Volume 3, Issue 2), Research Article; ISSN: 2378-346X
Received: February 29, 2016 | Accepted: May 16, 2016 | Published: May 19, 2016
Citation: Yazicioglu T, Kutluturk I (2016) The Surgical Outcome of two Different Tear Drainage Tube in the Proximal Obstruction of Lacrimal System. Int J Ophthalmol Clin Res 3:051. 10.23937/2378-346X/1410051
Copyright: © 2016 Yazicioglu T, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Purpose: To evaluate the results of conjunctival dacryocystorhinostomy surgery with two different tear drainage tube.
Methods: A total of 13 eyes of 13 patients (female 53.8%, male 46.2%) with persistent epiphora and secretion due to different etiologies were treated with two different tear drainage tube; silicone (PVP Metaireau tube-FCI Opththalmic), and Medpor coated tear drainage tube (Porex Surgical INC., Newnan, GA, USA). The choice of the tube was random, PVP tube was inserted in 8 (61.5 %) eyes and Medpor coated tube was inserted in 5 (38.5 %) eyes of patients in proximal obstruction of lacrimal system with different etiologies (functional obstruction secondary to external dacryocystorhinostomy, traumatic upper lacrimal system damaged, punctal atresia). The mean follow-up period was 16 months (range 12 to 24 months) in PVP and 24 months (range 12 to 36 months) in Medpor coated tear drainage tube. During the follow-up period, patients were observed for the patency of the lacrimal system and the complications.
Results: The patency of lacrimal system with lacrimal irrigation test was found negative in 8 of 13 (61.5%) patients. There was statistically difference between the patency of lacrimal system and type of the lacrimal drainage tube (p = 0.032 < 0.05). During the follow-up period 9 of 13 (69.2%) patients had more then one complication, 2 of 13 (15.4%) patients had only one complication, and 2 (15.4%) of patients had no complication. The complication rates didn't changed according to type of the tube (p = 0.103 > 0.05).
Conclusion: Although variations in technique and tube design will reduce the complications rates of conjunctival dacryocystorhinostomy, Medpor coated tear drainage tube seems to have some advantages in the stabilization and the patency of the lacrimal system. But further studies with longer follow-up times and a larger number of cases are needed to determine certain superiority of these two materials.
Keywords
Canalicul, Conjunctivodacryocystorhinostomy, Jones tube, Lacrimal
Introduction
Complete proximal canalicular obstruction is one of the main cause of epiphora among patients complaining about ocular discomfort in their daily life [1]. This functional disturbance is traditionally treated by a lacrimal bypass surgery named conjunctivodacryocystorhinostomy (CDCR). Other indications for this surgery are lacrimal pump failure associated with facial palsy and epiphora despite a patent dacriocystorinostomy (DCR) [1].
In the management of proximal obstruction of the lacrimal drainage system, performing a new drainage system between the conjunctiva and the nasal cavity with the insertion of a tear drainage tube, is an accepted treatment technique [2-5]. It was first described by Von Hoffman in 1904, and later by Kraupa and Goar [2]. Lester Jones in 1962, described various procedures for the management of proximal obstruction and noted a high rate of closure of the fistula in cases of CDCR with anastomosis of the lacrimal sac to the conjunctiva and nasal mucosa [2]. To overcome this problem, he tried different stents before developing a Pyrex glass tube [2]. Thin Pyrex tubes provided excellent capillary attraction, low irritation to surrounding tissues, and a low risk of blockage; but as complication rates were still unacceptably high for some surgeons, new materials and techniques were developed [2]. Although these developments, in patients with CDCR, the most common complications associated with tear drainage tube, are tube extrusion, malposition, obstruction, and diplopia [3].
The purpose of this study is to evaluate and compare the efficiency of CDCR surgery with the use of tear drainage tubes with different material, such as silicone and Medpor coated.
Materials and Methods
13 eyes of 13 patients; who underwent CDCR surgery with a tear drainage tube placement between September 2009 and November 2014 years were included in this retrospective study. The study was conducted in accordance with the ethical standards stated in the 1964 Declaration of Helsinki. The study was approved by the Local Ethics Committee of the participating center. All patients were informed about the purpose of the study and provided their consent.
The patients had grade 4 symptomatic epiphora according to Munk scale. The mean duration of symptom was 10.07 ± 1.7 months. All patients underwent preoperative lacrimal irrigation for detecting the patency of the lacrimal system. Functional obstruction secondary to external DCR surgery was seen in 8 of 13 (61.5%) eyes, traumatic upper lacrimal system damaged was seen in 4 of 13 (30.8%) eyes, and punctal atrezia was seen in 1 of 13 (7.7%) eyes. Patients with evidence of lacrimal hypersecretion with an anatomically patent DCR, eyelid malposition, idiopatic canalicular obstruction or canalicular obstruction due to viral infections of eye surface, Stevens - Johnson syndrome, lachrymal surgery in patients' previously undergone chemotherapy and radiotherapy were not included in the study.
The surgery was performed under general anesthesia. Initial surgical steps were same as classical external dacryocystorhinostomy surgery; after the lower flaps (lacrimal sac and mucosal flap) were sutured, passage was created from beneath of the caruncle to the lacrimal sac by using number 11 surgical blade. Initially we preferred to do anterior flaps between inferior nasal conjunctival flap and nasal mucosal flap, and inserting the tube into the nasal cavity. The choice of the tube to be implanted was random. Silicone (PVP Metaireau tube-FCI Opththalmic) were inserted in 8 of 13 (61.5%) eyes, and Medpor coated tubes were inserted in 5 of 13 (38.5%) eyes. The tube was fixed with 6-0 vicryl suture to the conjunctiva or lid margin. Patients were given topical antibiotics and steroid, as well as nasal decongestant postoperatively.
All patients were seen at 1 week and 1, 3, 6, 12 months after the surgery. Irrigation the bypass tube with normal saline was also done regularly in each control in order to maintain lacrimal passage patency. The patients were advised not to blow the noses for two weeks. As fluorescein test provides a quantitave measurement of lacrimal drainage function after DCR, we performed fluorescein dye disappearance test at the first visit postoperatively and also in patients complaining about persistent epiphora postoperatively.
Data were encoded and analyzed using the SPSS software (version 22.0, SPSS, Inc.). Descriptive statistics, frequency and percentage, were done. In consideration of total case number is lower than 30 (n < 30), nonparametric statistical tests (Chi-square test) was used for analysis. A P value less than 0.05 was determined to be significant. As the study consist of low number of patients, the results of the complications were given in percentage.
Results
Thirteen eyes of 13 patients were included in the study. The mean follow up period was 16 months in PVP tear drainage tube users and 24 months in medpor coated tear drainage tube users. The mean age of the patients were 45 ± 17.8 years (range from 17 to 70). 6 of 13 (46.1 %) patients were male, and 7 of 13 (53.8 %) patients were female. Seven patients had obstruction in the right eye, five patients had in the left eye, and one patient had bilateral obstruction. The etiological distribution of patients were shown in table 1. But in consideration of the size of the study, comparison of the success rates could not be done according to the etiological distribution.
![]()
Table 1: The etiological distribution of patients.
View Table 1
PVP tube was inserted in 8 (61.5 %) eyes and Medpor coated tube was inserted in 5 (38.5 %) eyes of 13 patients in proximal obstruction of lacrimal system with different etiologies (functional obstruction secondary to external dacryocystorhinostomy, traumatic upper lacrimal system damaged, punctal atresia). While the patency of lacrimal system with lacrimal irrigation test was found negative in 8 of 13 (61.5%) patients, it was positive in 5 (38.5%) of them. A comfortable, epiphora–free eye, with the patency of lacrimal passage and a normal dye clearance test was found in 4 of 5 (80%) eyes in Medpor coated tube, and 1 of 8 (12.5%) eyes in PVP tube. There is statistical difference between the patency of the lacrimal system with two different tubes (p = 0.032). In patients with epiphora (8 of 13, 61%) only one (2 of 8, 25%) and more than one complication (6 of 8, 75%), all which cause anatomical disorder, were seen (Table 2).
![]()
Table 2: The complication distribution of patients with epiphora.
View Table 2
During the follow-up period 9 of 13 (69.2%) patients had more than one complication, 2 of 13 (15.4%) patients had only one complication, and 2 (15.4%) of patients had no complication. There was no change in the complications according to tube type (p = 0.103 > 0.05). In the study symblepharon was observed in 2 (15.4%) eyes of 13 patients with anastomosis between inferior nasal conjunctival flap and nasal mucosal flap, and this complication didn't changed according to the material of the tube (p = 0.487 > 0.05). Tube malposition was only occurred in 1 of 8 (12.5%) patients with PVP tube. Obstruction at the distal end of the tube with nasal mucosal overgrowth was seen in 1 of 5 (20%) eyes with Medpor tube. Proximal end of the tube obstruction with conjunctival over-growth or granuloma was seen in 4 of 8 (50%) eyes with PVP tube eyes (Figure 1). Both distal and proximal end of obstruction was seen in 2 of 8 (25%) eyes with PVP tube (Table 3).
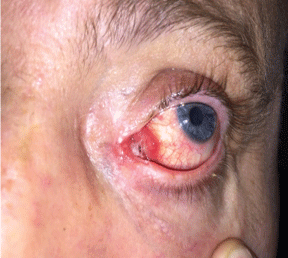
.
Figure 1: Proximal end obstruction with conjunctival over-growth in PVP tube patient.
View Figure 1
![]()
Table 3: The surgical results and complications income of lacrimal bypass surgery with different tear drainage tubes.
View Table 3
Discussion
Congenital absence of the canaliculi, destruction of the canaliculi following trauma, complete closure of either canaliculi or the inferior canaliculus following failure of conservative canalicular surgery, and cases with a permanent paralysis of the lacrimal pump are all known reasons for canalicular obstruction, and intermittent or constant tearing is the most common problem in canalicular obstruction. The frequency of canalicular obstruction is reported as 16-25 % in patients with epiphora [6]. Elodie et al. studied 71 patients with transcaruncular inserted angled extended Jones' tube and reported their ethiologic distrubition as idiopathic (48%, 34/71), viral (18%, 13/71), traumatic (13%, 9/71), congenital agenesis (9%, 6/71), chemotherapy (7%, 5/71), atopic (4%, 3/71), and also 19% unsuccessful external DCR [7]. Lim et al. studied 49 patients, and the causes of lacrimal obstruction in their study were such as idiopathic (20%), trauma (14%), congenital (12%), inflammation (10%), infection (10%), herpetic infection (8%), and tumor (8%) [8]. Our study mostly consist of unsuccessful external DCR patients.
Tear drainage tube can be made of various materials, such as polypropylene, silicone, Teflon, and Pyrex glass, and the ideal bypass tube should be straight rather than curved, hydrophobic, have some rigidity, less tissue reaction, and good biocompatibility [3]. The surgical success with different type of tear drainage tube was reported as 88% in the various studies [3,7]. An important factor in maintaining the patency of the desired fistula between the conjunctival sac and the nose is the retention of the tube [9]. The material of tube, anastomosis between the sac and the nasal mucosal flaps, length of the tube, depth of the insertion of the conjunctival end of the tube and the presence of fixation suture are all the factors affecting the retention of the tube [9].
Betharia et al. studied 20 CDCR patients with complaint of epiphora due to punctal or canalicular problem. They used Pyrex glass tubes in 10 patients and polyethylene tubes in 10 patients. They mentioned that Pyrex tubes have less irritation to the tissues and better drainage of tears due to their hdyrophilic nature than Polyethylene tubes. They also have lesser chances of obstruction with secretions because of the smooth surface thereby requiring syringing less often and hence less frequent follow-up [9].
The other known tube is Medpor (Porex Surgical Inc., Newnan, GA, USA) coated tear drainage tube, which provides good tissue tolerance and complete stability at a maximum follow-up of 2-6 years [3,7,10]. It is manufactured from borosilicate glass with a linear, high-density polyethylene porous (HDPP) biomaterial coating. The inner glass core conducts fluid into the nasal cavity, whereas the thin covering of linear HDPP permits tissue ingrowth for tube stabilization. A possible disadvantage of a too tight anchoring to the surrounding soft tissue may be difficult tube removal when re-intervention is required [3,7,11,12].
The most important tube related major complications are lateral or nasal tube displacement and obstruction. This is usually develops before the formation of a fistula and effects the surgical success [10]. Conjunctival over-growth or granuloma at the lateral end is known as minor complications [2,5,7].
Fan et al. used Medpor coated tube in their 26 of CDCR patients and reported tube obstruction in 9 of 26 patients (34.6%), and malposition in 1 (3.8%) of 26 patients [3]. They said that the most common complication in Medpor tube was tube obstruction either by soft tissue swelling (early obstruction), and overgrowth of conjunctiva or nasal mucosa (long-term obstruction) nearly 3 months after the surgery [3].
Elodie et al. used angled extended Pyrex glass Jones' tube, which has a standard length of 24 mm and angle of 130° in the middle, and reported as 31% major, and 8% minor complications [7]. Fernandez et al. used Jones tube between 17 and 26 mm in length and reported as 37.5% downward migration of the tube [5], and said that, surrounding tissue retraction following the edema and inflammation resulting after the surgery, as well as osteogenesis may responsible for the shifting of the tube [5].
In our study we used two different type of tube; silicon tear drainage tubes (PVP Metaireau tube-FCI Ophthalmic), and Medpor coated tear drainage tube (Porex Surgical INC., Newnan, GA, USA). The choice of the tube was random. We used polyglactin (6/0 vıcryl) suture in fixation of both tubes but found no advantages in the stabilization of the tubes. Major complications such as tube displacement was only seen in 1 (12.5%) of 8 patients with PVC tube. Distal tube occlusion with granulation tissue formation at the rhinostomy site was seen in 1 (20%) of 5 with medpor tube, and 2 (25%) of 8 with PVP tube. Proximal tube occlusion with conjunctival over-growth was seen in 2 (40%) of 5 Medpor coated tube, and 6 (75%) of 8 in PVP tube. There was no statistical difference in the complications according to type of the tube (p = 0.103). Epiphora–free eye, with the patency of lacrimal passage with lacrimal irrigation test and a normal dye clearance test, was observed in 4 (80%) of 5 eyes with Medpor coated tube, and 1 (12.5%) of 8 eyes with PVP tube. There was statistical difference between the patency of the lacrimal system with two different tubes (p = 0.032).
Granulation tissue may also occur at either end of the tube, and can usually be removed quite easily with toothed forceps. This problem occurs more often at the conjunctival end of the tube [13]. HDPP tube can lead to pyogenic granuloma formation, which leads to secondary tube obstruction [7]. In our study 8 of 13 patients (2 with medpor tube, and 6 with PVP tube) had pyogenic granuloma at the conjunctival end of the tube. This numeric discrepancy was not evaluated statistically because of limited case number.
Symblepharon formation was occurred in 1 of 5 (20%) Medpor coated, and 1 of 8 (12.5%) PVP tube patients who had conjunctival flap. This complication didn't changed according the material of the tube (p = 0.487 > 0.05). Because of this complication we gave up conjunctival flap formation technique, and preferred to do this surgery with tube insertion through the excised caruncule, which was really shortens the surgical time.
Less common complications mentioned in the studies were such as infection, discomfort, diplopia, corneal abrasion, and reflux of intranasal secretion into the fornix were not seen in our study.
In conclusion, present study showed that placement of Medpor coated tear drainage tube in patients undergoing conjunctival dacryocystorhinostomy, has better lacrimal passage stabilization and advantages in relieving tearing problems. But further studies are necessary with larger sample sizes for evaluating its stabilization and effectiveness.
Acknowledgement
Declaration of interest: The authors report no conflicts of interest. The authors alone are responsible for the content and writing of the paper.
Conflict of Interest
No conflicting relationship exists for any author.
References
-
Yuriani I, Lesmana MI, Cruz HO (2009) Conjunctivo-Dacryocystorhinostomy with Tube Implantation in Failed External Dacryocystorhinostomy: A Case Report. J Kedokteran Meditek 16: 1-9.
-
Athanasiov PA, Madge S, Kakizaki H, Selva D (2011) A review of bypass tubes for proximal lacrimal drainage obstruction. Surv Ophthalmol 56: 252-266.
-
Fan X, Bi X, Fu Y, Zhou H (2008) The use of Medpor coated tear drainage tube in conjunctivodacryocystorhinostomy. Eye (Lond) 22: 1148-1153.
-
Sekhar GC, Dortzbach RK, Gonnering RS, Lemke BN (1991) Problems associated with conjunctivodacryocystorhinostomy. Am J Ophthalmol 112: 502-506.
-
Alañón Fernández MA, Alañón Fernández FJ, Martínez Fernández A, Cárdenas Lara M (2008) [Conjunctivodacryocystorhinostomy with the assistance of diode laser. Endoscopic placement of Jones lacrimal tubes]. Acta Otorrinolaringol Esp 59: 11-15.
-
Kashkouli MB, Pakdel F, Kiavash V (2012) Assessment and management of proximal and incomplete symptomatic obstruction of the lacrimal drainage system. Middle East Afr J Ophthalmol 19: 60-69.
-
Elodie W, Ilse M (2015) The survival of an angled extended Jone's tube. Br J Ophthalmol 99: 1523-1526.
-
Lim C, Martin P, Benger R, Kourt G, Ghabrial R (2004) Lacrimal canalicular bypass surgery with the Lester Jones tube. Am J Ophthalmol 137: 101-108.
-
Betharia SM, Ritu A, Sushil K (1988) Comparative evaluation of Jones pyrex tubes and polyethylene tubes in conjunctivo-dacryocystorhinostomy. Indian J Ophthalmol 36: 162-164.
-
Lee JS, Jung G Lee JE, Oum BS, Lee SH, et al. (2001) The treatment of lacrimal apparatus obstruction with the use of an inner canthal jones tube insertion via a transcaruncular route. Ophthalmic Surgery and Laser 32: 48-54.
-
Pushker N, Khurana S, Shrey D, Bajaj MS, Chawla B, et al. (2013) Conjunctivodacryocystorhinostomy using a high-density porous polyethylene-coated tear drain tube. Int Ophthalmol 33: 329-333.
-
Wojno T (2010) Experience with a medpor-coated tear drain. Ophthal Plast Reconstr Surg 26: 327-329.
-
Bartley GB, Gustafson RO (1990) Complications of malpositioned Jones tubes. Am J Ophthalmol 109: 66-69.





