International Journal of Ophthalmology and Clinical Research
Lutein and Zeaxanthin Isomers Inhibit Cholinesterase and Modulate the Expression of Inflammation-Related Genes: In Vitro Models
Khadija Ghanam1, Jayant Deshpande1 and Vijaya Juturu2*
1OmniActive Health Technologies Ltd, Prince Edward Island, Canada
2OmniActive Health Technologies Inc, Morristown, NJ, USA
*Corresponding author: Vijaya Juturu, PhD, FACN, OmniActive Health Technologies, 67 East Park Place, Suite 500, Morristown, NJ 07950, USA, E-mail: v.juturu@omniactives.com
Int J Ophthalmol Clin Res, IJOCR-2-044, (Volume 2, Issue 6), Research Article; ISSN: 2378-346X
Received: October 30, 2015 | Accepted: December 14, 2015 | Published: December 16, 2015
Citation: Ghanam K, Deshpande J, Juturu V (2015) Lutein and Zeaxanthin Isomers Inhibit Cholinesterase and Modulate the Expression of Inflammation-Related Genes: In Vitro Models. Int J Ophthalmol Clin Res 2:043. 10.23937/2378-346X/1410044
Copyright: © 2015 Ghanam K, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Purpose: Exposure to solar ultraviolet (UV) radiation is believed to increase oxidative stress and may cause oxidative damage to retinal pigment epithelial (RPE) cells. UV light triggers a number of deleterious cellular responses in retinal cells that lead to changes in gene and protein expression. Lutein and zeaxanthin isomers-RR- and RS-zeaxanthin-(L/Zi) are carotenoids that play a functional role in the macula and retina of the eye. Optimal levels of L/Zi may reduce the risk of macular degeneration, due in large part to their antioxidant properties and ability to absorb light within the UV range. This study is to evaluate macular carotenoids' (Lutemax® 2020) effect on cholinesterase activity, antioxidant enzymes and in response to UV irradiation in an in vitro model.
Methods: Retinal pigment epithelial (RPE) cells play a key protective role by shielding the retina from damaging UV rays, and are used as a model to study the effect of therapeutic interventions. In this study, a human RPE cell line [ARPE-19 cells were acquired from ATCC (CRL-2302)] was either mock treated or given L/Zi (Lutemax® 2020) for 24 h prior to mild UV irradiation (100 μJ/cm2 for 7 sec). Cells were maintained for another 18 h, then harvested for gene expression, cytokine gene expression (qPCR), cytokine protein levels (protein array chip), and alteration in antioxidant enzymes activities. In another study, acetylcholinesterase and catalase activities in the presence of L/Zi were tested.
Results: In an in vitro model, L/Zi treatment inhibited cholinesterase activity and enhanced catalase activity. These results suggest that inhibition of cholinesterase enzyme and enhancing antioxidant enzymes activities may have several therapeutic applications such as neurodegeneration disorders and myasthenia gravis. Mild UV irradiation affected significant changes in 545 genes, including down-regulation of c-SRC and β-catenin, and up-regulation of VEGF and FOXO-3A. L/Zi induced changes in 520 genes, most notably down-regulation of β-catenin, and up-regulation of specific G-protein constituents that support neurophysiologic processes in vision and enhanced immune system poise. L/Zi supplemented cells were mild UV irradiated, 573 genes were significantly affected, most notably an up-regulation of c-SRC. There were changes in cytokine gene expression and enhancement in SOD and GPx activities.
Conclusions: L/Zi treated cells may ameliorate the effects of mild UV irradiation on RPE cells, as shown by expression of genes involved in cell proliferation, inflammation, immune function and wound healing.
Keywords
UV radiation, Oxidative stress, Inflammation, Acetylcholinesterase, Antioxidants, Age-related macular degeneration, Alzheimer's disease, Lutein, Zeaxanthin isomers, Meso-zeaxanthin, RS-zeaxanthin, RR-zeaxanthin, Visual function, Eye fatigue
Introduction
It is well established that ultraviolet (UV) radiation induces immunomodulatory effects involved in different chronic conditions including skin cancer, age-related macular degeneration (AMD), refractive errors, glaucoma and cataracts. Epidemiological studies suggest solar radiation is responsible for some of the deteriorating changes that lead to AMD [1-3] Recent studies have shown that UVA (320-400 nm) and UVB (290-320 nm) radiation are immunosuppressive [4,5]. Experimental studies have demonstrated that bright light preferentially damages precisely the region that degenerates in AMD [2,6-8]. The retina is highly susceptible to photochemical damage from continuous exposure of light and oxygen. The cornea and the lens block a major part of the ultraviolet (UV) radiation from reaching the retina (< 295 nm). The relationship between UV light exposure and AMD is unclear, although short wavelength radiation and the blue light induce significant oxidative stress to the retinal pigment epithelium [2].
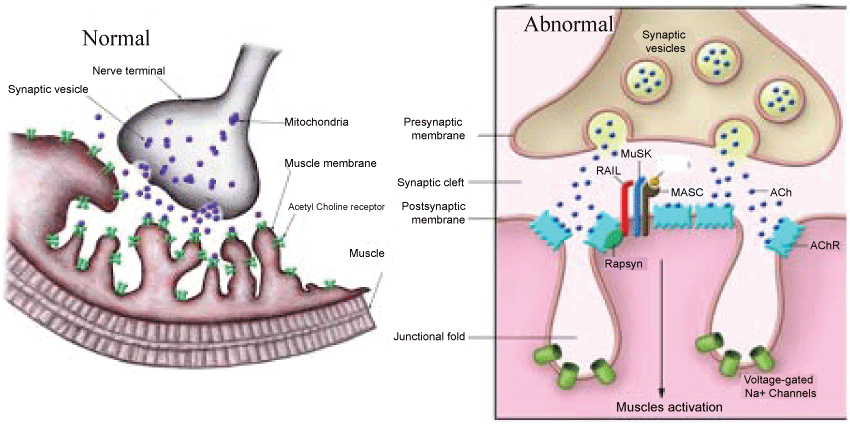
As electronic devices such as IPads, computers, lab tops, cell phones and tablets are part of our life and more and more people experiencing a variety of ocular symptoms. These include eye fatigue, eyestrain, tired eyes, irritation, redness, blurred vision, and double vision, collectively referred to as computer vision syndrome with so many other symptoms. The lighting conditions, luminance and contrast affect eye sight. The muscles around the eyes are commonly affected become weak and easily tired. Normally when impulses travel down the nerve, the nerve endings release a neurotransmitter substance called acetylcholine. The acetylcholine quickly attached to receptors on the muscles. This in turn triggers the muscles to tighten. There are many acetylcholine receptors on each muscle fiber. The eyelid and extra-ocular muscles are commonly involved in myasthenia, either in isolation or with other skelet al muscles. When acetylcholine released from a nerve and tries to induce contraction in a muscle, the receptors cannot attach to the acetylcholine resulting in muscle weakness and fatigue. Approximately 90% of people with myasthenia gravis have ocular involvement and 50% present with ocular symptoms. Double vision from eye misalignment (strabismus) and drooping of the eyelids (ptosis) are the most common symptoms. These symptoms classically vary and worsen throughout the day. The incidence estimated at 20/100,000. There appears no racial or sex predilection in myasthenia. This is due to weakness by blocking acetylcholine receptors at the post-synaptic neuromuscular junction, inhibiting the stimulative effect of the neurotransmitter acetylcholine (Figure 1).
.
Figure 1: Acetylcholine and Neuromuscular Junction in Normal and Abnormal conditions. Source: J Clin Invest 2001;116(11):2843-2854.
View Figure 1
Lutein and zeaxanthin are isomers differ location of a single double bond. Zeaxanthin exists as 3 stereoisomeric forms; (3R, 3'R)-zeaxanthin and (3R, 3'S)-zeaxanthin (also called as meso-zeaxanthin) are the main forms present in the macula of the retina, while small amounts of (3S, 3'S)-zeaxanthin have also been detected. Xanthophylls reported to be protective against photo-induced oxidative damage. Acetylcholine can affect the muscles around the eyes and be useful in controlling twitching and affecting the focusing and size of the pupil. Its main role would be in brain health. Supplementation with lutein/zeaxanthin isomers has been shown to increase circulating and tissue levels of these xanthophylls [9]. Lutein and zeaxanthin isomers are capable of quenching ROS such as singlet oxygen, and are effective scavengers of free radicals [10-13]. Exposure to UV light can result in lipid peroxidation and generation of peroxyl radicals; thus, L/Zi play an important role, particularly in the protection against photo-induced oxidative damage [11].
Lutein and zeaxanthin are potent antioxidants, and they can also act as filters of high-energy blue light. These xanthophylls are protective against photo-induced oxidative damage, in highly exposed tissues such as the skin and eyes. Recent data [14] support a protective role for lutein and zeaxanthin isomers against UV-induced damage to the skin, as well as improving skin health attributes. In preclinical and clinical studies, supplementation with lutein and zeaxanthin reduced reactive oxygen species (ROS) [15,16] and inflammatory responses [17,18] resulting from ultraviolet (UV) exposure [19]. The role of lutein and zeaxanthin in eye health has been further supported by epidemiological studies reporting by an inverse relationship between lutein intake and eye disease, and by some intervention studies wherein supplementation with lutein/zeaxanthin enhanced measures of visual function. The retina, cornea and lens are highly susceptible to photochemical damage from continuous exposure of light and oxygen. Catalase is a ubiquitous antioxidant enzyme that degrades oxidative stress induced by hydrogen peroxide [20]. It helps in protecting the cell from oxidative damage. Reactive oxygen species cause oxidative damage to cellular proteins, lipids and carbohydrates and inflammation, free radicals hydrogen peroxide and UV causes depletion of cellular antioxidants and antioxidant enzymes (SOD).
Thus, the present study designed to study the effects of lutein and zeaxanthin isomers (L/Zi) supplementation on cholinesterase, catalase activity and effect of mild UV radiation (with and without lutein supplementation) induced oxidative damage and inflammation-related genes.
Methods
Study I
Acetylcholinesterase assay: The inhibitory effect of cholinesterase activity by the extract was spectrophotometrically assessed in 96 well plates.
Acetylcholinesterase assay basis: AChE hydrolyzes acetylthiocholine to release thiocholine, which then reacts with DTNB to produce a yellow colour that can be detected as a change in absorbance at 412 nm.
Acetylthiocholine iodide + AChE = thiocholine + acetic acid; thiocholine + DTNB = yellow color
To perform acetylcholinestarse assay, 140 μl of 0.1 M sodium phosphate buffer (pH 8) were preincubated with 20 μl extract and 20 μl of 0.5 unit/ml acetylcholinestaerse enzyme for 15 min at 25°C. Then 10 μl of 10 mM of 5'5'-dithiobis-2-nitrobenzoic acid (DTNB) was added to each well, followed by 10 μl of 14 mM acetylcholine iodide used a substrate. The end product of the hydrolysis of acetylthiocholine was determined by spectrophotometrically monitoring a colorometric end product 5-thio-2-nitrobenzoate anion at 412 nm using a BioTek Synergy H1 plate reader. Eserine salicylate (10 mM), an acetylcholinesterase inhibitor was used as positive control. Vehicle diluent at equivalent concentrations (v/v) as the samples being tested and a DD H2O blank lacking AChE served as negative controls [C3389-500UN (SIGMA Aldrich)].
Each test was conducted in triplicate. A set of 6 dilutions was used to calculate 50% inhibitory concentration (IC50). Inhibition was calculated according to the equation: % Inhibition = (1-X/Y) * 100, where X= A414nm of diluent control and Y= A414nm of test sample.
Catalase (CAT) activity: Catalase activity was performed using a commercially available kit (Amplex Red Catalase Assay, Thermo Scientific (Catalog number A22180). In this assay, catalase first reacts with H2O2 to produce water and Oxygen. Then the amplex red reacts with any unreacted H2O2 in the presence of horseradish peroxidase (HRP) to produce a fluorescent oxidation product, resorufin. Changes in fluorescence are proportional to the breakdown of H2O2. As the catalase activity increases, the signal from resorufin decreases.
To perform catalase assay, in each well, 25 μl of H2O2 (40 μM), 25 μl catalase (4 U/ml) and 25 μl of cell lysates diluted in the assay buffer were added in each well and were incubated for 30 min. Then, an Amplex Red/ HRP working solution (50 μl) was added to each well and incubated for 30 min at 37°C, protected from light. The fluorescence emitted by resofurin was measured at multiple time points at an excitation of 530 nm and an emission detection at 590 nm using a BioTek Synergy H1 plate reader. The blank value was subtracted from each sample.
Study II
The human retinal pigment epithelia (RPE) cell line plays a key protective role by shielding the retina from damaging UV rays. This cell line is frequently used as a model to study the effect of therapeutic interventions. ARPE-19 cells were acquired from ATCC (CRL-2302). Figure 2 provides experimental study design. ATCC complete growth medium ATCC-formulated DMEM F12 Medium Catalog No. 30-2006 fet al bovine serum.
Sample preparation: Stock solutions of the test extracts were prepared in dimethylsulfoxide (DMSO) at 5 mM (53 mg lutein in 18.63 ml DMSO) and stored in 200 μl aliquots at -20°C until used. The effective vehicle concentration was equalized to 0.5% in complete culture medium for all test conditions.
Investigational product: Lutemax® 2020 [OmniActive Health Technologies Ltd., India] is a concentrate containing at least 80% carotenoids, with a minimum of 63.75% lutein and 11.25% zeaxanthin isomers in the free form. (3R, 3'R)-zeaxanthin and (3R, 3'S)-zeaxanthin (i.e., meso-zeaxanthin) are present at a ratio of approximately 50:50, and batch analytical data suggest the ratio of these 2 isomers may vary between 40:60 to 60:40. Lutemax® 2020 provides higher levels of zeaxanthin compared to other commercial lutein preparations, which reflects more accurately the ratio of these xanthophylls present in dietary sources.
Positive controls and primer assay products used in the study are Etoposide (E1383-25 mg), from Sigma Aldrich, 216763-100 mL H2O2 Sigma, Lutein and zeaxanthin isomers (Lutemax® 2020) from OmniActive Health Technologies Ltd. India and M50-OkCAFOY Biorad Human cytokine 27 Plex kit and MLL-9601 Multiplate™ Low-Profile 96 Well Unskirted PCR Plates from Biorad. MLL-9601 Multiplate™ Low-Profile 96 Well Unskirted PCR Plates from Biora, 170-8842 iScript™ Advanced cDNA Synthesis Kit for RT from Biorad, 4307281 Total RNA Control (Human) from Life Technologies,172-5202 SsoFast™ EvaGreen® Supermix (10 plates) from Biorad, PPH00171C-200 RT2 qPCR Primer Assay for Human IL1B , PPH00560C-200 RT2 qPCR Primer Assay for Human IL6, PPH00568A-200 RT2 qPCR Primer Assay for Human IL8, PPH00251C-200 RT2 qPCR Primer Assay for Human VEGFA and PPH00341F-200 RT2 qPCR Primer Assay for Human TNF purchased from Qiagen.
Evaluation of cytotoxicity and cytoprotection: Cells were seeded in quadruplicate in 96-well plates at 1.5 × 104 cells/well and treated with lutein for 24 or 48 h hours. Studies were completed in triplicate. Cell proliferation was initially assessed using a standard colorimetric indicator of metabolic activity (CIMA) assay. In this assay, the reduction of yellow tetrazolium salt (MTT) to purple formazan by mitochondrial reductase enzymes in viable cells was evaluated as a change in absorbance (ʎ = 500-600). Samples were diluted as required to ensure that values obtained with the MTT assay fell within the linear range of the protocol. Mock treated cells (0 μM lutein), cells incubated with vehicle alone (Vehicle or DMSO) and cells treated with staurosporine (STS, 5 nM) or hydrogen peroxide (H2O2, 100 μM) served as references. Qualitative microscopic evaluation of treated cultures was used to supplement the quantitative CIMA data as a routine measure. Cells seeded were pretreated with L/Zi for 24 h and then exposed to UV (100 μJ/cm2 for 7 sec). MTT assays were conducted at 24 h after UV exposure.
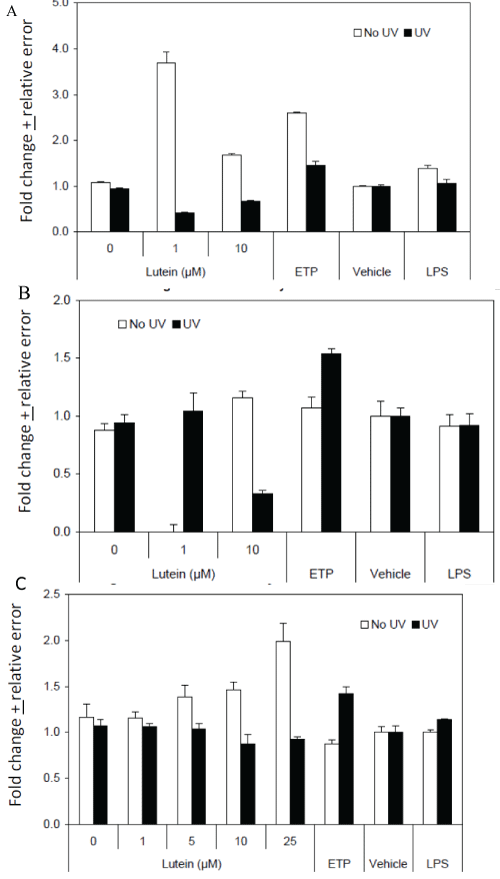
Release of lactate dehydrogenase (LDH): LDH is a measure of cytotoxicity. Cells were treated with L/Zi for 24 h and either mock-exposed (no UV) or exposed to UV as above. Cells treated with etoposide (ETP, 100 μM), lipopolysaccharide (LPS 100 ng/mL) served as references. Complete disruption of the cell membrane to assess 100% LDH release was obtained with Triton X-100 (10%). Conditioned media was harvested 24h after UV exposure and LDH activity evaluated at ʎ = 490 nm.
As per figure 1, RPE cells were mock-treated or given lutein plus zeaxanthin supplement for 24-hours prior to UV irradiation (100 μJ/cm2 for 7 sec). This is a relatively mild challenge insufficient to cause cell death but nonetheless sufficient to induce cellular stress responses. Following the UV challenge the cells were maintained for another 18-hours at which time they were harvested for gene expression profiling, cytokine expression, and alteration in the activities of antioxidant enzymes.
Cytokine expression: ARPE cells were mock-treated or treated with 10 μM of lutein for 24 h and either exposed to UV as above or not exposed. After 24 h, cells and conditioned media were harvested for subsequent analysis. Cells were used for the isolation of RNA and preparation of cDNA. Expression of IL-1β, IL-6, IL-8, TNF and VEGF was evaluated by QRT-PCR. Cytokine expression in conditioned media was assessed using a 27-plex liquid protein array and a Luminex MagPix bead analyzer. Three sets of triplicate wells were combined for analysis. Cells treated with vehicle (DMSO), etoposide (100 μM) or LPS (100 ng/ml) served as reference controls (Table 1A and Table 1B).
![]()
Table 1a: Cytokine gene expression in L/Zi-treated, UV-exposed ARPE cells.
View Table 1a
![]()
Table 1b: Cytokine gene expression in L/Zi-treated, no UV Exposure (B) ARPE cells.
View Table 1b
Glutathione peroxidase (GPx) activity: Cells were seeded and treated as above. After mock-exposure or UV-exposure, cells were harvested and subjected to three freeze-thaw cycles and then cleared by centrifugation. GPx activity was evaluated using a commercially available kit. Cell lysates were combined with assay buffer and NADH, and tert-butyryl hydroxide added to initiate the reaction. Absorbance at ʎ = 340 nm was evaluated at 34 sec intervals for six minutes and GPx activity expressed a units/ml/min.
Superoxide dismutase (SOD) activity: Cell lysates collected as for the GPx assay were also used to evaluate SOD activity using a commercially available kit. Cell lysates were combined with assay buffer and absorbance at ʎ = 450 nm was evaluated after 20 minutes. SOD activity was determined as % inhibition using a reference curve.
Peroxidase activity: Cell lysates collected as for the GPx assay were also used to evaluate non-specific peroxidase activity using Amplex Red reagent. The basis for this assay is the quantitative conversion of Amplex Red reagent to the fluorogenic oxidation product, resorufin, in the presence of oxidative species. Cell lysates were combined with assay buffer and fluorescence evaluated at ex: 530 nm and em: 590 nm after 30 min.
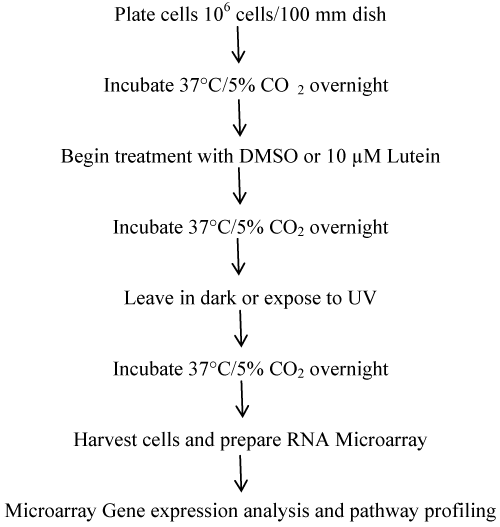
Microarray gene erxpression analysis and pathway profiling: Plate ARPE-19 cells at a density of 1 × 106 cells per 100 mm dish in ATCC, incubate for 37°C and 5% CO2 overnight compete growth medium overnight. It was organized into 4 sets of plates in triplicate (Total: 12 samples) such as (A) No treatment, (B) Lutein (10 μM; 24 h), (C) UV (18 h), and (D) Lutein (10 μM; 24 h) followed by UV (18 h). Harvest cells and prepare RNA for microarray gene expression analysis and pathway profiling (Figure 2).
Microarray processing: Total RNA was extracted from tissues and cells using Qiagen RNeasy Mini Kit according to manufacturer's instruction. Illumina Whole Genome HumanHT12 v4 Expression BeadChip was used in this study. Each RNA sample with 0.5 μg of total RNA was amplified using the Illumina TotalPrep RNA amplification kit with biotin UTP (Enzo) labeling. The Illumina® TotalPrep RNA amplification kit uses T7 oligo(dT) primer to generate single-stranded cDNA followed by a second strand synthesis to generate double-stranded cDNA which is then column purified. In Vitro transcription was done to synthesize biotin labeled cRNA using T7 RNA polymerase. The cRNA was then column purified. The cRNA was then checked for size and yield using the Bio-Rad Experion system. 1.5 μg of cRNA was hybridized for each array using standard Illumina protocols with streptavidin-Cy3 (Amersham) being used for detection. Slides were scanned on an Illumina Bead station. Summarized expression values for each probe sets were generated using Bead Studio 3.1 (Illumina, Inc). The data were background subtracted and quantile-quantile normalized across samples using MBCB algorithm [21].
Statistical analysis: A two-sample t-test was performed between each pair of control and treated sample of interest. Genes with p < 0.01 and fold change greater than 2 were considered as changed with statistical significance. The gene lists of significant changes were applied to functional enrichment and pathway analysis using Ingenuity Pathway Analysis software (IPA).
Results
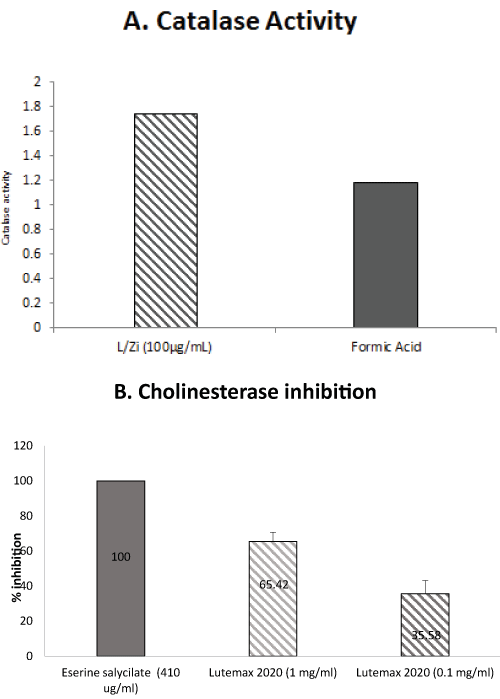
L/Zi treatment inhibited cholinesterase activity and enhanced catalase activity. These results suggest that inhibition of cholinesterase enzyme and enhancing antioxidant enzymes activities may have several therapeutic applications such as neurodegeneration disorders and myasthenia gravis. (Figure 3A and Figure 3B).
.
Figure 3: (A) L/Zi significantly increases catalase activity; (B) Inhibits cholinesterase.
View Figure 3
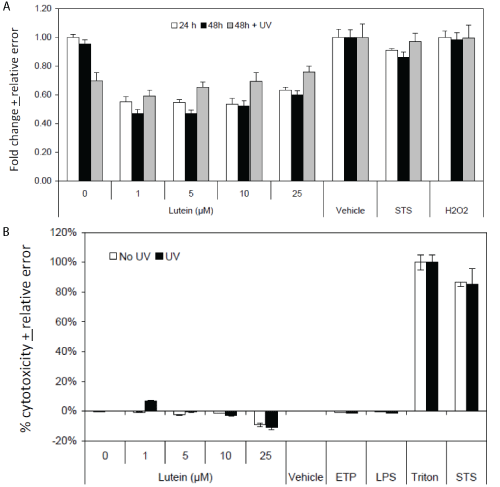
MTT analysis of L/Zi-treated ARPE cells and lutein-treated, UV-exposed ARPE cells.
Figure 4A provides MTT analysis and Figure 4B provides release of lactate dehydrogenase activity. In comparison, STS induced significant amounts of cytotoxicity. UV exposure had no discernible effect. Analysis of apoptosis using a DNA fragmentation assay under similar experimental conditions revealed no evidence of apoptosis under any of the test conditions (data not shown).
.
Figure 4: Cytoprotection and Cytotoxicity Effects in UV Exposed ARPE cells. (A) MTT analysis of L/Zi Treated Cells; (B) LDH Analysis of L/Zi Treated Cells.
View Figure 4
Gene expression profiling
UV-induced changes: The fold changes in gene expression were modest (3-fold or lower). There were 545 genes with significant change in expression. Most notably down-regulation of c-Src and β-catenin implying a significant trend in anti-proliferative, pro-differentiation signaling pathways and up-regulation of VEGF suggesting a trend towards angiogenesis/neo-vascularization and up-regulation of FOXO-3A, a cellular response to UV-induced stress and trends towards apoptosis was observed (Table 2A).
![]()
Table 2a: Identification of UV-responsive gene expression.
View Table 2a
Lutein/zeaxanthin isomers -induced changes: Fold changes in gene expression were modest (2.5-fold or lower). There were 520 genes with significant change in expression. Most notably upregulation of specific G-protein constituents that support the neurophysiological process in vision and upregulation of other G-protein constituents, trends that support enhance poise of immune system and down regulation of β-catenin that parallels the UV-induced trend towards pro-differentiation signaling pathways (Table 2B).
![]()
Table 2b: Identification of L/Zi-responsive gene expression.
View Table 2b
Effect of Lutein/zeaxanthin isomers supplementation on UV-induced changes: The fold changes in gene expression were modest (2.5-fold or lower). There were 573 genes with significant change in expression. A significant trend towards anti-inflammatory, anti-apoptosis pathways that are opposite to those were observed with UV exposure alone (Table 2C). Implications are lutein may reverse UV-induced pro-inflammatory, pro-angiogenic patterns of gene expression towards patterns of gene expression involved in cellular differentiation and wound healing. For example, c-SRC was upregulated.
![]()
Table 2c: Effects of L/Zi on UV-responsive gene expression.
View Table 2c
Discussion
The watermen in the Chesapeake Bay area survey [22] and Beaver Dam Eye Study [23] reported chronic exposure to blue or visible light related to development of AMD. In these studies, the amount of time spent outdoors in summer associated with increased retinal pigment (odds ratio [OR], 1.44). Wearing eyeglasses was inversely associated with increased retinal pigment (OR, 0.75), and the use of hats and sunglasses was inversely associated with soft indistinct drusen (OR, 0.61). The amount of leisure time spent outdoors in summer was definitely associated with exudative macular degeneration (OR, 2.26) and late maculopathy (OR, 2.19). In another study [24], participants in their 30s exposed for more than 5 hours a day in sunlight reported a higher risk of developing increased retinal pigment (risk ratio [RR], 2.99, P =0.02) and by 10 years they developed early ARM (RR, 2.20, P = 0.04) than those exposed less than 2 hours per day during the same age. Antioxidants especially L/Zi should cut the risk of AMD and more specifically their protective effects greatest for those with the greatest concentration of lipofuscin. Few studies reported the progression of AMD risk associated with an increase in blue light exposure [23,25].
High-energy part of the solar spectrum is most hazardous to retinal molecules, with damaging effects increasing as photon energy rises. High-energy visible and ultraviolet photons can produce molecular damage by a photochemical mechanism. The lesion is exacerbated by oxygen, which initiates free-radical chain reactions (photodynamic effects). Melanin exerts a protective effect against damage from sunlight. Radiation exposure can lead to impaired vision and transient or permanent blindness. Ultraviolet radiation is also a risk factor for damage to the retinas of children. The removal of these wavelengths from ocular exposure will greatly reduce the risk of early cataract and retinal damage [26]. Lutein and zeaxanthin have peak absorption at 446 nm of the visual light spectrum, which corresponds to the blue wavelength of light, the visible light with the highest energy [27].
Macular pigment has a broad band absorbance spectra peaking at 460 nm. MPOD typically absorbs about 70% of incident light at this wavelength. Optimal levels of lutein and zeaxanthin isomers [L/Zi] reduce the risk of macular degeneration, due in large part to their antioxidant properties and ability to absorb light within the UV range. Although the protective effect of lutein and zeaxanthin against photo-induced damage in the skin and eye attributed primarily to their anti-oxidant and UV-filtering properties [28], it has been recently suggested that lutein may also act by directly influencing immune responses and inflammation pathways [29].
The antioxidant enzymes pathway considered the most important pathway involved in the repair of reactive oxygen species (ROS)-induced damage. In the lens, superoxide dismutase (SOD), catalase (CAT), and glutathione peroxidase (GPX), are some of the antioxidant enzymes that protect the organisms from oxidative damage [30-32]. CAT catalyzes decomposition of hydrogen peroxide into water and oxygen, thereby preventing cell damage from high levels of ROS. Acetylcholine is a chemical messenger that carries signals between nerve and muscle. An enzyme called acetylcholinesterase breaks down acetylcholine. L/Zi inhibited acetylcholinesterase this may potentially increase the amount of acetylcholine available and help muscle activation and contraction. Further human clinical trials required to explore L/Zi role as acetylcholinesterase inhibitor and eye fatigue. In addition, cholinesterase inhibitors (ChE-Is) are the standard of therapy for treatment of patients with Alzheimer disease (AD) and are the only class of drugs approved by the Food and Drug Administration (FDA) for treatment. Recently Nolan et al. [33,34] reported patients with Alzheimer's disease (AD) show much less macular pigment (MP) and poorer vision when compared to control subjects. Nechifor et al. [35] show a decrease in catalase activity as an effect of UVA exposure and the tendency of the irradiated cells to restore the antioxidant activity of catalase in an exposure time-dependent manner (Figure 5A, figure 5B and figure 5C). In cellular antioxidant systems catalase as a source of antioxidant compound provided by its heme prosthetic group released and modified after catalytic photo-inactivation induced by UVA.
.
Figure 5: Effects on Cellular Antioxidant Defense Enzymes. (A) GPx Activity in L/Zi Treated Cells; (B) SOD Activity in Lutein Treated Cells; (C) Peroxidase activity in Lutein Treated Cells.
View Figure 5
Kim et al. [36] and Ott et al. [37] reported that oxidative stress causes apoptosis. Macular pigment believed to limit retinal oxidative damage by absorbing incoming blue light and/or quenching ROS. Lutein and zeaxanthin associated with reduced risk of neovascular changes [38-40]. Macular pigment consisting of lutein and zeaxanthin through its ability to filter light and by direct antioxidative properties, proposed as the most effective protective factor in the central retina and important to cut light induced oxidative retinal damage [41].
Supplementation with the macular carotenoids (MZ, Z, and L) benefits patients with AD, in terms of clinically meaningful improvements in visual function and in terms of MP augmentation. AD patients were observed to show much less MP, lower serum concentrations of L and Z, poorer vision, and a higher occurrence of AMD when compared to control subjects.
The antioxidative properties of lutein and zeaxanthin isomers have garnered a lot of interest beyond protection against photo-oxidative damage, to include effects on DNA damage repair and cancer. In Vitro and in vivo studies have confirmed that lutein and zeaxanthin have the capacity to: decrease double strand breaks and DNA cross-linking, inhibit mutagenicity of aflatoxin, suppress the initiation stages of hepatocarcinogenesis, and protect against hepatotoxins (i.e., ethanol, paracetamol, and carbon tetrachloride) [42-46].
Supplementation of lutein enhances macular pigment and an increase in MPOD may decrease the risk of AMD and other visual disease conditions. Lutein supplementation also showed its antioxidant properties and reduces oxidative stress markers. L/Zi treated cells may ameliorate the effects of mild UV irradiation on RPE cells, as shown by expression of genes involved in cell proliferation, inflammation, immune function and wound healing [14,47]. Supplementation of lutein reduces the risk of age related macular degeneration (AMD) or cataract formation based on its biological properties. Lutein reduces progression of ocular inflammation models, neovascularization, diabetes, retinal ischemia and reperfusion. An acetylcholinesterase inhibitor that promotes RGC survival, can protect the retinal microvasculature and enhance blood flow [48]. Limitations are due to the low dose of UV used and/or to the late time-point of gene sampling we did not see all DNA damage effects. Further studies planned to test different doses of UV exposure and L/Zi on gene profiling and cytokines.
Conclusions
L/Zi treatment improves antioxidants, gene proteins and antioxidant activity, decreases oxidative stress and eye fatigue due to inhibition of cholinesterase activity and mild UV irradiation on RPE cells reduced oxidative stress and inflammation as shown by expression of genes involved in cell proliferation, inflammation, immune function and wound healing.
Acknowledgement
Authors are grateful to NRC, Canada for providing lab facilities and Dr. Chaitanya Nirodi, University of South Alabama for valuable suggestions.
References
-
Young RW (1988) Solar radiation and age-related macular degeneration. Surv Ophthalmol 32: 252-269.
-
Chalam KV, Khetpal V, Rusovici R, Balaiya S (2011) A review: role of ultraviolet radiation in age-related macular degeneration. Eye Contact Lens 37: 225-232.
-
Beatty S, Koh H, Phil M, Henson D, Boulton M (2000) The role of oxidative stress in the pathogenesis of age-related macular degeneration. Surv Ophthalmol 45: 115-134.
-
Halliday GM, Damian DL, Rana S, Byrne SN (2012) The suppressive effects of ultraviolet radiation on immunity in the skin and internal organs: implications for autoimmunity. J Dermatol Sci 66: 176-182.
-
Matthews YJ, Halliday GM, Phan TA, Damian DL (2010) Wavelength dependency for UVA-induced suppression of recall immunity in humans. J Dermatol Sci 59: 192-197.
-
Ramchani-Ben Othman K, Cercy C, Amri M, Doly M, Ranchon-Cole I (2015) Dietary supplement enriched in antioxidants and omega-3 protects from progressive light-induced retinal degeneration. PLoS One 10: e0128395.
-
Margrain TH, Boulton M, Marshall J, Sliney DH (2004) Do blue light filters confer protection against age-related macular degeneration? Prog Retin Eye Res 23: 523-531.
-
Brandstetter C, Mohr LK, Latz E, Holz FG, Krohne TU (2015) Light induces NLRP3 inflammasome activation in retinal pigment epithelial cells via lipofuscin-mediated photooxidative damage. J Mol Med (Berl) 93: 905-916.
-
Stringham JM. Macular carotenoids, psychological stress and general health study in young adults. Eur J Ophthalmol 25: e72.
-
Stahl W, Sies H (2002) Carotenoids and protection against solar UV radiation. Skin Pharmacol Appl Skin Physiol 15: 291-296.
-
Stahl W, Sies H (2003) Antioxidant activity of carotenoids. Mol Aspects Med 24: 345-351.
-
Sies H, Stahl W (2003) Non-nutritive bioactive constituents of plants: lycopene, lutein and zeaxanthin. Int J Vitam Nutr Res 73: 95-100.
-
Loane E, Kelliher C, Beatty S, Nolan JM (2008) The rationale and evidence base for a protective role of macular pigment in age-related maculopathy. Br J Ophthalmol 92: 1163-1168.
-
Juturu V, Deshpande J, Ghanam K, Doyle L (2015) Soluble lutein inhibits cholinesterase and reduces ultraviolet radiation induced inflammation and immunosuppression: In vitro model. Eur J Ophthalmol 25: e63.
-
Meinke MC, Friedrich A, Tscherch K, Haag SF, Darvin ME, et al. (2013) Influence of dietary carotenoids on radical scavenging capacity of the skin and skin lipids. Eur J Pharm Biopharm 84: 365-373.
-
Gao S, Qin T, Liu Z, Caceres MA, Ronchi CF, et al. (2011) Lutein and zeaxanthin supplementation reduces H2O2-induced oxidative damage in human lens epithelial cells. Mol Vis 17: 3180-3190.
-
Gong X, Rubin LP (2015) Role of macular xanthophylls in prevention of common neovascular retinopathies: retinopathy of prematurity and diabetic retinopathy. Arch Biochem Biophys 572: 40-48.
-
Bian Q, Gao S, Zhou J, Qin J, Taylor A, et al. (2012) Lutein and zeaxanthin supplementation reduces photooxidative damage and modulates the expression of inflammation-related genes in retinal pigment epithelial cells. Free Radic Biol Med 53: 1298-1307.
-
Santocono M, Zurria M, Berrettini M, Fedeli D, Falcioni G (2006) Influence of astaxanthin, zeaxanthin and lutein on DNA damage and repair in UVA-irradiated cells. J Photochem Photobiol B 85: 205-215.
-
Loewen PC, Switala J, Triggs-Raine BL (1985) Catalases HPI and HPII in Escherichia coli are induced independently. Arch. Biochem. Biophys 243: 144-149.
-
Ding LH, Xie Y, Park S, Xiao G, Story MD (2008) Enhanced identification and biological validation of differential gene expression via Illumina whole-genome expression arrays through the use of the model-based background correction methodology. Nucleic Acids Res 36: e58.
-
Cruickshanks KJ, Klein R, Klein BE (1993) Sunlight and age-related macular degeneration. The Beaver Dam Eye Study. Arch Ophthalmol 111: 514-518.
-
Tomany SC, Cruickshanks KJ, Klein R, Klein BE, Knudtson MD (2004) Sunlight and the 10-year incidence of age-related maculopathy: the Beaver Dam Eye Study. Arch Ophthalmol 122: 750-757.
-
Cruickshanks KJ, Klein R, Klein BE, Nondahl DM (2001) Sunlight and the 5-year incidence of early age-related maculopathy: the beaver dam eye study. Arch Ophthalmol 119: 246-250.
-
Klein R, Klein BE, Moss SE, Cruickshanks KJ (1998) The Wisconsin Epidemiologic Study of Diabetic Retinopathy: XVII. The 14-year incidence and progression of diabetic retinopathy and associated risk factors in type 1 diabetes. Ophthalmology 105: 1801-1815.
-
Roberts RL, Green J, Lewis B (2009) Lutein and zeaxanthin in eye and skin health. Clin Dermatol 27: 195-201.
-
Alves-Rodrigues A1, Shao A (2004) The science behind lutein. Toxicol Lett 150: 57-83.
-
Krinsky NI, Landrum JT, Bone RA (2003) Biologic mechanisms of the protective role of lutein and zeaxanthin in the eye. Annu Rev Nutr 23: 171-201.
-
Kijlstra A, Tian Y, Kelly ER, Berendschot TT (2012) Lutein: more than just a filter for blue light. Prog Retin Eye Res 31: 303-315.
-
Yildirim Z, Yildirim F, Ucgun NI, Kilic N (2009) The evaluation of the oxidative stress parameters in nondiabetic and diabetic senile cataract patients. Biol Trace Elem Res 128: 135-143.
-
Spector A (1984) The search for a solution to senile cataracts. Proctor lecture. Invest Ophthalmol Vis Sci 25: 130-146.
-
Chandrasena LG, Chackrewarthy S, Perera PT, de Silva D (2006) Erythrocyte antioxidant enzymes in patients with cataract. Ann Clin Lab Sci 36: 201-204.
-
Nolan JM, Loskutova E, Howard A, Mulcahy R, Moran R, et al. (2015) The impact of supplemental macular carotenoids in Alzheimer's disease: a randomized clinical trial. J Alzheimers Dis 44: 1157-1169.
-
Nolan JM, Loskutova E, Howard AN, Moran R, Mulcahy R, et al. (2014) Macular pigment, visual function, and macular disease among subjects with Alzheimer's disease: an exploratory study. J Alzheimers Dis 42: 1191-1202.
-
Nechifor MT, Niculite CM, Urs AO, Regalia T, Mocanu M, et al. (2012) UVA irradiation of dysplastic keratinocytes: oxidative damage versus antioxidant defense. Int J Mol Sci 13: 16718-16736.
-
Kim KW, Ha KY, Lee JS, Rhyu KW, An HS, et al. (2007) The apoptotic effects of oxidative stress and antiapoptotic effects of caspase inhibitors on rat notochordal cells. Spine (Phila Pa 1976) 32: 2443-2448.
-
Ott M, Gogvadze V, Orrenius S, Zhivotovsky B (2007) Mitochondria, oxidative stress and cell death. Apoptosis 12: 913-922.
-
Delcourt C, Carriere I, Delage M, Barberger-Gateau P, Schalch W (2006) Plasma lutein and zeaxanthin and other carotenoids as modifiable risk factors for age-related maculopathy and cataract: the POLA Study. Invest Ophthalmol Vis Sci 47: 2329-2335.
-
Snellen EL, Verbeek AL, Van Den Hoogen GW, Cruysberg JR, Hoyng CB (2002) Neovascular age-related macular degeneration and its relationship to antioxidant intake. Acta Ophthalmol Scand 80: 368-371.
-
Age-Related Eye Disease Study 2 (AREDS2) Research Group, Chew EY1, Clemons TE2, Sangiovanni JP3, Danis RP4, et al. (2014) Secondary analyses of the effects of lutein/zeaxanthin on age-related macular degeneration progression: AREDS2 report No. 3. JAMA Ophthalmol 132: 142-149.
-
Pauleikhoff D, van Kuijk FJ, Bird AC (2001) Macular pigment and age-related macular degeneration. Ophthalmologe 98: 511-519.
-
Moreno FS, Toledo LP, de Conti A, Heidor R, Jordao A, et al. (2007) Lutein presents suppressing but not blocking chemopreventive activity during diethylnitrosamine-induced hepatocarcinogenesis and this involves inhibition of DNA damage. Chem Biol Interact 168: 221-228.
-
Wang Y, King JM, Xu Z, Losso J, Prudente A (2008) Lutein from ozone-treated corn retains antimutagenic properties. J Agric Food Chem 56: 7942-7949.
-
Serpeloni JM, Grotto D, Mercadante AZ, de Lourdes Pires Bianchi M, Antunes LM (2010) Lutein improves antioxidant defense in vivo and protects against DNA damage and chromosome instability induced by cisplatin. Arch Toxicol 84: 811-822.
-
Sindhu ER, Firdous AP, Preethi KC, Kuttan R (2010) Carotenoid lutein protects rats from paracetamol-, carbon tetrachloride- and ethanol-induced hepatic damage. J Pharm Pharmacol 62:1054-1060.
-
He RR, Tsoi B, Lan F, Yao N, Yao XS, Kurihara H (2011) Antioxidant properties of lutein contribute to the protection against lipopolysaccharide-induced uveitis in mice. Chin Med 6: 38.
-
Juturu V, Sahin, K, Sahin, N, Tuzcu, M, Yilmaz, I, et al. (2015) Mesozeaxanthin protects retina from oxidative stress in a rat model. AVRO Annual Meeting.
-
Almasieh M, MacIntyre JN, Pouliot M, Casanova C, Vaucher E, (2013) Acetylcholinesterase inhibition promotes retinal vasoprotection and increases ocular blood flow in experimental glaucoma. Invest Ophthalmol Vis Sci 54: 3171-3178.