International Journal of Ophthalmology and Clinical Research
Clinical Evidence of Ambient Temperature Variations on Femtosecond Laser Performance and Comparison with a Mechanical Microkeratome
Bojan Pajic1,2,3,4, Brigitte Pajic-Eggspuehler1, Iraklis Vastardis1, Michael Mrochen1,5 and Zeljka Cvejic2
1Swiss Eye Research Foundation - Eye Clinic ORASIS, Reinach AG, Switzerland
2Departments of Physics, Faculty of Sciences, University of Novi Sad, Serbia
3Medical Faculty, Military Medical Academy, University of Defense, Belgrade, Serbia
4Division of Ophthalmology, Department of Clinical Neurosciences, Geneva University Hospitals, Switzerland
5IROC Science, Technoparkstr. 1, Zürich, Switzerland
*Corresponding author: Bojan Pajic, MD, PhD, FEBO, Swiss Eye Research Foundation, Eye Clinic ORASIS, Reinach AG, Switzerland, Tel: +41 62 765 60 80, Fax: +41 62 765 60 81, E-mail: bpajic@datacomm.ch
Int J Ophthalmol Clin Res, IJOCR-2-043, (Volume 2, Issue 6), Research Article; ISSN: 2378-346X
Received: October 21, 2015 | Accepted: December 12, 2015 | Published: December 15, 2015
Citation: Pajic B, Pajic-Eggspuehler B, Vastardis I, Mrochen M, Cvejic Z (2015) Clinical Evidence of Ambient Temperature Variations on Femtosecond Laser Performance and Comparison with a Mechanical Microkeratome. Int J Ophthalmol Clin Res 2:043. 10.23937/2378-346X/1410043
Copyright: © 2015 Pajic B, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Purpose: To assess the consistency of flaps created by a femtosecond laser compared to a mechanical microkeratome as part of LASIK procedure.
Methods: Clinical outcomes were analyzed in 652 myopic eyes after LASIK surgery. Minimum follow-up time was 6 months. Mean age of patients was 35 ± 8.6 years (median: 34 years). Pre-operative mean refractive spherical equivalent was -3.71 ± 2.68 in the femtosecond laser group (range -9.75 to -1.50 D) and -3.40 ± 2.56 in the mechanical microkeratome group (range -9.88 to -1.25 D). Flaps were created either using a 60 kHz IntraLase Femtosecond Laser, with an intended flap thickness of 120 μm, or with a Ziemer Amadeus II microkeratome equipped with 140 μm head. Pre- and postoperative assessment was performed using confocal corneal laser-scanning microscopy. For all procedures the same excimer laser was used.
Results: Mean visual acuity comparing baseline and first day after surgery improved in both groups significantly and remained stable over the follow-up period of 6 months. Difference in mean visual acuity was not statistically significant between the groups during the follow up period (p > 0.05). The mean flap thickness in the femtosecond laser group was 119 ± 4.6 μm (range 111 to 129 μm) and 143 ± 21.9 μm (range 93 to 178 μm) in the microkeratome group.
Conclusions: Creation of LASIK flaps using a femtosecond laser provides a higher degree of precision and safety during the procedure but shows more inhomogeneity in the cut quality related to a temperature fluctuation in the surgery room.
Introduction
The mechanism of flap creation with the IntraLase femtosecond laser has already been described extensively in the literature [1-5] over the years. Femtosecond laser systems produce energy pulses (600 to 800 femtosecond) at an infrared wavelength of 1053 nm. The ultra-short impulses are focused at a spot size of a few micrometers in diameter at a preset depth within the stroma, leading to photo disruption and to a creation of a corneal resection plane. Once the flap has been created, the device creates a side cut at the level of the resection plane, moving anteriorly through the corneal stroma and epithelium. Any cavitation bubbles created during the photo disruption generally remain at the resection plane and disappear after several minutes.
The initial experience in the United States with the IntraLase FS laser began in May 2000 and showed excellent results with no postoperative complications [2,3]. Meanwhile, several studies showed an improved flap predictability and refractive outcomes compared with that of the mechanical microkeratome [6-8]. The use of this technology helped to reduce or eliminate many of the minor or even severe complications associated with the flap creation [2,4,6-9].
However, the question remains whether there are definitive clinical benefits of using a femtosecond laser over a mechanical system for corneal flap creation. In an attempt to answer this question, a prospective clinical study was carried out to investigate the predictability of the flap thickness. The analysis included the evaluation of the corneal endothelial cell loss and of all refractive outcomes for both groups.
Methods and Patients
In this study, corneal flaps created from 60 kHz femtosecond laser (IntraLase FS Laser, AMO, Abbott Park, Ill, USA) were compared to those created from a mechanical microkeratome (Amadeus II, Ziemer Group, Port, Switzerland). A total of 651 myopic eyes were selected for the procedure with 311 patients formed the femtosecond laser group and 341 patients in the microkeratome group (Table 1). In patients where bilateral treatment was performed, only data from the right eyes were used. The mean age of patients in the femtosecond laser group was 35 ± 8.5 years (range: 20-60 years) (median: 35) and 35 ± 8.6 years (range: 21-62 years) (median: 35) for the microkeratome group. There was no statistically significant difference in age (p = 0.41) between the two groups. One-hundred-sixty-two patients (52.1%) in the femtolaser group and 187 patients (54.8%) in the microkeratome were male. There was no statistically significant difference regarding the sex in each group or between the two groups (p > 0.05). The preoperative mean refractive spherical equivalent was -3.71 ± 2.68 D in the femtosecond laser group (range -9.75 to -1.50 D) and -3.40 ± 2.56 D in the microkeratome group (range -9.88 to -1.25 D). The mean astigmatic values were -0.99 D in the femtosecond group (range -5.00 to 0.00 D) and -1.07 D in the microkeratome group (range -6.00 to -0.50 D). There was no significant difference between the groups regarding the mean preoperative spherical equivalent or astigmatism (p > 0.05). All contact lens use (soft and rigid gas permeable) was terminated 2 weeks before preoperative evaluation began.
![]()
Table 1: Preoperative characteristics of two groups.
View Table 1
For all patients enrolled, a complete ophthalmological examination was performed including the determination of uncorrected distal visual acuity (UDVA), best corrected refraction, best-corrected distal visual acuity (CDVA), cycloplegic refraction, intraocular pressure (IOP) measurement, corneal topography and pachymetry, and slit lamp examination to assess any apparent pathology of the eye. The OCP uses 1310 nm wavelength light waves, which allows noncontact online measurements of pachymetry before, during and after excimer laser procedure. For measurement, the eye is illuminated with low coherent light of a super-luminescence diode. This light is reflected and scattered on the surface of the eye and at the posterior corneal surface at the interface of the cornea and aqueous humor. In addition to the light beam directed onto the cornea, a second parallel light beam with a changeable length of path is produced, which is a reference beam. In the interferometer, the reflected beam and reference beam are superposed. The superimposed beams are detected in a photo diode and converted into an electronic signal it is said in an interference pattern. This electronic signal is evaluated with respect to the length of path of the reference beam and the travel time. Using the refractive index of the tissue, the pachymetry is calculated. Use of online optical coherence pachymetry (OCP) served to obtain pachymetric values for all patients before surgery and after the creation of the flap. After surgery corneal morphology and pachymetry were analyzed using confocal laser microscopy. All patients were evaluated preoperatively, at 1 day, 1 week and 1, 3 and 6 months after surgery.
All flaps created with the femtosecond laser were designed to deliver equivalent geometric dimensions 120 μm flap thickness, 8.5 mm flap diameter, 45-degree superior hinge angle. In order to achieve equivalent corneal stromal surface exposure, a 70-degree side-cut angle with a laser raster pattern spot/line separation of 8/8 m with a 60 KHz repetition rate, as well as a stromal energy of 0.85 μJ with a side-cut energy of 0.85 μJ was used. For the microkeratome group, flaps were created using a 140 μm head and 8.5 or 9.5 mm ring depending on the corneal radius and the manufacturer's recommendations.
Ablation was performed using the Technolas 217P100 excimer laser (Bausch & Lomb, Rochester, NY, USA). Surgery was based on the patients' manifest refraction using our predefined nomogram adjustment for all eyes. The optical treatment zones were 6.0 to 7.0 mm depending of the scotopic pupil diameter it is said the optical treatment zone was 0.5 mm larger than the scotopic pupil diameter. The same optical zone diameter was used for the patients receiving bilateral surgery.
Laser-scanning microscopy and pachymetry
Corneal confocal digital in vivo laser microscopy was performed using the Heidelberg Retina Tomograph (HRT II. Heidelberg Engineering GmbH, Germany) selected as the basic device for a digital confocal corneal laser-scanning microscope. During the laser-scanning ophthalmoscopy of the anterior segment a high-quality microscope lens was used to provide a laser focus of 1 μm in diameter. The system provides a high-resolution, high-speed, digital confocal image of the cornea. All images were acquired by continuous through-focusing [10].
This technique allows a rapid and reliable visualization and evaluation of all the microstructures of the cornea, including the epithelium, the corneal nerves and the keratocytes, as well as the endothelium and bulbar conjunctiva [11]. Flap thickness was measured by determining the distance between the surface epithelium and the interface structure of the image.
Results
Visual acuity
The mean visual acuity improved significantly from baseline to 1 day after surgery and remained stable over the follow-up period in both groups. No differences were found between the groups during the follow up (p > 0.05). In the femtosecond laser group, 94% (292 eyes) of all treated eyes were within ± 0.5D of the intended manifest spherical equivalent refraction at the first month, improving to 95% (295 eyes) at the third month and 97% (302 eyes) at the sixth month. In the microkeratome group, at first month 96% (327 eyes) of all treated eyes were within ± 0.5D, 97 %, (331 eyes) at third month and 95% (324 eyes) at the sixth month.
Flap thickness
Predictability of the flap thickness was found to be more precise for the femtosecond laser group compared to the microkeratome group. The mean flap thickness in the femtosecond laser group was 119 ± 4.6 μm (range 111 to 129 μm). In the microkeratome group thickness were found to be 143 ± 21.9 μm (range 93 to 178 μm) (Figure 1). The difference between intended flap thickness and measured flap thickness was 4.1 ± 2.56 μm (range 0 to 10 μm) in the femtosecond laser group and 18.1 ± 11.78 μm (range 1 to 47 μm) in the microkeratome group. The difference between intended and measured flap thickness was significantly lower for the femtosecond laser group (p < 0.001).
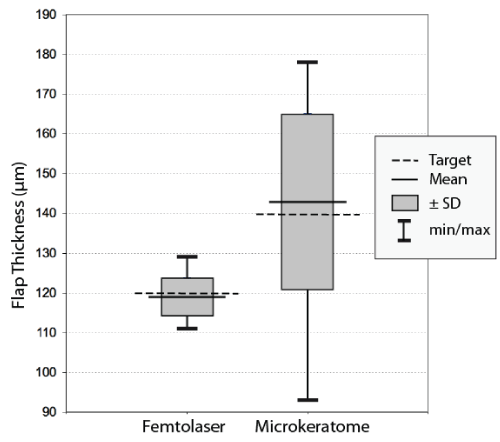
.
Figure 1: Central flap thickness in femtosecond laser and microkeratome groups following LASIK, measured by Optical Coherence Pachymetry. (Dotted lines: Target; solid horizontal lines: mean result; solid vertical lines: maximum/minimum values; boxes: range representing ± 1 standard deviation).
View Figure 1
Safety
No differences regarding safety were found between the two groups. Results of measured BCVA and line gain or loss are listed in table 2. In the femtosecond laser group no eye demonstrated any loss of line during the time interval of assessment. In the microkeratome group, some line loss was found for all time periods recorded (Figure 2); however the visual acuity results and the number of eyes losing or gaining lines was not or different between the groups at any time during follow-up (p = 0.36).
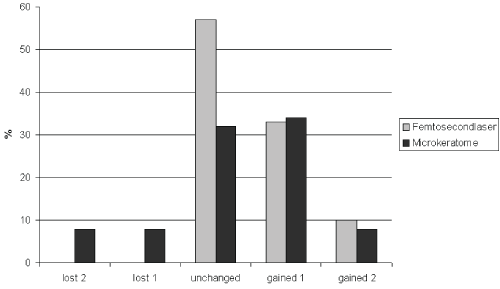
.
Figure 2: Changes in Corrected Distance Vision Visual Acuity (CDVA) in femtosecond laser and microkeratome groups after 6 months.
View Figure 2
![]()
Table 2: Summary of CDVA and line loss or gain for the time period from 1st to 6th month after surgery for both groups of the study.
View Table 2
Complications
No statistical differences were found in complication rates between the two groups. Complications were observed in the femtosecond group in 6 cases. 3 eyes developed striae and 3 eyes diffuse lamellar keratitis was observed. Complications in the microkeratome group occurred in 10 cases: 4 eyes developed striae, in 3 eyes a diffuse lamellar keratitis and 1 occurrence each of epithelial ingrowth, flap dislocation and an incomplete flap. In both groups the diffuse lamellar keratitis was treated with Dexamethason eye drops. The inflammation was disappeared within 5 days. Regarding the striae, only in two cases, one in each group, a flap re-lifting with hydration and consecutive PTK was necessary. In both cases the complaint disappears completely. The epithelial ingrowth was removed mechanically while the flap dislocation was re-lifted and put it again in the right position.
Confocal microscopy analysis
The preoperative corneal images were captured from a depth equivalent to the postoperative interface. Comparing both groups the postoperative images were focused to the level of the intracorneal interface. Corneal images in both groups did not reveal any significant signs of keratocyte activation. A more extensive degree of corneal edema was visible in the interface in the microkeratome group with a mean of 9.2 ± 4.9 μm at the first postoperative day (Figure 3A) in comparison to the femtolaser group showing a mean of 4.4 ± 3.7 μm, (Figure 3B). This difference was statistically significant in terms of edema thickness (P < 0.001).
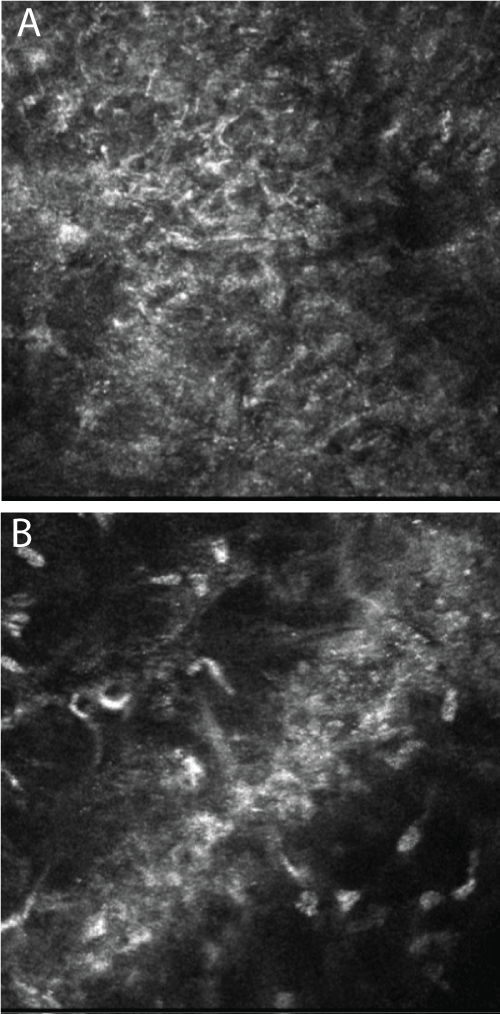
.
Figure 3: Confocal micrograph of edema in the corneal interface. (A) Amadeus microkeratome group; (B) femtosecond laser group.
View Figure 3
Room temperature
The focus stability of the IntraLase femtosecond laser is strongly dependent on the room temperature. For each procedure the room temperature was noted. There was temperature fluctuation during a surgery day thus there was possible to detect the differences between different surgery room temperature. An interesting finding of this study was the fact that the external (surgery room) temperature also had an impact on the delivery of femtosecond laser energy to the tissue. With increasing room temperature the femtosecond laser spot seems to tend to lose homogeneity. Thus the confocal laser microscopy analysis revealed a more extended interface edema when the femtosecond laser was applied at 22°C (Figure 4A) compared to 19°C of room temperature (Figure 4B).
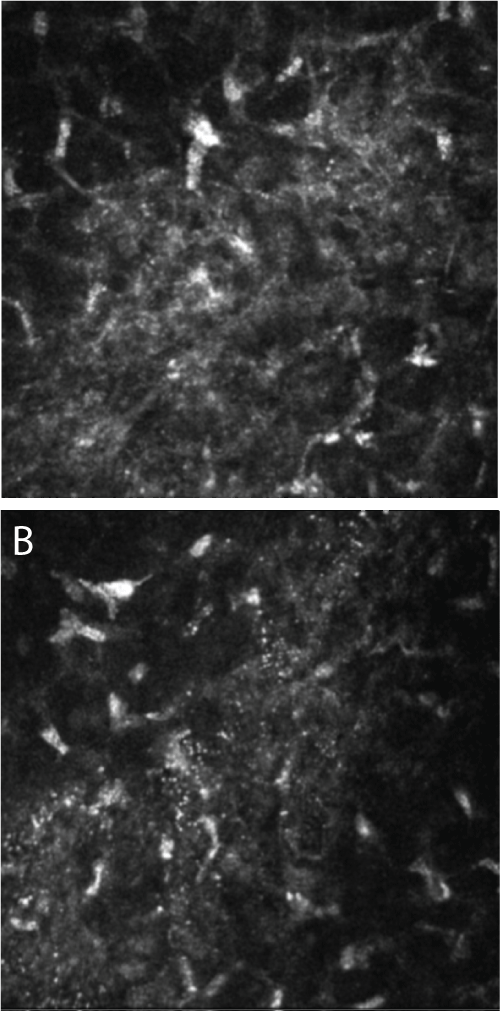
.
Figure 4: Confocal micrograph of edema in the corneal interface after femtosecond laser surgery. (A) Procedure performed at 22°C room temperature; (B) procedure performed at 19°C room temperature.
View Figure 4
Suction time
Mean suction time was 61.8 ± 3.6 seconds in the femtosecond laser group and 24.7 ± 2.1 seconds in the microkeratome group. The difference in suction time was statistically significant (p < 0.001). A longer suction time was required for the femtosecond laser.
Discussion
This prospective study compared surgery outcomes obtained by using two devices for flap creation, the Intralase FS Femtosecond Laser and the standard compression head Ziemer Amadeus microkeratome. The flap creation with the femtosecond laser showed a higher predictability and reproducibility when compared to the mechanical microkeratome. Both methods had low and comparable complication rates. A further advantage of the femtosecond laser compared with the microkeratome was that the precision and safety of the corneal incision was independent of the preoperative corneal curvature. The interface edema has a higher degree in the microkeratome than in the femtosecond laser group. The hypothesis is, that the cutting procedure of the microkeratome with a steel blade is more invasive that the femtosecond laser, what leads to a higher liquid accumulation in the extra cellular matrix space, it is said to a higher interface edema.
An interesting correlation was found between the room temperature and the amount of interface edema. As evidenced by confocal microscopy of the cornea after surgery, the femtosecond laser generates more extensive edema at higher room temperatures. The difference of the edema thickness when the flaps were created using both methods was found to be statistically significant. The reason is theorized to be associated with deterioration of homogeneity of the focal spot size outside the recommended surgery room temperature which is at 19°C. An increase in the focal spot size due to reduced beam quality results in a lower probability and stability for an optical breakdown. As a consequence the resulting disruptive effect might vary from pulse to pulse leading to an incomplete resection plane. More bridges between the stromal bed and the flap can occur and this might require a more powerful manual manipulation within the interface, resulting in a higher stress on the flap necessary to separate it from the corneal bed and thus creating a consecutive higher edema. Based on our findings we propose that the ideal operating room temperature should be between 17° and 22°C. This is in our knowledge the first description of variations in the effect of femtosecond laser application related to the operation room temperature. On the other side the temperature has only a very small impact on the steel blade of the microkeratome, thus there was not detected an edema thickness fluctuation in the interface as seen with the femtosecond laser. It seems that the temperature has not an influence on the microkeratome procedure regarding the cut quality and consecutively interface edema.
The IntraLase femtosecond laser is capable of consistently creating thinner corneal flaps with a lower standard deviation of flap thickness compared to the mechanical microkeratome. The femtosecond laser therefore offers the surgeon an opportunity to preserve a thicker residual stromal bed. The edge angle of the flap in the Amadeus group is a reciprocal function of the corneal diameter and corneal curvature, due to the design of the microkeratome. In contrast, the Intralase femtosecond laser [12] generates a fixed sidecut. Confocal microscopy revealed less postoperative edema after the femtosecond laser procedure. In contrast to other studies [13], we did not find a difference between the two groups relative to activation of keratocytes in the early postoperative period. The reason for this difference could be that the 60 kHz laser used in our study generates a smoother interface compared to the 15 kHz model used in the other study [13]. Unlike a mechanical microkeratome that does not perform the lamellar keratectomy under the hinge of the flap, the femtosecond laser creates a 360-degree lamellar dissection including the corneal area under the hinge. In addition, the femtosecond laser creates a lamellar resection of uniform thickness. This type of resection may produce a more symmetrical biomechanical response, with reduced potential effects due to variations in flap size or location. A small, predictable edge may also be important in maximizing stromal exposure for excimer laser ablation thus reducing any asymmetry during ablation [8].
Modern femtosecond laser systems with a higher pulse frequency, lower pulse energy and a shorter treatment time have meanwhile become available. The bubble formation caused by photodisruption can be significantly reduced in newer femtosecond lasers, leading to smoother cutting surfaces. The IntraLase has introduced a unit with a pulse frequency of 60 kHz that can reduce the treatment time to less than 25 seconds per eye. Regarding the intraocular perfusion the suction time is important, which is significantly longer than that with the mechanical microkeratome. Nevertheless we did not see any damage to the optic nerve or any evidence of retinal pathology due to the procedures in either group.
Conclusion
Based on the results of this study the femtosecond laser provides the refractive surgeon with a high degree of precision and safety. All flaps created with this laser system showed a high degree of confidence in relation to flap thickness predictability. This is of clinical relevance particularly for high myopic and thin cornea patients. A statistical difference between the two groups in this study in terms of visual acuity, predictability and safety was not found.
An increase to more than 22°C in the operating room during the procedure may lead to loss of spot homogeneity in the IntraLase femtosecond laser.
References
-
Kurtz RM, Liu X, Elner VM, Squier JA, Du D, et al. (1997) Photodisruption in the human cornea as a function of laser pulse width. J Refract Surg 13: 653-658.
-
Nordan LT, Slade SG, Baker RN, Suarez C, Juhasz T, et al. (2003) Femtosecond laser flap creation for laser in situ keratomileusis: six-month follow-up of initial U.S. clinical series. J Refract Surg 19: 8-14.
-
Ratkay-Traub I, Juhasz T, Horvath C, Suarez C, Kiss K, et al. (2001) Ultra-short pulse (femtosecond) laser surgery: initial use in LASIK flap creation. Ophthalmol Clin North Am 14: 347-355.
-
Ratkay-Traub I, Ferincz IE, Juhasz T, Ron MK, Ronald RK (2003) First dinical results with the femtosecond neodynium-glass laser in refractive surgery. J Refract Surg 19: 94-103.
-
Sugar A (2002) Ultrafast (femtosecond) laser refractive surgery. Curr Opin Ophthalmol 13: 246-249.
-
Binder PS (2004) Flap dimensions created with the IntraLase FS laser. J Cataract Refract Surg 30: 26-32.
-
Tran DB, Sarayba MA, Bor Z, Garufis C, Duh YJ, et al. (2005) Randomized prospective clinical study comparing induced aberrations with IntraLase and Hansatome flap creation in fellow eyes: potential impact on wavefront-guided laser in situ keratomileusis. J Cataract Refract Surg 31: 97-105.
-
Kezirian GM, Stonecipher KG (2004) Comparison of the IntraLase femtosecond laser and mechanical keratomes for laser in situ keratomileusis. J Cataract Refract Surg 30: 804-811.
-
Anderson NJ, Edelhauser HF, Sharara N, Thompson KP, Rubinfeld RS, et al. (2002) Histologic and ultrastructural findings in human corneas after successful laser in situ keratomileusis. Arch Ophthalmol 120: 288-293.
-
Guthoff RF, Stave J (2006) Essentials in Ophthalmology Cornea AND External Eye Disease. In: Hrsg. Reinhard Th, Larkin F (eds) In Vivo Micromorphology of the Cornea: Confocal Microscopy Principles and Clinical Applications, Springer-Verlag Berlin Heidelberg, 173-208.
-
Suzuki T, Sano Y, Kinoshita S (2000) Conjunctival inflammation induces Langerhans cell migration into the cornea. Curr Eye Res 21: 530-533.
-
Lin RT, Lu S, Wang LL, Kim ES, Bradley J (2003) Safety of laser in situ keratomileusis performed under ultra-thin corneal flaps. J Refract Surg 19: S231-236.
-
Porter J, MacRae S, Yoon G, Roberts C, Cox IG, et al. (2003) Separate effects of the microkeratome incision and laser ablation on the eye's wave aberration. Am J Ophthalmol 136: 327-337.





